Scalable Production of 2-Trifluoromethyl Quinazolinones via Iron Catalysis for Global Pharmaceutical Supply Chains
Scalable Production of 2-Trifluoromethyl Quinazolinones via Iron Catalysis for Global Pharmaceutical Supply Chains
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes. A pivotal advancement in this domain is detailed in patent CN111675662B, which discloses a robust preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These heterocyclic scaffolds are not merely academic curiosities; they form the core structure of numerous bioactive molecules with potent anti-cancer, anticonvulsant, and anti-inflammatory properties, as illustrated by the diverse biological activities associated with the quinazolinone framework shown in the reference structures. The strategic incorporation of the trifluoromethyl group into these scaffolds is a critical medicinal chemistry tactic, known to drastically improve metabolic stability, lipophilicity, and overall bioavailability of drug candidates. This patent presents a breakthrough methodology that leverages inexpensive iron catalysis to construct these valuable motifs, addressing long-standing challenges in yield and substrate scope that have plagued previous synthetic efforts.
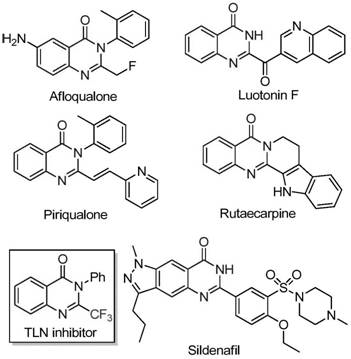
Quinazolinone compounds represent a privileged class of nitrogen-containing fused ring six-membered heterocycles that are ubiquitous in modern drug discovery pipelines. Their structural versatility allows them to interact with a wide array of biological targets, making them indispensable building blocks for developing new therapeutic agents. The specific subclass of 2-trifluoromethyl substituted quinazolinones has garnered intense interest due to the unique electronic and steric properties imparted by the CF3 group. However, accessing these specific derivatives has historically been fraught with difficulties. Traditional synthetic strategies often rely on the cyclization of synthons containing the trifluoromethyl group with substrates like anthranilamide or isatoic anhydride. While effective in some contexts, these conventional methods are frequently hampered by the use of expensive and hazardous reagents such as trifluoroacetic anhydride or ethyl trifluoroacetate. Moreover, these legacy processes often suffer from narrow substrate tolerance, requiring harsh reaction conditions that limit the diversity of analogues that can be practically synthesized for SAR studies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The historical reliance on precious metal catalysts or stoichiometric amounts of aggressive fluorinating agents has created significant bottlenecks in the supply chain for these high-value intermediates. Conventional routes typically demand rigorous exclusion of moisture and oxygen, necessitating specialized equipment and increasing operational expenditures. Furthermore, the purification of products generated via these older methods can be cumbersome, often resulting in lower overall yields due to the formation of difficult-to-remove side products. The economic burden is compounded by the high cost of starting materials; many traditional trifluoromethylating agents are commodity chemicals with volatile pricing, directly impacting the cost of goods sold (COGS) for the final API. For procurement managers, this translates to unpredictable budgeting and potential supply disruptions. Additionally, the environmental footprint of these methods is often substantial, generating significant waste streams that require costly treatment and disposal, thereby conflicting with the increasingly stringent green chemistry mandates imposed by global regulatory bodies.
