Advanced Synthesis of 5-Trifluoromethyl Triazoles: Enabling Commercial Scale Production for Pharmaceutical Applications
The patent CN113121462A introduces a groundbreaking methodology for synthesizing 5-trifluoromethyl substituted 1,2,3-triazole compounds, representing a significant advancement in the production of critical pharmaceutical intermediates. This innovative approach addresses longstanding challenges in heterocyclic chemistry by eliminating the need for toxic azides and transition metal catalysts that have plagued conventional synthesis routes. The process leverages readily available starting materials including cesium carbonate, molecular sieves, trifluoroethylimidoyl chloride, and diazo compounds, operating under remarkably mild conditions of 50–70°C for 8–16 hours in organic solvents. This breakthrough not only enhances reaction efficiency but also establishes a robust foundation for industrial-scale manufacturing of these biologically active compounds, which serve as essential building blocks in numerous therapeutic agents targeting human beta-3 adrenoceptors and other pharmacological pathways. The method's scalability from gram-level laboratory demonstrations to potential commercial production volumes offers substantial strategic advantages for pharmaceutical supply chains seeking reliable sources of high-purity intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
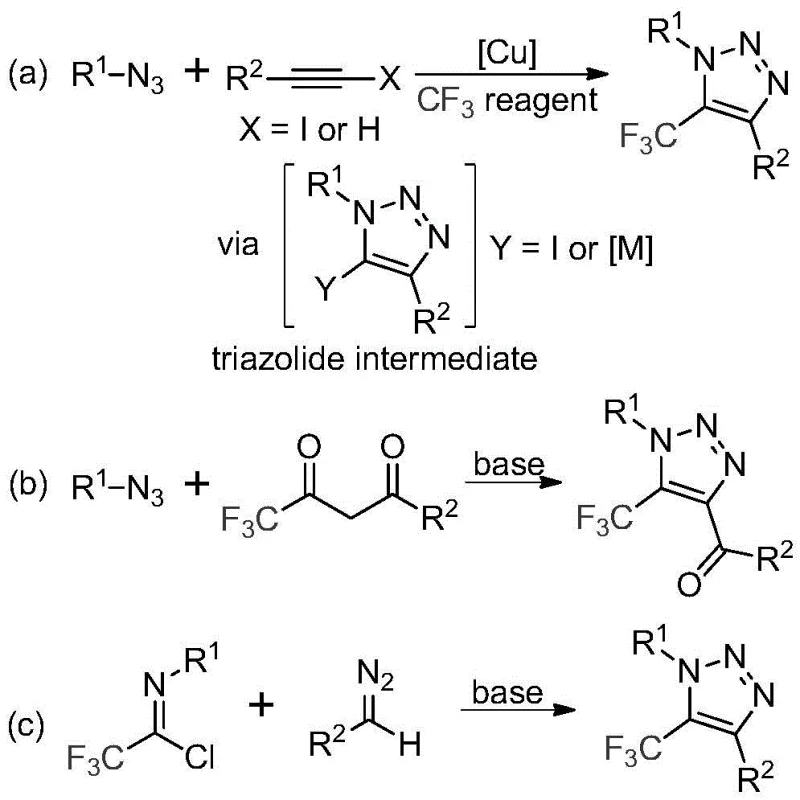
Traditional approaches for synthesizing trifluoromethyl-substituted triazoles have been severely constrained by multiple critical limitations that hinder commercial viability. The predominant copper-catalyzed [3+2] cycloaddition method requires toxic and explosive organic azides as starting materials, creating significant safety hazards and regulatory complications during manufacturing. These processes also necessitate expensive transition metal catalysts followed by complex purification steps to remove residual metals, substantially increasing production costs and complicating quality control procedures. Furthermore, the narrow substrate scope of conventional methods restricts structural diversity, limiting the ability to customize intermediates for specific pharmaceutical applications. The requirement for specialized equipment to handle hazardous azide compounds creates additional barriers to scale-up, while the multi-step nature of these processes results in lower overall yields and higher impurity profiles that fail to meet stringent pharmaceutical quality standards. These cumulative challenges have historically prevented cost-effective commercial production of these valuable heterocyclic compounds.
The Novel Approach
The patented methodology overcomes these limitations through an elegant metal-free cyclization process that utilizes stable and commercially available diazo compounds instead of hazardous azides. By employing cesium carbonate as a base promoter in acetonitrile solvent at moderate temperatures (60°C), the reaction achieves high efficiency without requiring transition metals or specialized safety protocols. This innovation eliminates the need for costly metal removal steps while maintaining excellent functional group tolerance across diverse R¹ and R² substituents. The process demonstrates remarkable scalability from laboratory to industrial production volumes while delivering consistent high-purity products suitable for pharmaceutical applications. Crucially, the simplified reaction pathway reduces both capital expenditure requirements and operational complexity, making it particularly attractive for contract manufacturing organizations seeking to establish reliable supply chains for critical intermediates. The method's compatibility with standard manufacturing equipment further enhances its commercial viability by minimizing facility modification costs.
