Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Pharmaceutical Applications
Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Pharmaceutical Applications
In the rapidly evolving landscape of medicinal chemistry, the demand for fluorinated heterocycles has surged due to their exceptional ability to enhance the metabolic stability and bioavailability of drug candidates. Patent CN113121462B introduces a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds, addressing critical bottlenecks in current synthetic methodologies. This technology leverages a base-promoted cyclization strategy that bypasses the need for toxic transition metals and hazardous azide reagents, offering a streamlined pathway for generating high-value scaffolds. For R&D teams and procurement specialists alike, this innovation represents a significant leap forward in constructing robust molecular architectures essential for next-generation therapeutics and agrochemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
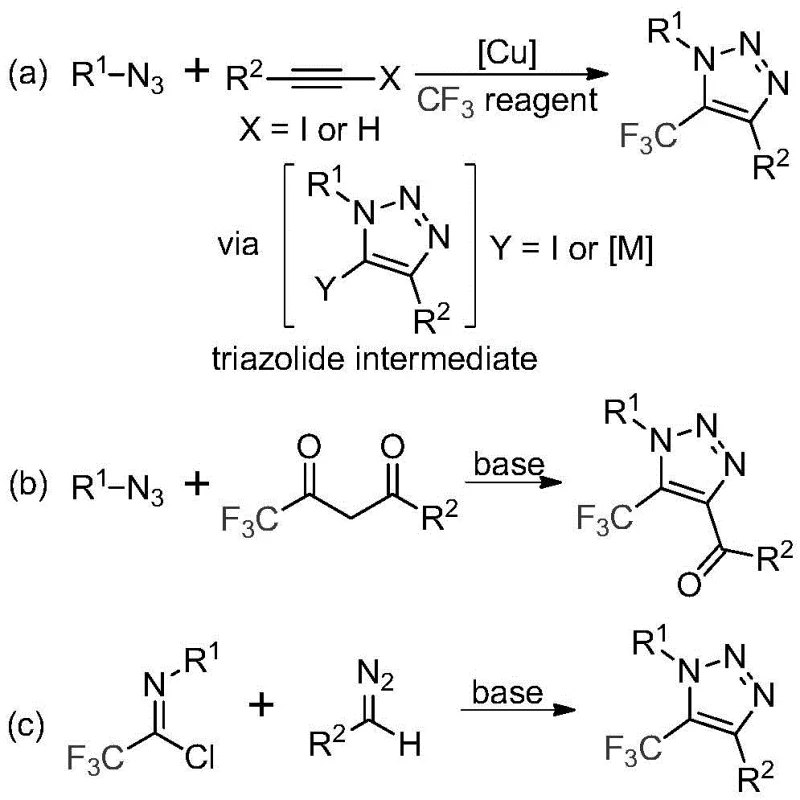
Historically, the construction of trifluoromethyl-substituted triazoles has relied heavily on copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, followed by trifluoromethylation steps. While effective in academic settings, these traditional routes present severe liabilities for industrial application, primarily concerning safety and purity. The use of organic azides introduces substantial explosion risks, necessitating specialized equipment and rigorous safety protocols that drive up operational costs. Furthermore, the reliance on copper catalysts creates a persistent challenge in downstream processing, as residual heavy metals must be meticulously removed to comply with stringent regulatory limits for active pharmaceutical ingredients (APIs).
The Novel Approach
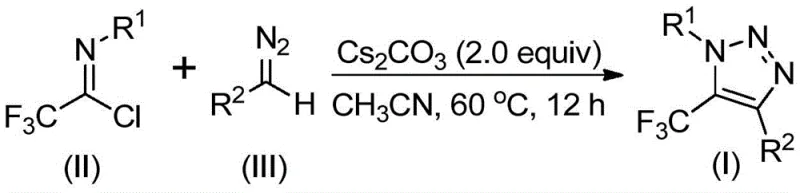
The methodology disclosed in CN113121462B fundamentally reimagines this synthetic challenge by employing a metal-free, base-promoted reaction between trifluoroethylimidoyl chlorides and diazo compounds. This approach completely eliminates the need for explosive azides and toxic copper catalysts, thereby simplifying the safety profile and reducing the environmental footprint of the manufacturing process. By utilizing readily available starting materials and mild reaction conditions, this novel route facilitates the efficient assembly of the triazole core with excellent functional group tolerance. The result is a cleaner reaction profile that minimizes impurity formation and drastically reduces the complexity of purification, offering a distinct competitive advantage for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway of this transformation is elegantly simple yet highly effective, proceeding through a base-promoted intermolecular nucleophilic addition-elimination sequence. Initially, the base activates the diazo compound, facilitating a nucleophilic attack on the electrophilic carbon of the trifluoroethylimidoyl chloride. This step promotes the formation of a crucial carbon-carbon bond while eliminating a chloride ion, setting the stage for the subsequent ring closure. The reaction then undergoes an intramolecular 5-endo-dig cyclization, which efficiently constructs the five-membered 1,2,3-triazole ring system. This mechanism avoids the formation of metal-carbene intermediates common in copper catalysis, thereby preventing side reactions associated with metal coordination and ensuring a high degree of regioselectivity.
