Scalable Production of Bioactive 2-Trifluoromethyl Quinazolinones via Iron Catalysis
Scalable Production of Bioactive 2-Trifluoromethyl Quinazolinones via Iron Catalysis
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive small molecules. Patent CN111675662B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing critical bottlenecks in current manufacturing paradigms. Quinazolinones serve as privileged scaffolds in medicinal chemistry, exhibiting a broad spectrum of pharmacological activities including anticancer, anticonvulsant, and anti-inflammatory properties. The strategic introduction of a trifluoromethyl group further enhances these profiles by improving metabolic stability, lipophilicity, and bioavailability. This technical insight analyzes the patented methodology, highlighting its potential to streamline the supply chain for reliable pharmaceutical intermediate suppliers seeking to optimize cost and efficiency in API manufacturing.
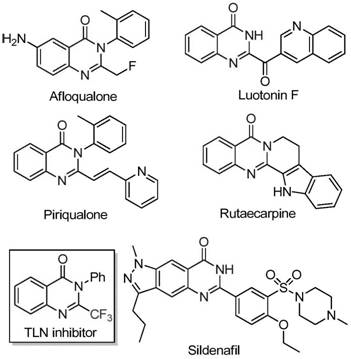
Traditional synthetic approaches for constructing the quinazolinone core often rely on the cyclization of anthranilamides or isatoic anhydrides with trifluoromethyl synthons like trifluoroacetic anhydride. While effective on a laboratory scale, these legacy methods frequently suffer from severe reaction conditions, limited substrate scope, and the utilization of costly reagents that complicate purification and waste management. The reliance on precious metal catalysts or harsh dehydrating agents in conventional protocols often leads to inconsistent impurity profiles, posing significant challenges for regulatory compliance in GMP environments. Furthermore, the narrow functional group tolerance of older methodologies restricts the structural diversity accessible to process chemists, limiting the ability to rapidly iterate on lead optimization campaigns for new drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethylated quinazolinones has been hindered by the high cost and handling difficulties associated with common trifluoromethylating agents. Methods utilizing ethyl trifluoroacetate or trifluoroacetic anhydride often require stoichiometric amounts of strong bases or Lewis acids, generating substantial quantities of salt waste that necessitate complex downstream processing. Additionally, these reactions frequently exhibit poor atom economy and require elevated temperatures that can degrade sensitive functional groups on the aromatic ring. For procurement managers, these inefficiencies translate into higher raw material costs and extended production cycles, directly impacting the bottom line. The inability to easily scale these processes without compromising yield or purity remains a persistent pain point for contract development and manufacturing organizations (CDMOs) aiming to deliver high-purity pharmaceutical intermediates.
The Novel Approach
The methodology described in CN111675662B represents a paradigm shift by employing readily available trifluoroethylimidoyl chloride and isatin derivatives as starting materials. This innovative route leverages an earth-abundant iron catalyst, specifically ferric chloride, to drive the cyclization under relatively mild conditions compared to traditional methods. By utilizing cheap and commercially accessible feedstocks, the process significantly lowers the barrier to entry for large-scale production. The reaction demonstrates excellent functional group tolerance, accommodating various substituents such as halogens, alkyl, and methoxy groups on both the isatin and the imidoyl chloride components. This versatility allows for the rapid generation of diverse compound libraries, facilitating faster drug discovery timelines while maintaining a lean and cost-effective manufacturing footprint.
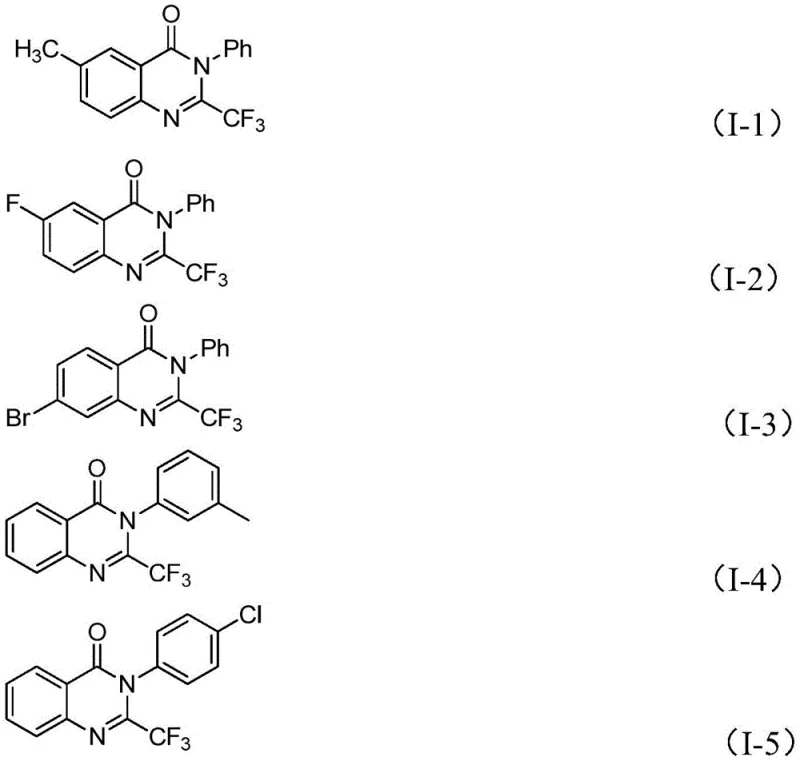
Mechanistic Insights into FeCl3-Catalyzed Cyclization
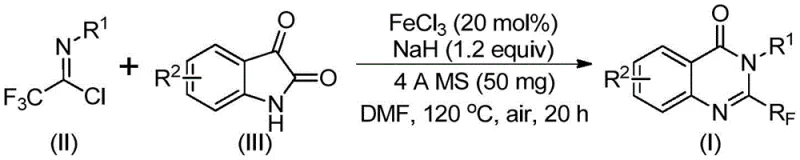
The core of this technological advancement lies in the efficient iron-catalyzed decarbonylation and cyclization mechanism. The reaction initiates with the formation of a carbon-nitrogen bond between the trifluoroethylimidoyl chloride and the isatin substrate, likely promoted by the base sodium hydride. Subsequently, the ferric chloride catalyst facilitates a decarbonylation event followed by an intramolecular cyclization to form the quinazolinone ring system. This mechanistic pathway avoids the formation of stable intermediates that typically plague other synthetic routes, ensuring a smoother conversion to the final product. The use of 4A molecular sieves plays a crucial role in sequestering moisture, which is vital for maintaining the activity of the sodium hydride and preventing hydrolysis of the sensitive imidoyl chloride species. Understanding this mechanism is essential for R&D directors aiming to replicate or modify the process for specific analog synthesis.
