Advanced Catalytic Route for Scalable 3-Trifluoromethyl-1,2,4-Triazole Production
Advanced Catalytic Route for Scalable 3-Trifluoromethyl-1,2,4-Triazole Production
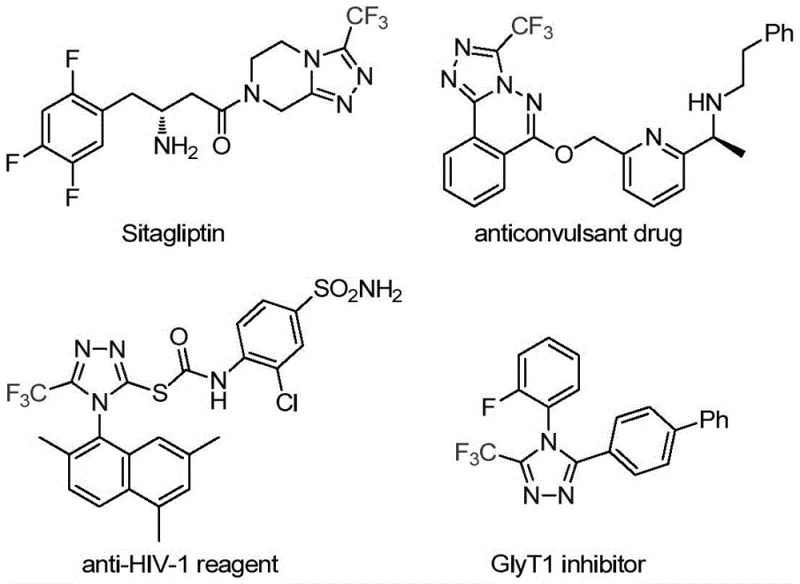
The pharmaceutical industry continuously seeks robust synthetic methodologies to access complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. A significant breakthrough in this domain is detailed in Chinese patent CN113307778A, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These nitrogen-containing heterocycles are ubiquitous in medicinal chemistry, found in prominent drugs such as Sitagliptin and various anticonvulsants, where the trifluoromethyl group plays a pivotal role in enhancing metabolic stability and lipophilicity. The disclosed technology leverages a novel dual-catalytic system involving molybdenum hexacarbonyl and cuprous acetate to drive a cycloaddition reaction between trifluoroethylimidoyl chloride and functionalized isonitriles. This approach represents a paradigm shift from traditional multi-step syntheses, offering a streamlined, one-pot solution that operates under relatively mild thermal conditions, thereby addressing key pain points for both process chemists and supply chain managers seeking reliable pharmaceutical intermediate supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazole core, particularly when substituted with electron-withdrawing trifluoromethyl groups, has been fraught with synthetic challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional literature methods often rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These legacy routes frequently suffer from harsh reaction conditions, requiring strong acids or bases that can degrade sensitive functional groups, leading to complex impurity profiles that are difficult to purge. Furthermore, alternative copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane pose significant safety hazards due to the explosive nature of diazo compounds, creating substantial bottlenecks in manufacturing environments. The reliance on unstable or hazardous reagents not only increases the cost of goods sold through specialized handling requirements but also introduces variability in batch-to-batch consistency, a critical metric for quality assurance teams.
