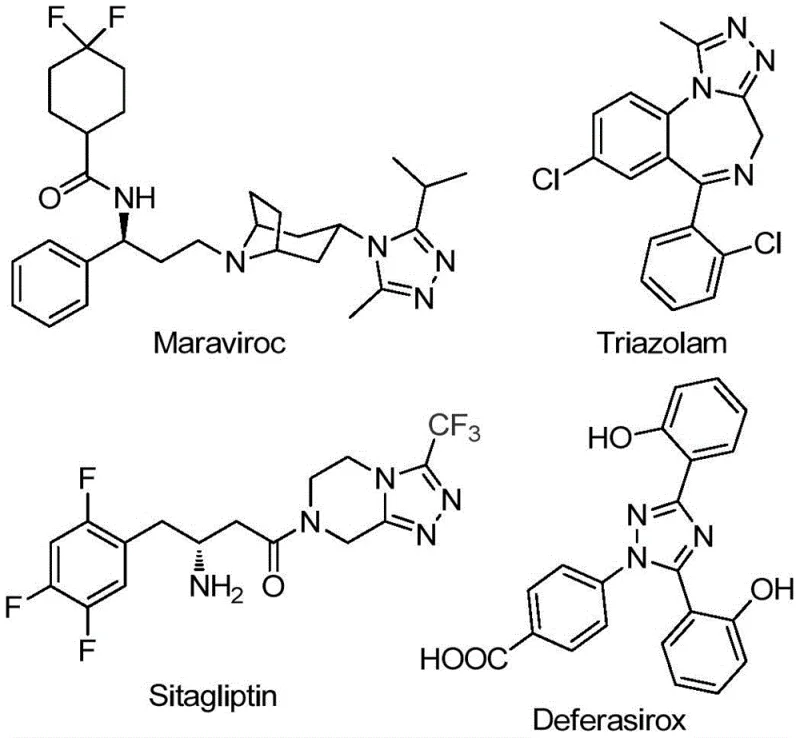
Transforming Pharmaceutical Intermediate Manufacturing: Scalable Production of High-Purity 5-Trifluoromethyl Triazole Derivatives
The Chinese patent CN111978265B discloses a groundbreaking methodology for synthesizing 5-trifluoromethyl substituted 1,2,4-triazole derivatives, addressing critical limitations in current pharmaceutical intermediate production. This innovation introduces an iron-catalyzed cyclization process that operates under ambient atmospheric conditions without requiring anhydrous or oxygen-free environments, representing a significant advancement over conventional synthetic routes. The methodology demonstrates exceptional substrate flexibility across diverse functional groups while maintaining high yield consistency across multiple reaction scales. By utilizing cost-effective catalysts and commercially available starting materials, this approach provides a robust foundation for manufacturing high-purity triazole intermediates essential for next-generation pharmaceutical development. The patent's detailed experimental validation confirms the process's reliability for producing structurally complex derivatives required in modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for trifluoromethyl-substituted triazoles face significant operational constraints that hinder industrial adoption. The first four documented methods—oxadiazole condensation with primary amines, trifluoromethyl hydrazide cyclization with amidines, hydrazone-nitrile cyclization, and oxadiazole hydrazinolysis—all require harsh reaction conditions including elevated temperatures above 100°C or cryogenic environments below -20°C. These processes typically involve multi-step sequences with extended reaction times exceeding 24 hours and demonstrate narrow substrate scope that limits structural diversity. Critically, they consistently deliver low yields ranging from 30% to 60% due to competing side reactions and poor functional group tolerance. The fifth method previously developed by the inventors showed promise but failed completely with alkyl hydrazones, preventing synthesis of crucial 3-alkyl fluoro derivatives required for certain therapeutic applications. These collective limitations create substantial barriers to commercial production including high purification costs, inconsistent quality profiles, and extended manufacturing timelines that directly impact drug development schedules.