Mechanistic Insights into Metal-Free Triazole Cyclization
The reaction mechanism proceeds through a well-defined sequence that begins with intermolecular nucleophilic addition between the diazo compound and trifluoroethylimidoyl chloride, facilitated by cesium carbonate as a base promoter. This initial step forms a key triazolide intermediate through elimination processes that avoid hazardous azide intermediates entirely. Subsequent intramolecular 5-endo-dig cyclization then occurs spontaneously under mild thermal conditions to generate the final triazole product with precise regioselectivity at the 5-position. The absence of transition metals prevents unwanted side reactions and eliminates concerns about metal contamination in the final product. Molecular sieves play a critical role in maintaining anhydrous conditions that prevent hydrolysis of sensitive intermediates while promoting high conversion rates. This mechanistic pathway represents a significant departure from conventional copper-catalyzed methods by fundamentally redesigning the reaction sequence to prioritize safety, simplicity, and purity.
Impurity control is achieved through multiple built-in mechanisms within this novel process design. The elimination of transition metals removes a major source of potential contaminants that would require extensive purification steps in traditional methods. The mild reaction conditions (60°C) prevent thermal degradation pathways that typically generate byproducts in higher-temperature processes. Careful selection of cesium carbonate as the base promoter minimizes unwanted side reactions compared to stronger bases that could cause decomposition of sensitive functional groups. The use of molecular sieves effectively controls moisture content, preventing hydrolysis of key intermediates that would otherwise lead to impurity formation. Furthermore, the reaction's high functional group tolerance allows precise control over substitution patterns without generating complex impurity profiles associated with less selective methods. This comprehensive impurity management strategy ensures consistent production of high-purity intermediates meeting pharmaceutical quality requirements.
How to Synthesize 5-Trifluoromethyl Triazoles Efficiently
This patented synthesis route represents a significant advancement in the production of trifluoromethyl-substituted triazole intermediates through its innovative metal-free approach that eliminates hazardous reagents while maintaining high efficiency. The process leverages commercially available starting materials and standard laboratory equipment to achieve consistent results across diverse substrate combinations. Detailed standardized synthesis steps are provided below to ensure reliable implementation in manufacturing environments while maintaining the high purity standards required for pharmaceutical applications.
- Combine trifluoroethylimidoyl chloride (II), diazo compound (III), and cesium carbonate in acetonitrile solvent under nitrogen atmosphere
- Maintain reaction temperature at 60°C for 12 hours with continuous stirring while monitoring conversion
- Perform post-processing through filtration, silica gel mixing, and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial strategic advantages for procurement and supply chain operations by addressing critical pain points in intermediate sourcing. The elimination of hazardous materials reduces regulatory compliance burdens while enhancing supply chain resilience through simplified logistics and reduced safety management requirements. The use of readily available starting materials creates multiple sourcing options that mitigate supply disruption risks, while the process's inherent scalability ensures consistent product availability across varying demand volumes. These combined benefits position this technology as a transformative solution for organizations seeking reliable access to high-quality triazole intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and hazardous azide handling protocols significantly reduces raw material costs while avoiding substantial expenditures associated with metal removal processes and specialized safety infrastructure. The simplified purification requirements stemming from cleaner reaction profiles further decrease processing costs without requiring additional capital investment in new equipment or facilities.
- Enhanced Supply Chain Reliability: Utilization of commercially available starting materials with multiple global suppliers creates robust sourcing options that minimize vulnerability to single-point failures. The process's compatibility with standard manufacturing equipment enables rapid technology transfer between facilities, ensuring consistent production capabilities across different geographic locations while maintaining product quality specifications.
- Scalability and Environmental Compliance: The demonstrated scalability from gram-level reactions to potential commercial volumes eliminates development risks associated with process scale-up. The absence of toxic reagents and heavy metals simplifies waste stream management while meeting increasingly stringent environmental regulations, reducing both operational complexity and compliance costs throughout the manufacturing lifecycle.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN113121462A's methodology and implementation requirements. These insights provide essential information for procurement teams evaluating this technology for integration into their supply chain strategies.
Q: How does this metal-free process improve impurity profile compared to conventional copper-catalyzed methods?
A: The elimination of transition metal catalysts prevents heavy metal contamination in the final product, significantly simplifying purification requirements and ensuring stringent purity specifications required for pharmaceutical intermediates. This directly addresses regulatory concerns regarding residual metal content in API manufacturing.
Q: What substrate flexibility does this method offer for custom intermediate production?
A: The process demonstrates broad functional group tolerance with diverse R¹ (alkyl/aryl) and R² (aroyl/phospholipid) substitutions, enabling tailored synthesis of specialized triazole derivatives. This adaptability supports customized intermediate production for specific drug development pipelines without process revalidation.
Q: How does the solvent system contribute to commercial scalability?
A: The use of acetonitrile as the preferred aprotic solvent provides optimal solubility for reactants while maintaining high conversion rates. Its industrial availability and established handling protocols facilitate seamless scale-up from laboratory to commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Triazole Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs and advanced analytical capabilities. As a specialized CDMO partner, we possess deep expertise in implementing this patented metal-free triazole synthesis methodology across multiple production scales, ensuring seamless technology transfer from laboratory development to full commercial manufacturing. Our dedicated technical teams work closely with clients to optimize process parameters for specific intermediate requirements while maintaining the highest quality standards demanded by global pharmaceutical regulators.
Request a Customized Cost-Saving Analysis from our technical procurement team today to explore how this innovative synthesis route can enhance your supply chain resilience. We provide comprehensive support including specific COA data and route feasibility assessments tailored to your production requirements and quality specifications.
