Scalable Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to incorporate trifluoromethyl groups into heterocyclic scaffolds, driven by the profound impact these moieties have on metabolic stability, lipophilicity, and bioavailability. Patent CN113121462B, published in late 2022, introduces a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds that addresses long-standing synthetic challenges. Unlike conventional approaches that rely on hazardous reagents or expensive catalysts, this invention leverages a base-promoted cyclization strategy using readily available trifluoroethylimidoyl chlorides and diazo compounds. For R&D directors and process chemists, this represents a significant leap forward in constructing complex nitrogen-containing heterocycles with high efficiency and operational simplicity. The technology not only streamlines the synthetic route but also aligns perfectly with modern green chemistry principles by avoiding toxic azides and heavy metals, thereby offering a compelling value proposition for the reliable pharmaceutical intermediate supplier market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
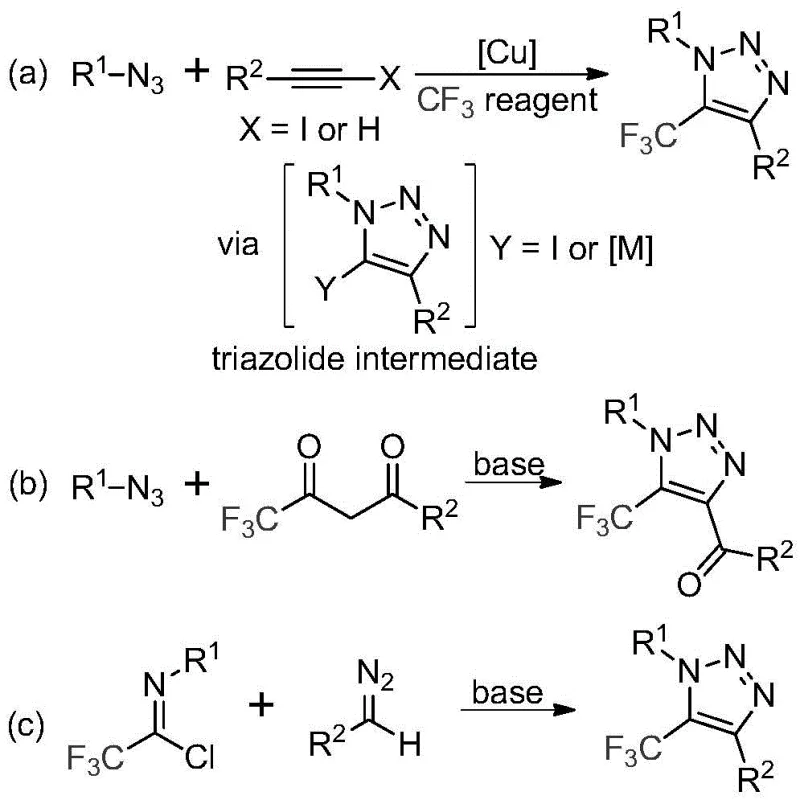
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has been fraught with significant safety and economic hurdles that complicate commercial scale-up. The most prevalent literature methods typically involve copper-catalyzed [3+2] cycloadditions between alkynes and organic azides, followed by trifluoromethylation, or organocatalytic 1,3-dipolar cycloadditions involving azides and trifluoromethyl ketones. A critical bottleneck in these traditional pathways is the reliance on organic azides, which are notoriously toxic and possess high explosive potential, necessitating stringent safety protocols and specialized infrastructure that drive up capital expenditure. Furthermore, copper-catalyzed variants introduce the persistent issue of heavy metal contamination, requiring additional downstream purification steps such as scavenger resin treatment to meet the rigorous purity specifications demanded by regulatory bodies for active pharmaceutical ingredients. These factors collectively result in prolonged lead times, increased waste generation, and elevated manufacturing costs, making the commercial scale-up of complex pharmaceutical intermediates via these routes less attractive for large-scale production.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN113121462B offers a transformative solution by utilizing a metal-free, azide-free protocol that fundamentally reshapes the synthetic landscape for these valuable heterocycles. As illustrated in the comparative reaction schemes below, the new method bypasses the dangerous azide intermediates entirely, opting instead for a direct coupling between trifluoroethylimidoyl chlorides and diazo compounds promoted by a simple inorganic base. This shift not only mitigates the severe safety risks associated with handling explosive precursors but also eliminates the need for expensive transition metal catalysts like copper. The reaction proceeds under mild thermal conditions in common aprotic solvents, demonstrating exceptional functional group tolerance across a wide range of substrates. By removing the constraints of metal catalysis and hazardous reagents, this approach facilitates a drastically simplified workflow that is inherently safer, more cost-effective, and easier to implement in standard chemical manufacturing facilities without the need for specialized explosion-proof reactors.