Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, scalable, and cost-effective synthetic routes. A significant breakthrough in this domain is detailed in patent CN111423381B, which discloses a novel preparation method for 2-trifluoromethyl substituted imidazole compounds. These heterocyclic scaffolds are critical building blocks in modern medicinal chemistry, often found in the molecular backbones of drugs with potent biological activities. The introduction of a trifluoromethyl group significantly enhances electronegativity, metabolic stability, and lipophilicity, making these compounds highly desirable for drug discovery programs. This patent outlines a transition metal palladium-catalyzed carbonylation series reaction that utilizes cheap and easily obtained starting materials, marking a substantial shift away from traditional, more hazardous synthetic methodologies.
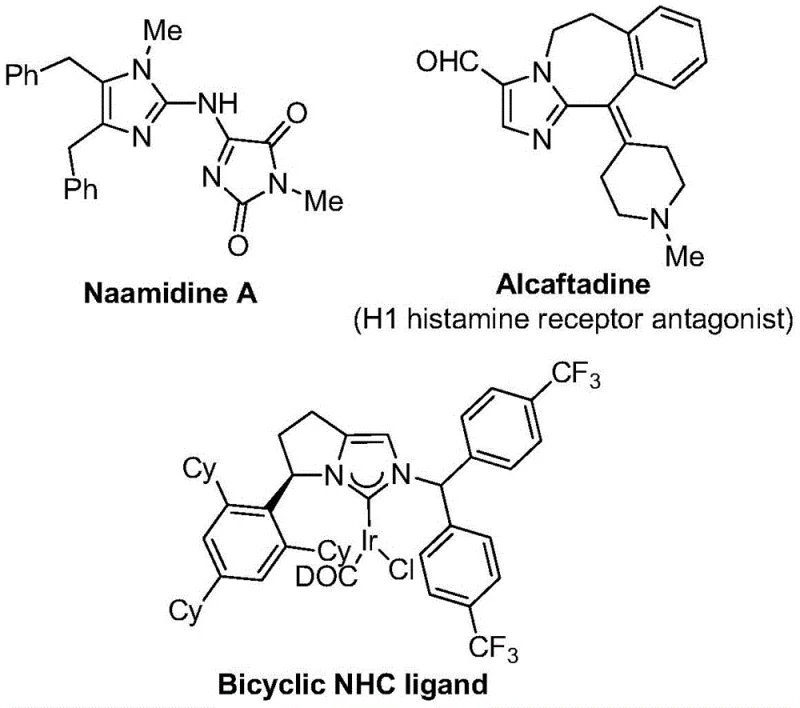
The strategic importance of this technology cannot be overstated for R&D directors and procurement managers alike. By leveraging a mild reaction condition of 30°C and a robust catalytic system, this method addresses key pain points in the supply chain of high-purity pharmaceutical intermediates. The ability to synthesize diversely substituted imidazole compounds through substrate design not only broadens the utility of this method but also ensures a reliable supply of specialized chemical entities required for next-generation therapeutics. As a leading entity in the fine chemical sector, understanding and adopting such innovative pathways is crucial for maintaining competitiveness in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has been fraught with challenges that hinder large-scale commercialization. Conventional methods often rely on direct reactions between synthons containing trifluoromethyl groups and suitable substrates, frequently utilizing reagents like trifluorodiazoethane or trifluoroethylimidoyl halides. While these reagents have application potential, their widespread use has been limited due to issues regarding availability, stability, and safety. Many traditional protocols require harsh reaction conditions, including elevated temperatures and pressures, which can lead to decomposition of sensitive functional groups and lower overall yields. Furthermore, the handling of gaseous carbon monoxide in traditional carbonylation reactions poses significant safety risks and requires specialized high-pressure equipment, increasing both capital expenditure and operational complexity for manufacturing facilities.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN111423381B introduces a streamlined, palladium-catalyzed approach that fundamentally simplifies the synthetic workflow. This novel route employs trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts as the core starting materials. The reaction proceeds efficiently at a remarkably mild temperature of 30°C, eliminating the need for energy-intensive heating or cryogenic cooling. Crucially, the method utilizes a combination of formic acid and acetic anhydride as a safe and convenient alternative to gaseous carbon monoxide, generating the necessary CO in situ. This innovation drastically reduces safety hazards and equipment requirements. The general reaction scheme illustrates the convergence of these three components into the desired 2-trifluoromethyl imidazole scaffold with high atom economy.