Impurity control is inherently superior in this system due to the high selectivity of the iron catalyst and the specific reaction conditions employed. The protocol operates effectively in polar aprotic solvents like DMF, which solubilize the ionic intermediates while stabilizing the transition states. Experimental data indicates that the reaction proceeds through a well-defined pathway that minimizes side reactions such as polymerization or over-fluorination. The post-treatment process is straightforward, involving simple filtration and standard column chromatography, which ensures that the final API intermediate meets stringent purity specifications. For quality assurance teams, the predictability of the impurity profile simplifies the validation process and reduces the risk of batch failures during commercial production runs.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The operational simplicity of this synthesis makes it highly attractive for technology transfer from laboratory to pilot plant scales. The procedure involves mixing the catalyst, base, and molecular sieves with the substrates in a single pot, reducing the need for complex multi-step sequences. Detailed standardized synthesis steps are provided below to guide process engineers in implementing this route.
- Combine ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in anhydrous DMF solvent within a reaction vessel.
- Initiate the reaction by stirring at 40°C for 8-10 hours, then increase the temperature to 120°C and maintain for an additional 18-20 hours under air atmosphere.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify using column chromatography to isolate the target quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling advantages that directly address the priorities of procurement and supply chain leadership. The substitution of expensive reagents with commodity chemicals drastically simplifies the sourcing strategy and mitigates supply risk. By eliminating the need for specialized trifluoromethylating agents that often have long lead times and volatile pricing, manufacturers can achieve greater stability in their raw material inventory. Furthermore, the use of iron chloride as a catalyst removes the necessity for costly heavy metal scavenging steps, which are typically required when using palladium or rhodium catalysts. This reduction in downstream processing complexity translates to significant operational expenditure savings and a smaller environmental footprint, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the utilization of low-cost starting materials and catalysts. Isatin and aromatic amines used to prepare the imidoyl chloride are widely available bulk chemicals, ensuring competitive pricing and consistent supply. The elimination of precious metals not only reduces direct material costs but also lowers the cost of waste disposal and regulatory compliance associated with heavy metal residues. Additionally, the high conversion rates observed in the patent examples suggest that solvent recovery and recycling can be optimized, further enhancing the overall cost efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on globally sourced, non-proprietary raw materials. Unlike specialized reagents that may be subject to export controls or single-source dependencies, the key components of this synthesis are produced by multiple vendors worldwide. This diversification reduces the risk of production stoppages due to raw material shortages. Moreover, the robustness of the reaction conditions, which tolerate air and moisture to a reasonable extent thanks to the molecular sieves, allows for more flexible manufacturing scheduling and reduces the strictness of inert atmosphere requirements, thereby increasing throughput capacity.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, having been demonstrated to work efficiently with standard laboratory equipment and amenable to larger reactor vessels. The absence of highly toxic reagents and the generation of manageable byproducts simplify the environmental health and safety (EHS) profile of the operation. Waste streams are easier to treat, and the overall atom economy is superior to many alternative methods. For facilities operating under strict environmental regulations, this cleaner profile facilitates faster permitting and reduces the liability associated with hazardous waste management, making it an ideal candidate for sustainable commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These answers are derived directly from the experimental data and disclosures within the patent documentation to ensure accuracy and reliability for technical decision-makers.
Q: What catalyst system is utilized in this novel quinazolinone synthesis?
A: The process utilizes an inexpensive iron-based catalyst system, specifically ferric chloride (FeCl3) at 20 mol% loading, promoted by sodium hydride (NaH) and 4A molecular sieves in DMF solvent.
Q: What are the primary starting materials for this reaction?
A: The synthesis employs readily available isatin derivatives and trifluoroethylimidoyl chloride as the key building blocks, avoiding the need for expensive trifluoroacetic anhydride or ethyl trifluoroacetate.
Q: What yields can be expected from this methodology?
A: Experimental data indicates high efficiency, with isolated yields ranging significantly depending on substituents, achieving up to 93% for specific fluorinated derivatives and consistently above 70% for various aryl-substituted analogs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics. Our team of expert chemists has extensively evaluated the methodology described in CN111675662B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from gram-scale optimization to full commercial supply. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest industry standards for potency and impurity control.
We invite you to collaborate with us to leverage this advanced technology for your pipeline projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage potential partners to contact us directly to request specific COA data for our catalog compounds or to discuss route feasibility assessments for custom synthesis projects. Let us help you accelerate your drug development timeline with reliable, high-quality intermediates produced via cutting-edge, cost-effective chemistry.