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic elegance of this synthesis lies in its ability to forge the triazole ring through a cascade of nucleophilic additions and cyclizations without external metal activation. The reaction is initiated by the base-promoted intermolecular nucleophilic attack of the diazo compound onto the electrophilic carbon of the trifluoroethylimidoyl chloride. This initial addition-elimination sequence effectively constructs the critical carbon-carbon bond while expelling the chloride leaving group, generating a reactive intermediate that is primed for ring closure. Subsequently, the system undergoes an intramolecular 5-endo-dig cyclization, a process that is thermodynamically favorable under the specified reaction conditions, to close the five-membered triazole ring. This mechanism is particularly advantageous because it avoids the formation of stable metal-carbene complexes that often lead to difficult-to-remove impurities in transition metal catalysis. Instead, the pathway relies on the inherent reactivity of the diazo species and the electron-withdrawing nature of the trifluoromethyl group to drive the cyclization to completion with high regioselectivity.
From an impurity control perspective, the absence of transition metals significantly reduces the complexity of the impurity profile, as there are no metal-ligand complexes or metal-mediated side reactions to manage. The use of cesium carbonate as a promoter ensures a homogeneous reaction environment that facilitates efficient mixing and heat transfer, further minimizing the formation of byproducts associated with localized hot spots or poor solubility. The mild reaction temperatures, typically ranging from 50°C to 70°C, prevent thermal degradation of sensitive functional groups on the aromatic rings, ensuring that the final product retains high chemical integrity. This clean reaction profile translates directly to simplified downstream processing, where standard filtration and column chromatography are sufficient to achieve high-purity isolates, thereby reducing the overall process mass intensity and solvent consumption typically associated with more aggressive synthetic routes.
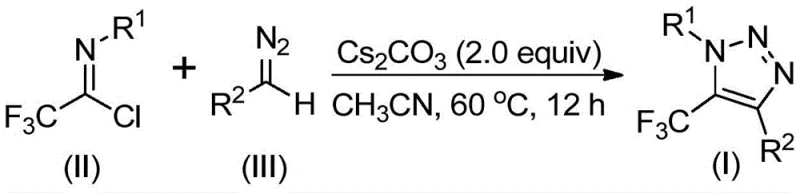
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it highly accessible for both laboratory optimization and industrial manufacturing. The protocol involves combining the key starting materials—trifluoroethylimidoyl chloride and the specific diazo compound—with cesium carbonate and 4 Å molecular sieves in an organic solvent such as acetonitrile. The molecular sieves play a crucial role in sequestering trace moisture, which could otherwise hydrolyze the sensitive imidoyl chloride or diazo species, thereby ensuring maximum yield and reproducibility. The reaction mixture is then heated to a moderate temperature and stirred for a defined period until conversion is complete, after which simple workup procedures yield the desired triazole derivative. For detailed operational parameters and specific stoichiometric ratios optimized for different substrates, please refer to the standardized guide below.
