Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Pharmaceutical Intermediates
The rapid evolution of medicinal chemistry demands robust, safe, and scalable synthetic methodologies for constructing complex heterocyclic scaffolds. A pivotal advancement in this domain is detailed in patent CN113121462B, which discloses a highly efficient preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. These structures are critical pharmacophores found in numerous bioactive molecules, ranging from beta-3 adrenergic receptor agonists to agrochemical fungicides. The introduction of the trifluoromethyl group significantly enhances metabolic stability and lipophilicity, yet its installation has historically been fraught with safety and cost challenges. This patent presents a transformative approach that bypasses the limitations of conventional transition-metal catalysis and hazardous azide chemistry, offering a streamlined pathway for the production of high-purity pharmaceutical intermediates.
For R&D directors and process chemists, the significance of this technology lies in its operational simplicity and broad substrate scope. By utilizing readily available trifluoroethylimidoyl chlorides and diazo compounds, the method achieves high reaction efficiency under mild conditions. This eliminates the dependency on expensive copper catalysts and the rigorous safety protocols required for handling explosive organic azides. Consequently, this innovation not only accelerates the drug discovery timeline but also provides a viable route for commercial scale-up, addressing the critical need for reliable suppliers of complex fluorinated heterocycles in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on two primary strategies, both of which present significant drawbacks for industrial application. The first involves copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, followed by trifluoromethylation. This route is inherently problematic due to the use of toxic copper salts, which necessitate extensive downstream purification to meet stringent residual metal limits in pharmaceutical products. Furthermore, the reliance on organic azides introduces severe safety risks, as these compounds are known to be thermally unstable and potentially explosive, complicating storage and handling on a large scale.
The second conventional approach utilizes organocatalytic 1,3-dipolar cycloaddition between azides and trifluoromethyl ketones. While this avoids transition metals, it retains the fundamental safety hazard of using azide reagents. Additionally, these traditional methods often suffer from limited functional group tolerance and require harsh reaction conditions or specialized trifluoromethylating reagents that drive up raw material costs. For procurement managers, these factors translate into higher COGS (Cost of Goods Sold) and increased liability regarding process safety and environmental compliance, making the search for alternative synthetic routes a top priority.
The Novel Approach
In stark contrast to legacy technologies, the method described in CN113121462B employs a base-promoted cyclization strategy that completely circumvents the use of metals and azides. As illustrated in the reaction scheme below, the process couples trifluoroethylimidoyl chloride with diazo compounds in the presence of cesium carbonate. This metal-free paradigm shift drastically simplifies the workup procedure, as there is no need for expensive metal scavengers or complex extraction protocols to remove heavy metal residues.
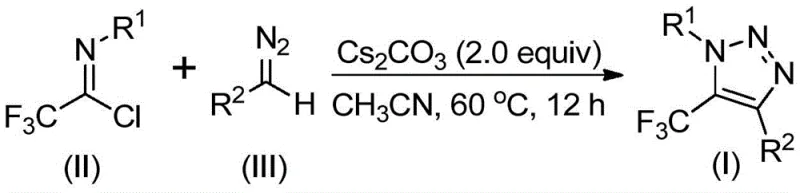
The novelty of this approach extends to its exceptional versatility. The reaction tolerates a wide array of substituents on both the nitrogen and carbon frameworks, allowing for the synthesis of diverse derivatives including esters, phosphonates, and ketones. By operating at moderate temperatures (50-70°C) in common solvents like acetonitrile, the process is inherently safer and more energy-efficient. This represents a substantial leap forward in process chemistry, enabling the cost reduction in pharmaceutical intermediate manufacturing by utilizing cheap, commercially available starting materials and minimizing waste generation.
Mechanistic Insights into Base-Promoted Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing reaction parameters and ensuring reproducibility at scale. The proposed mechanism involves a base-promoted intermolecular nucleophilic addition-elimination sequence. Initially, the diazo compound acts as a nucleophile, attacking the electrophilic carbon of the trifluoroethylimidoyl chloride. This step is facilitated by the cesium carbonate base, which likely assists in deprotonation or activation of the diazo species, promoting the formation of a key carbon-carbon bond while eliminating a chloride ion.