The Novel Approach
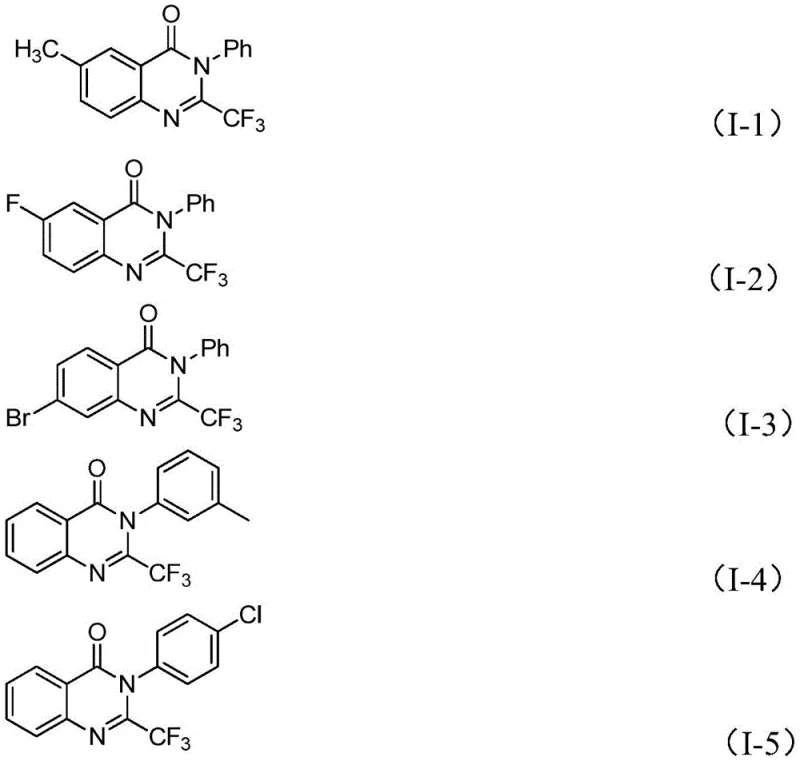
In stark contrast, the methodology outlined in the patent data introduces a paradigm shift by utilizing readily available trifluoroethylimidoyl chloride and isatin derivatives as the primary building blocks. This novel approach capitalizes on the abundance and low cost of isatin, a natural product scaffold that is easily functionalized. By employing a cheap metal iron catalyst, specifically ferric chloride, the process eliminates the dependency on scarce and expensive transition metals like palladium or rhodium. The reaction conditions are remarkably forgiving, proceeding efficiently in common polar aprotic solvents like DMF under air, which simplifies the engineering controls required for manufacturing. As demonstrated by the specific examples provided in the patent, such as compounds I-1 through I-5, this method exhibits exceptional functional group tolerance, accommodating various substituents including halogens, alkyl groups, and methoxy groups without compromising yield. This versatility is crucial for R&D teams aiming to rapidly generate libraries of analogues for biological screening.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
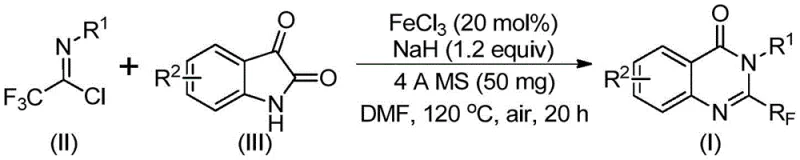
The mechanistic pathway of this transformation is a testament to the elegance of base-promoted cascade reactions mediated by Lewis acidic iron species. The reaction initiates with the deprotonation of the isatin nitrogen by sodium hydride, generating a nucleophilic anion that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step forms a key carbon-nitrogen bond, creating a trifluoroacetamidine intermediate. Subsequently, the ferric chloride catalyst plays a dual role: it acts as a Lewis acid to activate the carbonyl group of the isatin moiety and facilitates a decarbonylation event. This decarbonylation is the critical driving force that allows the system to rearrange and close the ring, forming the stable quinazolinone core. The presence of 4A molecular sieves in the reaction mixture is a subtle yet vital detail, as they serve to scavenge trace water that could otherwise hydrolyze the sensitive imidoyl chloride or deactivate the sodium hydride, ensuring high conversion rates. The entire sequence proceeds through a concerted mechanism that minimizes the formation of polymeric byproducts, leading to cleaner reaction profiles.