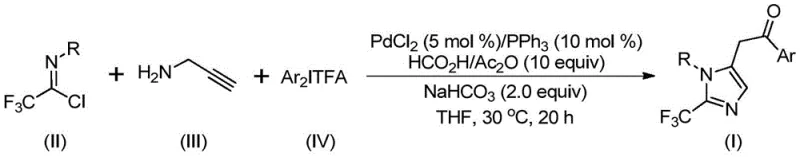
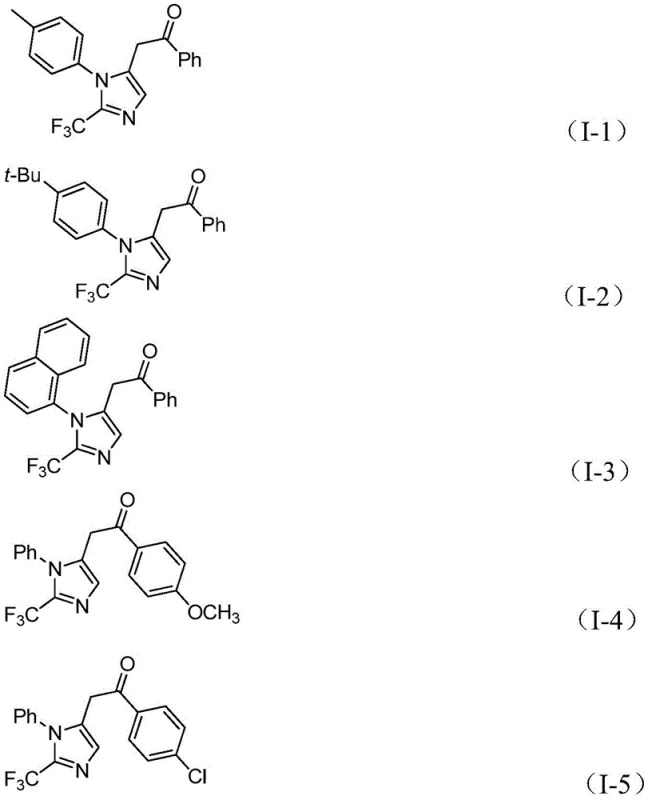
The versatility of this approach is demonstrated by its excellent substrate compatibility. As shown in the specific examples provided in the patent, a wide range of aryl groups with various substituents—such as methyl, tert-butyl, chlorine, bromine, trifluoromethyl, and nitro groups—can be successfully incorporated into the final product. This flexibility allows chemists to access a diverse library of compounds, such as those depicted in structures I-1 through I-5, facilitating rapid structure-activity relationship (SAR) studies without the need to develop new synthetic routes for each analog. The simplicity of the post-treatment process, involving filtration and standard column chromatography, further underscores the practicality of this method for both laboratory research and industrial production.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
For R&D directors focused on process robustness and impurity control, understanding the mechanistic underpinnings of this transformation is vital. The reaction is believed to proceed through a sophisticated catalytic cycle initiated by the formation of an intermolecular carbon-nitrogen bond promoted by the base, sodium bicarbonate. This initial step yields a trifluoroacetamidine intermediate, which subsequently undergoes isomerization. The palladium catalyst, generated from palladium chloride and triphenylphosphine, then engages with the alkyne moiety of the propargylamine derivative. This palladation event forms an alkenyl palladium intermediate, which isomerizes to a more stable alkyl palladium species. The critical carbonylation step follows, where carbon monoxide—released from the formic acid/acetic anhydride mixture—inserts into the palladium-carbon bond to generate an acyl palladium intermediate.
The cycle concludes with the oxidative addition of the diaryl iodonium salt to the palladium center, forming a transient tetravalent palladium intermediate. Finally, reductive elimination occurs, releasing the final 2-trifluoromethyl-substituted imidazole product and regenerating the active palladium catalyst. This intricate dance of organometallic steps ensures high selectivity and minimizes the formation of side products. The use of sodium bicarbonate as a mild base helps neutralize acidic byproducts without promoting unwanted hydrolysis of the sensitive imidoyl chloride or the final product. This mechanistic clarity allows for precise tuning of reaction parameters, ensuring that impurity profiles remain within strict specifications required for pharmaceutical applications. The high yields reported, ranging up to 97% for certain substrates, attest to the efficiency of this catalytic manifold.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis in a practical setting requires adherence to specific protocols to maximize yield and purity. The process is designed to be user-friendly, utilizing reagents that are generally commercially available or easily prepared. The reaction is typically conducted in tetrahydrofuran (THF), which effectively dissolves all reactants and promotes the catalytic cycle. The molar ratios are carefully optimized, with a preference for a slight excess of trifluoroethylimidoyl chloride and diaryl iodonium salt relative to propargylamine to drive the reaction to completion. The detailed standardized synthesis steps below outline the precise addition order and workup procedures necessary to achieve the high-quality results described in the patent literature.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, formic acid, trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt in an organic solvent such as THF.
