Advanced Base-Promoted Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Pharmaceutical Intermediates
Advanced Base-Promoted Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Pharmaceutical Intermediates
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for more efficient, safer, and scalable synthetic routes for high-value pharmaceutical intermediates. A significant breakthrough in this domain is documented in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology represents a paradigm shift away from traditional transition-metal catalysis towards a more sustainable, base-promoted methodology. For R&D directors and procurement specialists in the fine chemical sector, this patent offers a compelling alternative for constructing the ubiquitous 1,2,3-triazole scaffold, a motif found in countless bioactive molecules ranging from beta-3 adrenergic receptor agonists to advanced agrochemical fungicides. The strategic value of this invention lies not only in its chemical elegance but also in its potential to streamline supply chains by eliminating hazardous reagents and expensive catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has been dominated by two primary methodologies, both of which present significant operational and safety challenges for large-scale manufacturing. The first conventional route involves copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides to form triazole metal complexes, followed by trifluoromethylation. The second relies on organocatalytic 1,3-dipolar cycloaddition between azides and trifluoromethyl ketones. The critical bottleneck in both strategies is the reliance on organic azides, which are notoriously toxic, unstable, and prone to explosive decomposition, posing severe safety risks during kilogram-scale production. Furthermore, the use of copper catalysts introduces the risk of heavy metal contamination, necessitating rigorous and costly purification steps to meet the stringent residual metal limits imposed by regulatory bodies for active pharmaceutical ingredients (APIs).
The Novel Approach
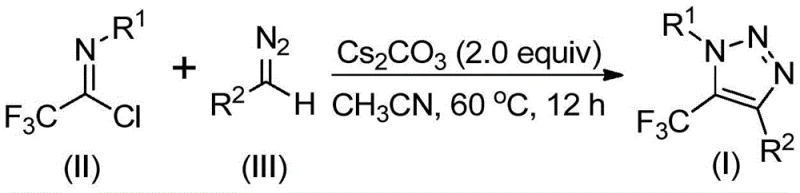
In stark contrast to these legacy methods, the technology described in CN113121462B utilizes a metal-free, base-promoted strategy that fundamentally alters the risk profile and cost structure of the synthesis. By employing cheap and readily available diazo compounds and trifluoroethylimidoyl chloride as starting materials, this method completely bypasses the need for explosive azides and toxic transition metals. The reaction proceeds efficiently under mild thermal conditions, typically between 50°C and 70°C, using cesium carbonate as a benign promoter. This shift not only enhances laboratory safety but also drastically simplifies the downstream processing, as there is no need for specialized equipment to handle high-pressure azide chemistry or complex protocols to scavenge residual copper. This approach exemplifies the principles of green chemistry while delivering high-purity products suitable for sensitive pharmaceutical applications.
Mechanistic Insights into Base-Promoted Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for process chemists aiming to optimize the reaction for specific substrates. The proposed mechanism involves a sophisticated cascade initiated by the base-promoted intermolecular nucleophilic addition of the diazo compound to the electrophilic carbon of the trifluoroethylimidoyl chloride. This initial step facilitates the formation of a critical carbon-carbon bond, setting the stage for the subsequent ring closure. Following this addition-elimination sequence, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This specific cyclization mode is kinetically favored under the reaction conditions and leads directly to the formation of the five-membered 1,2,3-triazole ring with the trifluoromethyl group positioned at the 5-position. The elegance of this mechanism lies in its atom economy and the avoidance of high-energy intermediates that often lead to polymeric byproducts in traditional azide chemistry.
From an impurity control perspective, the mild nature of this base-promoted system offers distinct advantages over harsher acidic or metal-catalyzed alternatives. The use of cesium carbonate in aprotic solvents like acetonitrile creates a homogeneous reaction environment that minimizes side reactions such as hydrolysis of the imidoyl chloride or dimerization of the diazo species. The patent data highlights that the reaction exhibits excellent functional group tolerance, accommodating various substituents on the aromatic rings of both the imidoyl chloride and the diazo compound without significant degradation in yield. This robustness suggests that the electronic properties of the substrates do not drastically alter the reaction pathway, allowing for a modular approach to library synthesis. Consequently, manufacturers can achieve consistent purity profiles across a wide range of derivatives, reducing the burden on analytical quality control teams.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
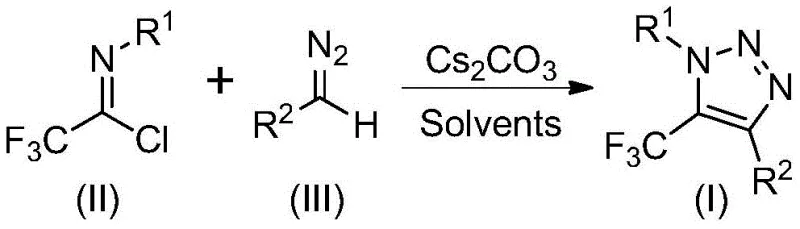
Implementing this synthesis in a pilot or production plant requires adherence to specific operational parameters to maximize yield and reproducibility. The patent outlines a straightforward protocol where the key reagents—trifluoroethylimidoyl chloride, the diazo compound, and cesium carbonate—are combined in an organic solvent. The choice of solvent is critical; while several aprotic solvents were screened, acetonitrile emerged as the superior medium, providing the highest conversion rates and facilitating easy product isolation. The reaction temperature is maintained in a narrow window, optimally at 60°C, which balances reaction kinetics with thermal stability. For R&D teams looking to replicate or scale this process, the detailed standardized synthesis steps are provided in the guide below, ensuring that the nuanced stoichiometry and mixing requirements are met precisely.