Understanding the impurity profile is paramount for any process intended for pharmaceutical application. In this iron-catalyzed system, the primary impurities likely stem from incomplete conversion of the starting isatin or hydrolysis of the imidoyl chloride. However, the robustness of the cyclization step ensures that once the intermediate is formed, it rapidly converts to the thermodynamically stable quinazolinone product. The use of DMF as a solvent is particularly advantageous here, as it effectively solubilizes both the ionic intermediates and the organic substrates, promoting homogeneous reaction kinetics. The tolerance for electron-withdrawing and electron-donating groups on the aromatic rings suggests that the electronic nature of the substrate does not drastically alter the rate-determining step, which is likely the initial nucleophilic attack or the subsequent cyclization. This mechanistic resilience provides R&D directors with the confidence that the process can be adapted to a wide range of substituted isatins without needing to re-optimize the entire protocol for each new derivative.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory scale optimization and pilot plant production. The protocol involves a straightforward one-pot procedure where all reagents are combined in a specific order to maximize safety and efficiency. The initial mixing at a lower temperature allows for the controlled formation of the reactive intermediate, preventing exothermic runaway, before the system is heated to drive the cyclization to completion. Detailed standardized operating procedures regarding stoichiometry, addition rates, and quenching protocols are essential for reproducibility. For those looking to implement this technology, the following guide outlines the critical steps derived from the patent examples to ensure successful isolation of the target compound with high purity.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then increase temperature to 120°C and continue reacting for 18-20 hours under air.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final quinazolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the pain points of modern chemical procurement and supply chain management. The shift from precious metal catalysts to iron represents a fundamental change in the cost structure of the molecule. Iron salts are commodities with stable pricing and abundant global supply, insulating manufacturers from the geopolitical and market volatility often associated with platinum group metals. Furthermore, the simplicity of the workup procedure—filtration followed by standard chromatography—reduces the processing time and solvent consumption per kilogram of product. This efficiency gain translates into a significantly reduced manufacturing footprint and lower utility costs. For supply chain heads, the use of commercially available starting materials like isatin and simple aromatic amines means that raw material sourcing is straightforward and reliable, minimizing the risk of production delays due to material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive catalysts and the use of cheap, bulk-available reagents drastically lowers the direct material costs. The high atom economy of the cyclization reaction ensures that a greater proportion of the input mass ends up in the final product, reducing waste disposal fees. Additionally, the ability to run the reaction under air rather than inert atmosphere removes the need for nitrogen or argon blanketing, further cutting operational expenses associated with gas consumption and equipment maintenance.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that the process is less sensitive to minor variations in temperature or reagent quality, leading to consistent batch-to-batch performance. This reliability is crucial for maintaining continuous supply to downstream API manufacturers. The broad substrate scope allows for the rapid synthesis of different analogues using the same core process infrastructure, providing flexibility to respond to changing market demands without significant retooling or process validation efforts.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently on gram scales with potential for tonnage production. The absence of toxic heavy metals in the catalyst system simplifies the regulatory compliance landscape, particularly regarding residual metal limits in the final drug substance. This 'green' aspect aligns with corporate sustainability goals and reduces the environmental liability associated with chemical manufacturing, making it an attractive option for companies striving to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when adopting this technology. Understanding these nuances helps in making informed decisions about process integration and resource allocation.
Q: What are the key advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) offers significant cost advantages as it is an inexpensive, earth-abundant metal compared to precious metal catalysts. Furthermore, it demonstrates high catalytic efficiency and excellent functional group tolerance, allowing for the synthesis of diverse derivatives without requiring complex ligand systems.
Q: How does the introduction of a trifluoromethyl group impact the biological profile of quinazolinones?
A: Introducing a trifluoromethyl group significantly enhances the electronegativity, lipophilicity, and metabolic stability of the target molecule. These physicochemical improvements often translate to better bioavailability and prolonged half-life in vivo, making these compounds highly desirable candidates for drug development in oncology and anti-inflammatory applications.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It utilizes readily available starting materials like isatin and avoids severe reaction conditions or expensive reagents. The simple workup procedure involving filtration and standard column chromatography facilitates easy scale-up from gram to kilogram levels for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed methodology for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our facilities are equipped with state-of-the-art reactors capable of handling the specific thermal profiles required for this two-stage heating process, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We understand that consistency is key in the pharmaceutical supply chain, and our commitment to quality assurance ensures that the 2-trifluoromethyl quinazolinones we deliver are free from critical impurities and ready for the next step in your synthesis.
We invite you to leverage our technical expertise to optimize your supply chain for these critical intermediates. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient route. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary derivatives. Let us help you secure a reliable, cost-effective, and sustainable supply of 2-trifluoromethyl quinazolinones for your next generation of therapeutic innovations.