- Stir the reaction mixture at a mild temperature of 30°C for a duration of 16 to 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl substituted imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere chemical curiosity. The shift towards this palladium-catalyzed method addresses several critical bottlenecks in the sourcing of complex heterocyclic intermediates. By utilizing starting materials that are cheap and widely available in the chemical market, such as aromatic amines and propargylamine, the dependency on exotic or monopolized reagents is significantly reduced. This diversification of the raw material base enhances supply chain resilience, mitigating the risk of disruptions that can occur when relying on single-source suppliers for specialized synthons. Furthermore, the operational simplicity of the process means that it can be executed in standard glass-lined reactors without the need for specialized high-pressure infrastructure.
- Cost Reduction in Manufacturing: The economic implications of this method are profound. By eliminating the need for high-pressure carbon monoxide gas and the associated safety measures, capital and operational expenditures are drastically lowered. The use of palladium chloride, while a precious metal, is employed at low loading (5 mol%), and the ligand triphenylphosphine is inexpensive. Moreover, the high reaction efficiency and yields mean less raw material is wasted, and downstream purification costs are minimized due to cleaner reaction profiles. The avoidance of cryogenic conditions or extreme heating further reduces utility costs, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent delivery timelines. Since the reaction tolerates a wide range of functional groups and proceeds under mild conditions, batch-to-batch variability is minimized. This reliability is crucial for maintaining continuous production schedules for downstream API manufacturing. The ability to scale the reaction from gram levels to industrial quantities without significant re-optimization provides a clear pathway for meeting increasing demand. Suppliers adopting this method can offer more reliable lead times, as the process is less susceptible to the delays often caused by complex safety checks or difficult-to-source reagents.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns well with green chemistry principles. The in situ generation of carbon monoxide avoids the transport and storage of toxic gases, reducing the facility's environmental footprint. The solvent system, primarily THF, is well-understood and can be efficiently recovered and recycled. The simple workup procedure involving filtration and chromatography reduces the volume of aqueous waste generated compared to multi-step extraction processes. These factors facilitate easier compliance with increasingly stringent environmental regulations, ensuring long-term operational sustainability and reducing the risk of regulatory shutdowns.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries based on the patent data. These questions address the practical aspects of implementation, including catalyst selection, substrate limitations, and purification strategies. Understanding these nuances is essential for integrating this synthetic route into existing production workflows or R&D pipelines. The following insights are derived directly from the experimental data and technical disclosures provided in the patent documentation.
Q: What are the key advantages of this palladium-catalyzed method over traditional synthesis?
A: This method operates under mild conditions (30°C) using cheap and readily available starting materials like propargylamine and diaryl iodonium salts. It avoids harsh reagents and offers excellent functional group tolerance, making it superior for complex molecule synthesis.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the patent explicitly states the method is expandable to the gram level and provides possibilities for industrial large-scale production due to its simple operation, high reaction efficiency, and use of common organic solvents like THF.
Q: What is the role of formic acid and acetic anhydride in this reaction?
A: Formic acid and acetic anhydride act as a carbon monoxide alternative (CO source). They generate CO in situ, which is essential for the carbonylation step that constructs the imidazole ring backbone without requiring high-pressure CO gas cylinders.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
The technological advancements detailed in patent CN111423381B represent a significant opportunity for the pharmaceutical and fine chemical industries to optimize their supply chains. At NINGBO INNO PHARMCHEM, we recognize the value of such innovative synthetic methodologies and have integrated similar state-of-the-art capabilities into our manufacturing portfolio. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global clients. We are committed to delivering high-purity intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation to verify every batch.
We invite you to explore how our expertise in palladium-catalyzed transformations can benefit your upcoming projects. Whether you require custom synthesis of novel imidazole derivatives or scale-up of existing routes, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how we can support your journey from discovery to commercial success.
