Scalable Production of 2-Trifluoromethyl Imidazoles via Mild Palladium Catalysis
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access fluorinated heterocycles, which serve as critical scaffolds in modern drug design. Patent CN111423381B introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing the growing demand for metabolically stable and lipophilic molecular building blocks. The introduction of a trifluoromethyl group into a heterocyclic core often dramatically enhances the bioavailability and electronegativity of the parent molecule, making these compounds highly desirable for medicinal chemistry programs targeting complex biological pathways. As illustrated in the structural diversity of bioactive molecules below, the imidazole motif is pervasive in high-value therapeutics and functional materials.
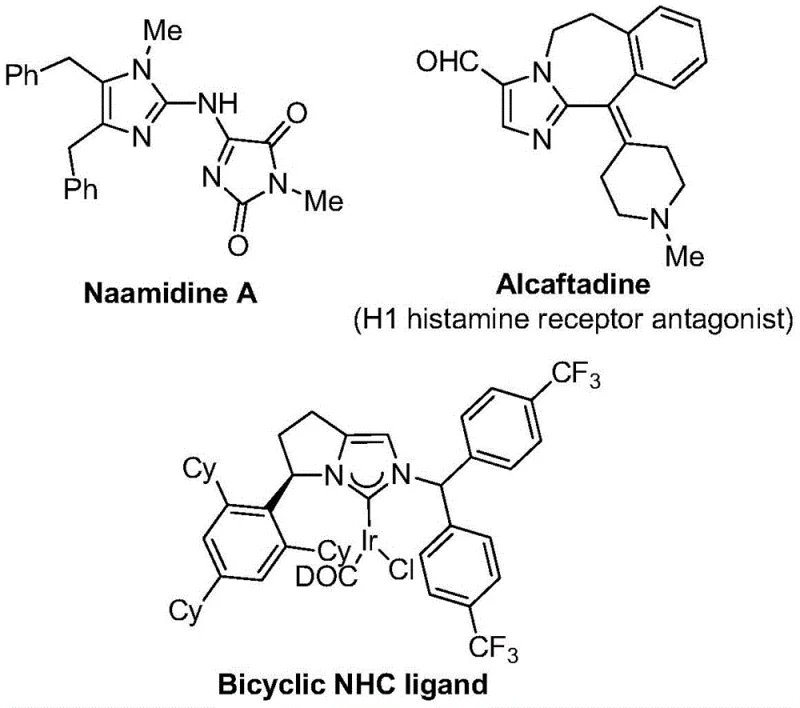
This patented technology leverages a transition metal palladium-catalyzed carbonylation series reaction, utilizing cheap and easily obtained starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts. Unlike traditional methods that may rely on unstable or hazardous reagents, this approach offers a streamlined pathway to diverse 2-trifluoromethyl imidazoles with high reaction efficiency and excellent substrate compatibility. For procurement managers and supply chain heads, this represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity compounds through a scalable and economically viable process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has been fraught with challenges related to safety, cost, and operational complexity. Conventional literature often reports the direct reaction of synthons like trifluorodiazoethane with suitable substrates, a method that poses significant safety hazards due to the explosive nature of diazo compounds. Furthermore, existing protocols frequently require harsh reaction conditions, including elevated temperatures or the use of expensive, specialized catalysts that are difficult to remove from the final product, leading to increased purification costs and potential heavy metal contamination. These factors collectively hinder the commercial scale-up of complex pharmaceutical intermediates, creating bottlenecks in the supply chain for API manufacturers who require consistent quality and volume.
The Novel Approach
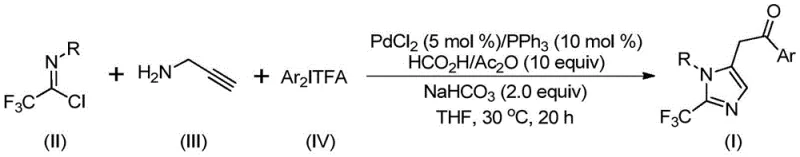
The methodology disclosed in CN111423381B fundamentally shifts the paradigm by employing a mild, palladium-catalyzed multicomponent reaction that operates efficiently at just 30°C. By utilizing trifluoroethylimidoyl chloride and diaryl iodonium salts as key precursors, the process avoids the need for dangerous diazo reagents while maintaining high atom economy. The reaction scheme below highlights the convergence of three distinct components—imidoyl chloride, propargylamine, and iodonium salt—into a single pot, facilitated by a carbon monoxide surrogate generated in situ from formic acid and acetic anhydride. This novel approach not only simplifies the operational workflow but also broadens the utility of the method, allowing for the synthesis of多样化 substituted imidazole derivatives through simple substrate design.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation is a sophisticated orchestration of organometallic steps that ensures high selectivity and yield. Initially, an intermolecular carbon-nitrogen bond is formed under alkaline promotion to generate a trifluoroacetamidine intermediate, which subsequently undergoes isomerization. The palladium catalyst then engages in a palladation of the alkyne moiety of the propargylamine derivative, forming a crucial alkenyl palladium intermediate. This species further isomerizes to an alkyl palladium intermediate, setting the stage for the carbonylation event. The carbon monoxide required for this step is elegantly released from the formic acid/acetic anhydride mixture, reacting with the palladium center to form an acyl palladium intermediate. Finally, oxidative addition of the diaryl iodonium salt generates a tetravalent palladium species, which undergoes reductive elimination to release the final 2-trifluoromethyl-substituted imidazole product and regenerate the active catalyst.
