Advanced Iodine-Promoted Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Scalable Pharma Manufacturing
Advanced Iodine-Promoted Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Scalable Pharma Manufacturing
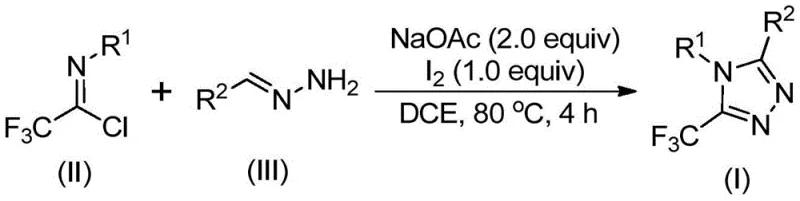
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly those incorporating fluorine motifs. Patent CN110467579B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in current synthetic routes. This technology leverages a non-metallic iodine-promoted cyclization strategy that transforms inexpensive hydrazones and trifluoroethylimidoyl chlorides into high-value heterocyclic scaffolds. For R&D Directors and Process Chemists, this represents a significant leap forward in accessing privileged structures found in numerous bioactive molecules, including antifungal agents and kinase inhibitors. The method's ability to operate under relatively mild thermal conditions without stringent exclusion of air or moisture marks a departure from the capricious nature of many trifluoromethylation protocols, offering a reliable pathway for the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a trifluoromethyl group into a 1,2,4-triazole ring has been fraught with synthetic challenges that hinder large-scale manufacturing. Conventional literature reports primarily describe two approaches: the direct trifluoromethylation of pre-synthesized triazole rings or the cycloaddition of trifluoromethyl synthons. The former often necessitates the use of specialized, expensive, and sometimes hazardous trifluoromethylating reagents, which drastically inflates the cost of goods sold (COGS) and complicates supply chain logistics. The latter approach, while more direct, frequently relies on unstable precursors like trifluorodiazoethane, which poses significant safety risks due to its explosive potential and requires specialized handling equipment. Furthermore, many existing catalytic systems depend on transition metals such as copper or palladium, introducing the risk of heavy metal contamination in the final Active Pharmaceutical Ingredient (API). Removing these trace metals to meet stringent regulatory limits adds complex purification steps, such as scavenging or recrystallization, which reduce overall yield and increase waste generation, making these conventional methods less attractive for cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in Patent CN110467579B offers a streamlined, metal-free alternative that fundamentally reshapes the economic and operational landscape of triazole synthesis. By utilizing trifluoroethylimidoyl chloride and hydrazones as building blocks, the process accesses the triazole core through a constructive annulation rather than a post-functionalization modification. This strategy bypasses the need for dangerous diazo compounds and expensive fluorinating agents. The reaction is promoted by elemental iodine, a benign and inexpensive halogen source, in the presence of a simple base like sodium acetate. Crucially, the protocol does not demand anhydrous or anaerobic conditions, allowing reactions to be performed in standard glassware without the need for inert gas manifolds or gloveboxes. This operational simplicity translates directly into reduced capital expenditure and lower barrier-to-entry for scale-up, positioning this method as a superior choice for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of nucleophilic attack, isomerization, and oxidative aromatization, driven by the unique reactivity of the iodine promoter. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond formation between the hydrazone nitrogen and the imidoyl chloride carbon, generating a trifluoroacetamidine intermediate. This species then undergoes a critical isomerization to align the reactive centers for cyclization. Subsequently, the addition of elemental iodine facilitates an oxidative iodination step, generating an electrophilic iodine species that activates the system for ring closure. This is followed by an intramolecular electrophilic substitution where the nitrogen attacks the activated carbon, closing the five-membered ring. The final step involves the elimination of hydrogen iodide to achieve aromatization, yielding the stable 5-trifluoromethyl-1,2,4-triazole core. Understanding this mechanism is vital for process optimization, as it highlights the dual role of iodine as both an oxidant and a leaving group facilitator.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based trifluoromethylation methods. Because the trifluoromethyl group is pre-installed on the imidoyl chloride starting material, there is no risk of random radical abstraction or multiple fluorinations that often plague late-stage C-H functionalization. The stepwise nature of the cyclization ensures that side reactions are minimized, provided the stoichiometry of the base and iodine is carefully controlled. The use of sodium acetate as a mild base helps to buffer the reaction medium, preventing the decomposition of acid-sensitive functional groups on the aromatic rings. This high level of chemoselectivity is essential for maintaining a clean impurity profile, reducing the burden on downstream purification teams and ensuring that the final product meets the rigorous quality standards required for reliable pharmaceutical intermediate supplier partnerships.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazoles Efficiently
The execution of this synthesis is designed for practicality and reproducibility in a pilot or production plant setting. The process begins by charging a reactor with the requisite molar equivalents of sodium acetate, trifluoroethylimidoyl chloride, and the specific hydrazone derivative in a suitable aprotic solvent such as 1,2-dichloroethane (DCE). The mixture is heated to approximately 80°C and maintained for an initial period to allow for the condensation and initial cyclization events to occur. Following this induction period, elemental iodine is introduced to the reaction vessel to drive the oxidative aromatization to completion. The detailed standardized synthesis steps, including precise stoichiometric ratios, temperature profiles, and workup procedures, are outlined in the guide below.
- Combine sodium acetate, trifluoroethylimidoyl chloride, and hydrazone substrates in an organic solvent such as dichloroethane (DCE).
- Heat the reaction mixture to 80°C and stir for 2 to 4 hours to facilitate the initial condensation and cyclization.