The Novel Approach
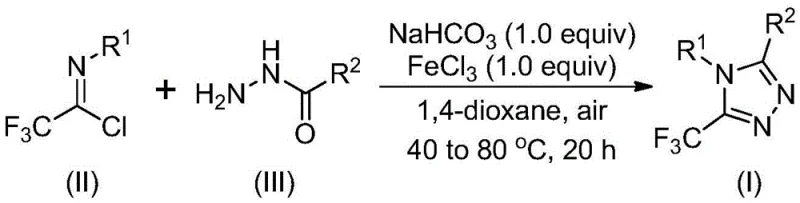
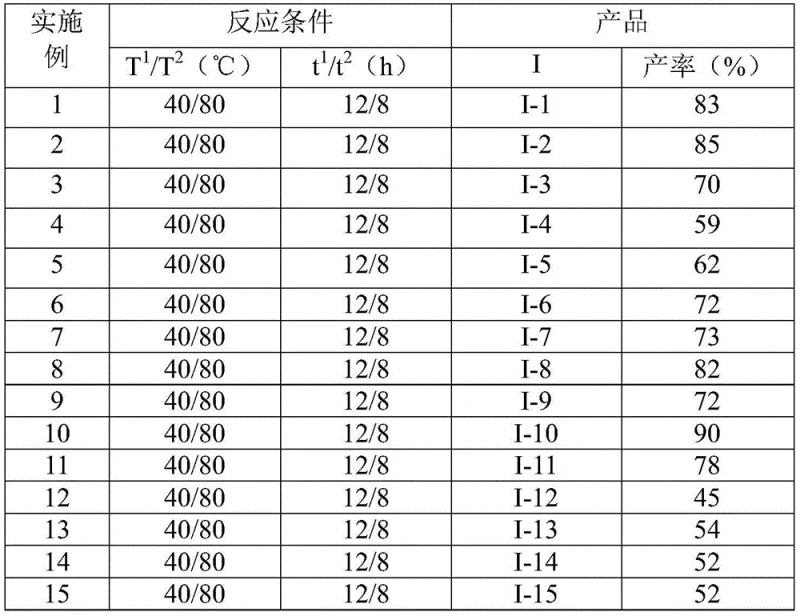
The patented methodology overcomes these challenges through a strategically designed two-stage reaction sequence that operates under remarkably mild conditions. Starting with commercially available hydrazides and trifluoroethyl imidoyl chlorides in aprotic solvents like 1,4-dioxane at temperatures between 30°C and 50°C for the initial phase, followed by FeCl3-catalyzed cyclization at 70°C to 90°C for the second phase. This approach achieves consistent yields between 59% and 90% across diverse substrates as validated in the patent's experimental section. The process eliminates the need for specialized equipment or inert atmospheres while accommodating a wide range of substituents including methyl, methoxy, halogen, and trifluoromethyl groups at various positions on the aromatic rings. Crucially, the methodology demonstrates exceptional scalability from laboratory to industrial production volumes without requiring process re-engineering. The simplified purification protocol involving basic filtration and standard column chromatography significantly reduces manufacturing complexity while maintaining stringent purity requirements essential for pharmaceutical applications. This innovation represents a paradigm shift in triazole derivative synthesis by transforming what was previously a bottleneck process into a streamlined manufacturing operation.
Mechanistic Insights into FeCl3-Catalyzed Triazole Synthesis
The reaction mechanism proceeds through a well-defined dual-stage pathway that explains both the high efficiency and broad substrate tolerance observed in the patent examples. Initially, sodium bicarbonate promotes intermolecular carbon-nitrogen bond formation between the hydrazide and imidoyl chloride to generate trifluoroacetamidine intermediates under mild thermal conditions (40°C). This base-mediated step occurs without requiring anhydrous conditions due to the carbonate's moisture tolerance and occurs within the specified timeframe of 8 to 16 hours as documented in the patent's experimental section. The second stage involves Lewis acid catalysis where FeCl3 facilitates intramolecular dehydration condensation at elevated temperatures (70°C to 90°C), driving the cyclization to form the final triazole ring structure. The iron catalyst's unique electronic properties enable selective activation of the amide nitrogen while tolerating various functional groups that would typically interfere with transition metal catalysts. This mechanistic pathway avoids common side reactions such as hydrolysis or oxidation that plague conventional methods, directly contributing to the observed yield improvements across diverse substrates.
Impurity control is achieved through precise stoichiometric management and temperature programming that minimizes decomposition pathways. The patent specifies exact molar ratios (trifluoroethyl imidoyl chloride : hydrazide : NaHCO3 : FeCl3 = 1:1.5:1:1) that prevent over-reaction or incomplete conversion while maintaining optimal reaction kinetics. The use of non-aqueous workup procedures combined with standard column chromatography effectively removes residual catalysts and unreacted starting materials without requiring specialized purification techniques. Notably, the process generates minimal byproducts as evidenced by the clean NMR spectra provided in the patent examples (e.g., Example 1 showing single product peaks at δ7.35 ppm and δ7.14 ppm in 1H NMR), indicating superior selectivity compared to alternative methodologies. This inherent selectivity reduces downstream processing requirements while ensuring consistent quality profiles essential for pharmaceutical intermediate production.