Following the initial coupling, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This ring-closing step constructs the characteristic five-membered triazole core, driven by the electronic properties of the trifluoromethyl group and the stability of the resulting aromatic system. The absence of transition metals suggests that the cyclization proceeds through a purely polar mechanism rather than a radical or organometallic pathway. This mechanistic clarity allows chemists to fine-tune the electronic nature of the substrates to enhance reaction rates and yields, ensuring consistent quality for high-purity OLED material or API precursors.
Furthermore, the choice of base and solvent plays a pivotal role in controlling the impurity profile. Cesium carbonate is identified as the optimal promoter due to its solubility characteristics and basicity, which effectively drive the reaction to completion without inducing side reactions such as diazo decomposition. The use of aprotic solvents like acetonitrile ensures that the reactive intermediates remain stable throughout the 8 to 16-hour reaction window. This level of control over the reaction environment is essential for reducing lead time for high-purity intermediates, as it minimizes the formation of difficult-to-remove byproducts.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot plant production. The protocol requires minimal specialized equipment, relying instead on standard glassware and heating mantles. The key to success lies in the precise stoichiometric balance of reagents and the maintenance of anhydrous conditions to prevent diazo hydrolysis. Detailed procedural guidelines ensure that operators can achieve reproducible results with yields comparable to those reported in the patent examples.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an aprotic organic solvent like acetonitrile.
- Heat the reaction mixture to 50-70°C and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via column chromatography to obtain the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this synthetic route offers compelling economic and logistical benefits. The elimination of transition metal catalysts removes a significant cost center associated with catalyst procurement and, more importantly, the downstream purification required to meet regulatory standards for residual metals. This simplification of the purification train directly contributes to substantial cost savings and shorter batch cycle times. Additionally, the avoidance of explosive azide reagents significantly lowers insurance premiums and reduces the regulatory burden associated with storing and transporting hazardous materials.
- Cost Reduction in Manufacturing: The utilization of inexpensive, commodity-grade starting materials such as trifluoroethylimidoyl chloride and cesium carbonate drastically lowers the raw material bill of materials. Unlike specialized trifluoromethylating reagents which can be prohibitively expensive, these precursors are widely available from multiple global suppliers, mitigating supply risk. Furthermore, the simplified workup procedure reduces solvent consumption and labor hours, leading to a leaner, more cost-effective manufacturing process that enhances overall margin potential.
- Enhanced Supply Chain Reliability: By removing dependency on scarce or regulated reagents like organic azides and copper catalysts, this method fortifies the supply chain against disruptions. The robustness of the reaction conditions means that production is less susceptible to variations in raw material quality or minor fluctuations in process parameters. This reliability ensures consistent delivery schedules for critical pharmaceutical intermediates, allowing downstream partners to maintain uninterrupted production of final drug substances without the fear of batch failures or safety incidents.
- Scalability and Environmental Compliance: The metal-free nature of this synthesis aligns perfectly with modern green chemistry principles and increasingly strict environmental regulations. The absence of heavy metals simplifies waste stream management and reduces the environmental footprint of the manufacturing process. The reaction is easily scalable from gram to kilogram quantities without the need for complex engineering controls, facilitating a smooth transition from R&D to commercial production. This scalability ensures that the technology can meet growing market demand for fluorinated heterocycles while maintaining compliance with global EHS standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: Why is this metal-free synthesis method safer than traditional Cu-catalyzed routes?
A: Traditional methods often rely on toxic and potentially explosive organic azides and copper catalysts. This novel protocol utilizes stable diazo compounds and imidoyl chlorides, eliminating the safety hazards associated with handling high-energy azide species and removing the need for costly heavy metal scavenging steps.
Q: What are the typical reaction conditions for scaling this process?
A: The process operates under mild thermal conditions, typically between 50°C and 70°C, using common aprotic solvents like acetonitrile. The use of inexpensive cesium carbonate as a promoter allows for straightforward scale-up without requiring specialized high-pressure or cryogenic equipment.
Q: Does this method support diverse functional groups for drug discovery?
A: Yes, the methodology demonstrates excellent functional group tolerance. It accommodates various substituents on both the imidoyl chloride and diazo components, including halogens, esters, phosphonates, and ketones, making it highly versatile for generating diverse libraries of bioactive intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN113121462B for accelerating drug development pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in fluorine chemistry positions us as a preferred partner for complex molecule synthesis.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce overall project costs.