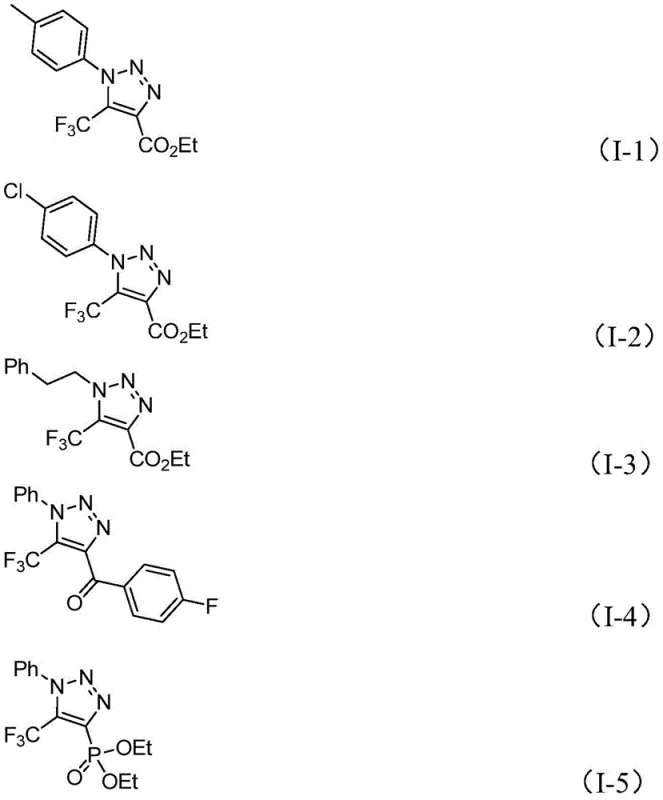
From an impurity control perspective, the absence of transition metals significantly simplifies the impurity profile, removing the risk of metal-complexed byproducts that are difficult to separate. The mild basic conditions provided by cesium carbonate ensure that sensitive functional groups on the aromatic rings remain intact, allowing for a broad substrate scope that includes various electron-donating and electron-withdrawing substituents. This robustness is critical for R&D directors who require reliable access to diverse analog libraries for structure-activity relationship (SAR) studies without the fear of decomposition or low yields due to harsh reaction environments.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
The operational simplicity of this protocol makes it highly attractive for both laboratory scale-up and commercial production. The process typically involves mixing cesium carbonate, the trifluoroethylimidoyl chloride, and the diazo compound in a polar aprotic solvent such as acetonitrile. The reaction is heated to a moderate temperature range of 50 to 70 degrees Celsius and stirred for approximately 8 to 16 hours. Upon completion, the workup is straightforward, involving filtration to remove inorganic salts followed by standard silica gel chromatography. This ease of operation lowers the barrier for adoption and ensures consistent batch-to-batch reproducibility.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an organic solvent like acetonitrile.
- Heat the reaction mixture to 50-70°C and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this metal-free methodology translates into tangible strategic benefits regarding cost, reliability, and scalability. By removing the dependency on precious metal catalysts and hazardous reagents, the overall cost of goods sold (COGS) is significantly optimized through reduced raw material expenses and simplified waste management. The supply chain becomes more resilient as the starting materials—aromatic amines and acid chlorides—are commodity chemicals with stable global availability, mitigating the risk of shortages associated with specialized reagents.
- Cost Reduction in Manufacturing: The elimination of copper catalysts removes the necessity for expensive metal scavenging resins and complex purification steps, leading to substantial cost savings in downstream processing. Additionally, the use of inexpensive cesium carbonate as a promoter instead of costly ligands or organometallic reagents further drives down the direct material costs, enhancing the overall economic viability of large-scale production runs.
- Enhanced Supply Chain Reliability: Since the key building blocks are derived from common industrial feedstocks, the supply chain is less vulnerable to the volatility often seen with specialized catalytic systems. The robustness of the reaction conditions ensures high conversion rates and consistent yields, allowing for predictable production scheduling and reliable delivery timelines for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The mild thermal requirements and absence of explosive azides make this process inherently safer and easier to scale from gram to multi-kilogram quantities without requiring specialized pressure vessels or blast-proof facilities. Furthermore, the reduction in heavy metal waste aligns with increasingly strict environmental regulations, simplifying the permitting process and reducing the long-term liability associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on its practical application in industrial settings.
Q: Why is this metal-free method superior to traditional copper-catalyzed routes?
A: Traditional methods often rely on copper catalysts which require expensive and complex removal steps to meet strict pharmaceutical purity standards. This novel base-promoted approach eliminates heavy metal contamination risks entirely.
Q: What are the safety advantages of using diazo compounds over organic azides?
A: Organic azides are notoriously unstable and pose significant explosion hazards, especially on a large industrial scale. The disclosed method utilizes stable diazo compounds and imidoyl chlorides, significantly enhancing operational safety.
Q: Can this synthesis be scaled for commercial production?
A: Yes, the patent explicitly demonstrates that the reaction conditions are mild (50-70°C) and the process can be easily extended to the gram level and beyond, making it highly suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the development of advanced therapeutic agents. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole intermediate meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with our technical team to explore how this innovative route can optimize your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, where we can provide specific COA data and route feasibility assessments tailored to your target molecule. Let us help you secure a sustainable and cost-effective supply chain for your critical fluorinated building blocks.