How to Synthesize 5-Trifluoromethyl Triazole Derivatives Efficiently
This section outlines the practical implementation framework for adopting this patented methodology in commercial manufacturing environments. The process has been validated across multiple scales from laboratory to pilot production with consistent results as demonstrated in the patent's experimental data tables. Detailed operational parameters have been optimized to ensure reproducibility while maintaining flexibility for different substrate requirements.
- Prepare reaction mixture with hydrazide and trifluoroethyl imidoyl chloride in aprotic solvent under ambient conditions
- Execute iron-catalyzed cyclization at controlled temperature with precise stoichiometric ratios
- Implement simplified purification protocol to achieve pharmaceutical-grade purity specifications
Step-by-Step Synthesis Guide
Commercial Advantages for Procurement and Supply Chain Teams
The patented methodology delivers substantial operational improvements that directly address critical pain points in pharmaceutical intermediate supply chains. By eliminating specialized equipment requirements and simplifying process parameters, this approach creates significant opportunities for cost optimization while enhancing supply reliability through more robust manufacturing processes.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and specialized reaction environments significantly reduces raw material costs while avoiding complex catalyst removal steps required in alternative processes. The use of commercially available starting materials at optimal stoichiometric ratios (as shown in Table 1) minimizes waste generation and improves atom economy without requiring costly purification infrastructure. This streamlined approach substantially lowers overall manufacturing costs through reduced processing complexity and energy consumption while maintaining high product quality standards required for pharmaceutical applications.
- Enhanced Supply Chain Reliability: The process operates effectively under standard atmospheric conditions without requiring inert gas environments or ultra-pure solvents, significantly reducing dependency on specialized supply chain components that often cause production delays. The demonstrated scalability from gram-scale reactions to potential industrial volumes (as noted in the patent's description) ensures consistent supply continuity without requiring major process re-engineering during scale-up phases. This reliability is further enhanced by the broad availability of starting materials through multiple global suppliers, creating a resilient supply chain foundation that minimizes disruption risks associated with single-source dependencies.
- Scalability and Environmental Compliance: The methodology's compatibility with standard manufacturing equipment enables seamless transition from laboratory to commercial production volumes without requiring specialized infrastructure investments. The simplified waste stream profile resulting from reduced byproduct formation lowers environmental impact while decreasing waste treatment costs associated with complex purification processes required by conventional methods. This environmentally favorable profile supports regulatory compliance in multiple jurisdictions while aligning with growing industry sustainability requirements without compromising on product quality or manufacturing efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial considerations regarding implementation of this patented synthesis methodology based on specific details from the patent documentation.
Q: Why do conventional methods for synthesizing trifluoromethyl triazoles often yield low purity and require harsh conditions?
A: Traditional approaches like oxadiazole condensation or hydrazone cyclization suffer from narrow substrate tolerance and require extreme temperatures or anhydrous environments, leading to inconsistent purity profiles and complex purification needs as documented in prior art limitations.
Q: How does the FeCl3-catalyzed process achieve higher substrate tolerance compared to previous methodologies?
A: The dual-stage mechanism (base-promoted C-N bond formation followed by Lewis acid-mediated cyclization) accommodates diverse functional groups including alkyl, aryl, and halogen substituents without requiring specialized protection/deprotection steps, as evidenced by the broad substrate scope in patent examples.
Q: What makes this iron-catalyzed synthesis particularly suitable for commercial scale-up of pharmaceutical intermediates?
A: The process operates under air without anhydrous requirements, uses cost-effective catalysts at stoichiometric ratios (FeCl3 at 0.3 mmol), and demonstrates consistent yields (59%-90%) across gram-scale reactions with straightforward column chromatography purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Triazole Derivatives Supplier
While iron-catalyzed methodologies represent significant advancements in heterocyclic chemistry, NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production for complex pharmaceutical intermediates. Our rigorous QC labs ensure stringent purity specifications are consistently met through advanced analytical capabilities specifically designed for challenging heterocyclic compounds like trifluoromethyl triazoles. As a CDMO partner specializing in fine chemical synthesis, we possess the technical expertise to adapt this patented approach to specific client requirements while maintaining all critical quality attributes essential for regulatory compliance.
We invite you to initiate a Customized Cost-Saving Analysis for your specific triazole intermediate requirements by contacting our technical procurement team. They will provide detailed route feasibility assessments and specific COA data demonstrating how our manufacturing capabilities can optimize your supply chain while meeting your exact quality specifications.