- Prepare the reaction mixture by adding cesium carbonate, 4 Å molecular sieves, trifluoroethylimidoyl chloride, and the diazo compound into an aprotic organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature range of 50°C to 70°C and maintain stirring for 8 to 16 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture to remove solids, concentrate the filtrate, and purify the crude residue via silica gel column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers substantial strategic advantages that extend far beyond simple yield improvements. By transitioning away from copper-catalyzed processes, manufacturers can eliminate the significant costs associated with purchasing precious metal catalysts and the subsequent expensive remediation steps required to lower metal residues to ppm levels. This reduction in process complexity directly correlates to a shorter overall production cycle time, allowing for faster turnaround on custom synthesis orders and improved responsiveness to market demands. Furthermore, the reliance on commercially available and stable starting materials like diazo compounds and imidoyl chlorides ensures a robust supply chain that is less susceptible to the volatility often seen with specialized organometallic reagents or hazardous azides. This stability in raw material sourcing is critical for maintaining continuous production schedules and meeting the just-in-time delivery requirements of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the need for costly metal scavengers and extensive analytical testing for heavy metal residues, which are mandatory for API intermediates. Additionally, the use of inexpensive inorganic bases like cesium carbonate instead of proprietary ligands or organocatalysts significantly lowers the raw material cost per kilogram. The simplified purification process, which avoids complex extraction sequences often needed to remove metal complexes, further reduces solvent usage and waste disposal costs, leading to a leaner and more economically efficient manufacturing process that enhances overall profit margins.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, including various substituted aromatic amines and diazo precursors, are widely available from multiple global suppliers, reducing the risk of single-source dependency. The avoidance of explosive azides means that the synthesis can be performed in standard chemical facilities without the need for specialized blast-resistant infrastructure, vastly expanding the number of qualified contract manufacturing organizations (CMOs) capable of producing these intermediates. This flexibility in manufacturing partners ensures greater supply security and reduces the risk of production bottlenecks caused by facility limitations or regulatory restrictions on hazardous materials.
- Scalability and Environmental Compliance: The reaction conditions are mild and operate at atmospheric pressure, making the scale-up from gram to multi-ton quantities straightforward and safe. The use of common solvents like acetonitrile simplifies solvent recovery and recycling efforts, aligning with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. The metal-free nature of the process results in a cleaner waste stream that is easier to treat and dispose of, reducing the environmental footprint of the manufacturing operation and ensuring compliance with green chemistry initiatives that are becoming a prerequisite for doing business with major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this base-promoted triazole synthesis. These insights are derived directly from the experimental data and scope analysis provided in the patent documentation, offering clarity on substrate compatibility, safety profiles, and purification strategies. Understanding these nuances is essential for process development teams evaluating this technology for integration into their existing pipelines or for procurement specialists assessing the feasibility of long-term supply agreements.
Q: What are the primary safety advantages of this new synthesis method compared to traditional routes?
A: Traditional methods often rely on organic azides, which are known to be toxic and potentially explosive. This novel base-promoted method utilizes stable diazo compounds and trifluoroethylimidoyl chlorides, significantly enhancing operational safety and eliminating the need for specialized explosion-proof equipment.
Q: Does this process require expensive transition metal catalysts?
A: No, the process is entirely metal-free. It utilizes cesium carbonate as a promoter instead of copper or other transition metals. This eliminates the costly and time-consuming steps associated with heavy metal removal and residual metal testing, which is critical for pharmaceutical grade intermediates.
Q: What is the substrate scope for the R1 and R2 groups in this reaction?
A: The method demonstrates broad functional group tolerance. R1 can be alkyl, substituted or unsubstituted aryl groups (including phenethyl and various substituted phenyls). R2 accommodates aroyl, phospholipid, alkoxycarbonyl, or trifluoromethyl groups, allowing for the synthesis of diverse triazole derivatives suitable for various drug discovery programs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of next-generation therapeutics and agrochemicals. Our technical team has thoroughly analyzed the potential of the base-promoted cyclization technology described in patent CN113121462B and is fully prepared to leverage this methodology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole delivered meets the highest international standards for quality and consistency.
We invite you to engage with our technical procurement team to discuss how this innovative synthetic route can be tailored to your specific molecule requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this metal-free process for your specific volume needs. We encourage you to contact us today to request specific COA data for our catalog compounds or to initiate a discussion on route feasibility assessments for your proprietary targets, ensuring a secure and cost-effective supply chain for your critical building blocks.