- Add elemental iodine to the system and continue heating for an additional 1 to 2 hours to promote oxidative aromatization.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits that extend beyond mere chemical yield. The shift away from precious metal catalysts and hazardous reagents fundamentally alters the cost structure and risk profile of the manufacturing process. By eliminating the need for expensive palladium or copper catalysts, the direct material costs are significantly reduced, and the complex, costly steps associated with metal scavenging are rendered unnecessary. This simplification of the downstream processing workflow leads to faster batch turnover times and higher throughput, directly impacting the bottom line. Furthermore, the reliance on commodity chemicals like iodine and sodium acetate ensures a stable and resilient supply chain, insulating production schedules from the volatility often seen in the market for specialized organometallic reagents.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the replacement of high-cost inputs with low-cost alternatives. Traditional trifluoromethylation often relies on reagents that are not only expensive to purchase but also require specialized storage and handling infrastructure. By utilizing trifluoroethylimidoyl chlorides and hydrazones, which are derived from widely available aldehydes and amines, the raw material bill of materials is drastically optimized. Additionally, the absence of heavy metals means that the purification process does not require expensive chromatographic resins or activated carbon treatments specifically designed for metal removal. This reduction in processing complexity translates to lower utility consumption and reduced waste disposal costs, contributing to substantial cost savings in pharmaceutical intermediate manufacturing without compromising on product quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for meeting the demanding delivery schedules of global pharmaceutical clients. This synthesis route enhances reliability by utilizing starting materials that are commercially available in bulk quantities from multiple vendors. Unlike proprietary catalysts or unstable diazo compounds that may have long lead times or single-source dependencies, the reagents for this process are commodity chemicals with robust global supply networks. The tolerance of the reaction to ambient moisture and oxygen further reduces the risk of batch failures due to environmental excursions, ensuring consistent output. This operational robustness allows for more accurate forecasting and inventory management, reducing lead time for high-purity pharmaceutical intermediates and strengthening the overall resilience of the supply chain against external disruptions.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the tonne scale often introduces unforeseen challenges, particularly regarding heat management and safety. This iodine-promoted method is inherently scalable because it avoids exothermic hazards associated with diazo decomposition and operates at moderate temperatures. The use of DCE as a solvent, while requiring appropriate recovery systems, is a well-understood industrial solvent with established recycling protocols. Moreover, the metal-free nature of the reaction aligns perfectly with modern green chemistry principles and increasingly stringent environmental regulations regarding heavy metal discharge. The simplified waste stream, devoid of toxic metal residues, facilitates easier treatment and disposal, lowering the environmental compliance burden and supporting sustainable manufacturing practices essential for long-term viability in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on the method's versatility and limitations. Understanding these nuances is critical for project managers evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the primary advantages of this iodine-promoted method over traditional trifluoromethylation?
A: Unlike traditional methods that often require expensive trifluoromethylating reagents or unstable diazo compounds, this method utilizes cheap, commercially available hydrazones and trifluoroethylimidoyl chlorides. Furthermore, it operates under mild conditions without the need for strict anhydrous or anaerobic environments, significantly simplifying operational complexity.
Q: Does this synthesis require toxic heavy metal catalysts?
A: No, a key feature of this patented process is that it is metal-free. It utilizes elemental iodine as a promoter rather than transition metals like palladium or copper, which eliminates the need for costly and difficult heavy metal removal steps in the final purification of pharmaceutical intermediates.
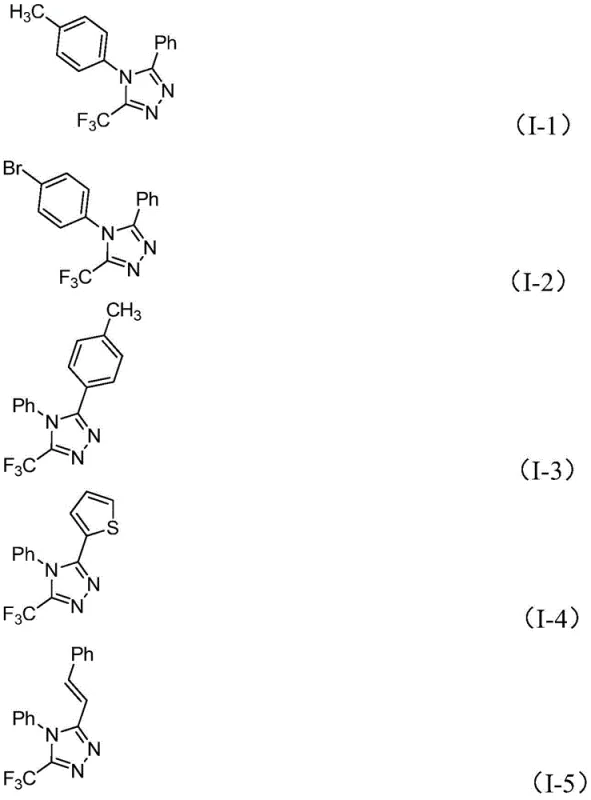
Q: What is the substrate scope for R1 and R2 groups in this triazole synthesis?
A: The method exhibits excellent functional group tolerance. R1 can be various substituted aryl groups (e.g., methyl, methoxy, bromo, trifluoromethyl phenyl), while R2 accommodates alkenyl, aryl, and heteroaryl groups (such as thiophene or pyridine derivatives), allowing for the design of diverse bioactive scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires deep technical expertise and unwavering commitment to quality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific requirements of halogenated heterocyclic synthesis, including corrosion-resistant reactors and advanced solvent recovery systems. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-trifluoromethyl-1,2,4-triazole intermediate meets the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this innovative synthesis technology for your next drug development program. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive quotation. Let us help you accelerate your path to market with a reliable, cost-effective, and scalable supply of high-quality triazole intermediates.