- Prepare the reaction mixture by adding cesium carbonate (2.0 equivalents), trifluoroethylimidoyl chloride, and diazo compound (1.5 equivalents) into anhydrous acetonitrile solvent within a Schlenk tube.
- Heat the reaction mixture to a temperature range of 50°C to 70°C (optimally 60°C) and maintain stirring for 8 to 16 hours to ensure complete conversion.
- Upon completion, filter the mixture to remove inorganic salts, concentrate the filtrate, and purify the crude residue via silica gel column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible strategic benefits that extend beyond mere chemical yield. The elimination of transition metal catalysts removes a significant cost center associated with ligand procurement and, more importantly, the extensive purification processes required to remove trace metals from the final API. Additionally, the replacement of hazardous azides with stable diazo compounds significantly lowers insurance premiums and safety compliance costs associated with handling explosive materials. The use of commodity chemicals like cesium carbonate and acetonitrile ensures that the raw material supply chain is robust and less susceptible to geopolitical disruptions that often affect specialized catalysts. This stability allows for more accurate long-term forecasting and inventory management.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is substantial. By removing the requirement for expensive copper catalysts and their associated ligands, the direct material cost of the reaction is significantly lowered. Furthermore, the simplified workup procedure, which involves basic filtration and standard column chromatography rather than complex metal scavenging or distillation under reduced pressure to remove volatile azides, reduces labor hours and energy consumption. The overall process mass intensity (PMI) is improved due to the higher atom efficiency and the ability to use simpler solvent systems, leading to reduced waste disposal costs and a smaller environmental footprint for the manufacturing facility.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically the trifluoroethylimidoyl chlorides and diazo compounds, are derived from widely available commodity precursors such as aromatic amines and acid chlorides. This contrasts sharply with traditional methods that may rely on bespoke azides or specialized trifluoromethylating reagents with limited global suppliers. By anchoring the synthesis in a supply chain built on bulk chemicals, manufacturers can secure multiple sourcing options, mitigating the risk of single-supplier dependency. This diversification ensures continuity of supply even during market fluctuations, guaranteeing that production schedules for critical pharmaceutical intermediates remain uninterrupted.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies, but this base-promoted reaction demonstrates excellent scalability potential. The mild reaction temperatures (50-70°C) mean that standard stainless steel reactors can be used without the need for specialized cryogenic cooling or high-pressure vessels required for some azide reactions. The absence of heavy metals simplifies the environmental compliance landscape, as wastewater treatment does not require specialized protocols for heavy metal precipitation. This ease of scale-up from gram to multi-kilogram quantities allows for rapid technology transfer from the lab to the plant, accelerating time-to-market for new drug candidates while maintaining strict adherence to environmental, health, and safety (EHS) regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on reaction scope, safety, and optimization strategies. Understanding these nuances is essential for process development teams evaluating this route for inclusion in their manufacturing portfolio.
Q: Why is this base-promoted method superior to traditional copper-catalyzed routes?
A: Traditional methods often rely on toxic and explosive organic azides and require expensive transition metal catalysts like copper, which necessitate complex removal steps to meet pharmaceutical purity standards. This novel base-promoted approach eliminates the need for transition metals and hazardous azides, utilizing safer diazo compounds and inexpensive cesium carbonate, thereby simplifying purification and reducing safety risks.
Q: What are the optimal reaction conditions for scaling this synthesis?
A: The patent data indicates that using acetonitrile as the solvent with cesium carbonate (2.0 equivalents) at 60°C provides the highest efficiency. The reaction tolerates a wide range of functional groups on both the imidoyl chloride and the diazo compound, making it robust for various substrate derivatives.
Q: How does the trifluoromethyl group benefit the final pharmaceutical application?
A: The introduction of a trifluoromethyl group significantly enhances the electronegativity, metabolic stability, and lipophilicity of the parent molecule. These properties are critical for improving the bioavailability and binding affinity of drug candidates, particularly in the development of beta-3 adrenergic receptor agonists and agrochemical agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the base-promoted synthesis described in CN113121462B for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical building blocks. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, including the detection of trace impurities and residual solvents, which is paramount for API manufacturing. We are committed to leveraging this advanced, metal-free technology to deliver cost-effective solutions that align with your sustainability goals.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver high-purity 5-trifluoromethyl-1,2,3-triazoles that meet the exacting standards of the global pharmaceutical industry.