From a quality control perspective, this mechanism offers inherent advantages in impurity management. The use of specific ligands like triphenylphosphine and the controlled generation of CO prevent the formation of uncontrolled polymerization byproducts often seen in free-radical processes. The mild reaction temperature of 30°C further suppresses thermal degradation pathways, ensuring a cleaner crude reaction profile. This level of mechanistic control is vital for R&D directors focused on purity and杂质谱 (impurity profiles), as it minimizes the burden on downstream purification units and ensures that the final high-purity OLED material or API intermediate meets stringent regulatory specifications without extensive recrystallization cycles.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The synthesis protocol described in the patent is designed for operational simplicity, making it highly attractive for both laboratory optimization and pilot plant operations. The process involves mixing the palladium catalyst system with the three key organic precursors in a solvent like tetrahydrofuran (THF), followed by a prolonged but low-energy incubation period. The detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below, ensuring that technical teams can rapidly validate the process for their specific substrate requirements.
- Combine palladium chloride (5 mol %), triphenylphosphine (10 mol %), sodium bicarbonate, acetic anhydride, and formic acid in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 18 to 20 hours, then filter and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere chemical novelty. The shift towards milder reaction conditions and safer reagents directly translates to reduced operational risks and lower overhead costs associated with safety compliance and waste disposal. By eliminating the need for high-pressure equipment or cryogenic cooling, facilities can utilize existing standard reactor infrastructure, thereby accelerating the timeline from process development to commercial production. This adaptability is crucial for maintaining supply continuity in a volatile global market.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of commodity chemicals such as propargylamine and palladium chloride, which are significantly cheaper than specialized organometallic reagents. The elimination of hazardous diazo compounds removes the need for expensive safety containment systems and specialized waste treatment protocols, leading to substantial cost savings in overall manufacturing operations. Furthermore, the high conversion rates observed across various substrates minimize raw material waste, optimizing the cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including aromatic amines and aryl boronic acids (precursors to the iodonium salts), are widely available from global chemical suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by utility fluctuations, such as minor variations in cooling water temperature. This reliability ensures a steady flow of high-purity pharmaceutical intermediates to downstream customers, supporting just-in-time manufacturing models.
- Scalability and Environmental Compliance: The process is explicitly noted for its scalability to gram levels and potential for industrial application, with simple post-processing steps like filtration and silica gel chromatography. The use of THF as a solvent, while requiring recovery, is well-established in industrial settings with mature recycling technologies. Additionally, the avoidance of heavy stoichiometric oxidants and the use of catalytic amounts of palladium align with green chemistry principles, facilitating easier environmental permitting and reducing the ecological footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the method's capabilities and limitations for potential partners.
Q: What are the key advantages of this Pd-catalyzed method over traditional diazo routes?
A: This method avoids the use of hazardous trifluorodiazoethane, utilizing safer trifluoroethylimidoyl chloride instead. It operates at a mild 30°C, significantly reducing energy consumption and safety risks associated with high-pressure or high-temperature reactions.
Q: What is the substrate scope for the aryl groups in this synthesis?
A: The process demonstrates excellent compatibility with various substituents including methyl, tert-butyl, methoxy, chloro, bromo, trifluoromethyl, and nitro groups on both the imidoyl chloride and the diaryl iodonium salt components.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is scalable to gram levels and suitable for industrial application due to the use of cheap, commercially available starting materials and simple post-processing procedures like filtration and chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has extensively evaluated the pathway described in CN111423381B and confirmed its potential for generating diverse libraries of fluorinated heterocycles. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from benchtop curiosity to market-ready reality. Our state-of-the-art rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of 2-trifluoromethyl imidazole delivered meets the highest international standards.
We invite you to collaborate with us to leverage this innovative chemistry for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in palladium-catalyzed carbonylation can drive efficiency and value for your organization.