The Novel Approach
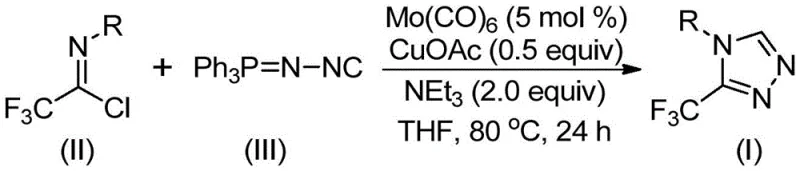
In stark contrast to these cumbersome legacy protocols, the methodology described in CN113307778A utilizes a sophisticated yet operationally simple co-catalytic system to forge the triazole ring with exceptional precision. By employing trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as stable, commercially accessible starting materials, the process eliminates the need for dangerous diazo reagents entirely. The reaction proceeds smoothly in tetrahydrofuran (THF) at temperatures between 70°C and 90°C, typically optimized at 80°C, which is well within the operational range of standard glass-lined reactors found in most fine chemical facilities. As illustrated in the reaction scheme below, this transformation achieves high atom economy and delivers the target 3-trifluoromethyl-1,2,4-triazole scaffold in yields reaching up to 99% for certain substrates, such as the 4-methylthio phenyl derivative. This dramatic improvement in efficiency translates directly into reduced waste generation and lower solvent consumption, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into Mo/Cu Co-catalyzed Cycloaddition
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors evaluating the robustness of the pathway for high-purity OLED material or API intermediate production. The reaction initiates with the activation of the functionalized isonitrile by molybdenum hexacarbonyl, forming a reactive metal-isocyanide complex that lowers the energy barrier for subsequent bond formation. Concurrently, cuprous acetate acts as a Lewis acid promoter, facilitating the nucleophilic attack of the activated isonitrile species onto the electrophilic carbon of the trifluoroethylimidoyl chloride. This synergistic interaction drives a formal [3+2] cycloaddition, constructing the five-membered triazole ring intermediate with high regioselectivity. The final step involves the elimination of triphenylphosphine oxide, likely assisted by trace water or the basic environment provided by triethylamine, to aromatize the ring and yield the stable triazole product. This mechanistic clarity ensures that process engineers can predict potential side reactions and optimize parameters to minimize byproduct formation.
From an impurity control perspective, the mildness of the triethylamine base and the moderate temperature profile are instrumental in preserving the integrity of sensitive substituents on the aromatic ring. Unlike strongly acidic cyclizations that might cause dehalogenation or ester hydrolysis, this neutral-to-basic catalytic environment tolerates a wide array of functional groups, including halogens, nitro groups, and thioethers. The patent data demonstrates this versatility across a broad substrate scope, where electron-rich and electron-deficient aryl groups alike are converted efficiently. For instance, substrates bearing electron-donating groups like tert-butyl or methoxy proceed with high conversion, while those with electron-withdrawing nitro or chloro groups also react successfully, albeit with slightly varied kinetics. This broad tolerance simplifies the purification workflow, as fewer degradation products are generated, allowing for simpler crystallization or chromatographic purification steps to achieve the stringent purity specifications required for clinical candidates.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for seamless integration into existing laboratory and pilot plant workflows, minimizing the need for specialized equipment or exotic reagents. The protocol dictates a straightforward charging sequence where all components—catalysts, base, and substrates—are combined in an aprotic solvent, preferably THF, which offers superior solubility for the polar intermediates. The reaction is then heated to reflux or maintained at 80°C for a period of 18 to 30 hours, ensuring complete consumption of the limiting reagent. Following the reaction, the workup is remarkably simple, involving filtration to remove metal residues followed by adsorption onto silica gel for final purification. This operational simplicity reduces the technical burden on operators and shortens the overall cycle time per batch. For a detailed breakdown of the specific stoichiometric ratios and step-by-step operational parameters, please refer to the standardized guide below.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), trifluoroethylimidoyl chloride, and functionalized isonitrile in THF solvent.
- Heat the reaction mixture to 80°C and stir for 24 hours under inert atmosphere to ensure complete conversion.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and logistical benefits that extend far beyond mere chemical yield. The primary driver for cost reduction in pharmaceutical intermediate manufacturing lies in the replacement of expensive or hazardous reagents with commodity chemicals that are readily available in the global market. By utilizing trifluoroethylimidoyl chloride and functionalized isonitriles, which can be sourced from multiple vendors, the supply chain becomes more resilient against single-source disruptions. Furthermore, the elimination of cryogenic conditions or high-pressure hydrogenation steps significantly lowers the utility costs associated with heating, cooling, and pressure containment, contributing to a leaner cost structure. The ability to run the reaction in common solvents like THF also simplifies solvent recovery and recycling processes, further driving down the variable costs per kilogram of produced material.
- Cost Reduction in Manufacturing: The economic viability of this process is underscored by the use of earth-abundant metal catalysts rather than precious metals like palladium or rhodium, which are subject to volatile market pricing. Cuprous acetate and molybdenum hexacarbonyl are relatively inexpensive compared to noble metal catalysts, and their usage at low loadings (5 mol% and 0.5 equiv respectively) minimizes the financial impact of metal loss. Additionally, the high reaction efficiency observed, with yields frequently exceeding 80-90% for optimized substrates, means that less raw material is wasted as byproduct, directly improving the mass balance and reducing the cost of goods sold. The simplified post-treatment procedure, which avoids complex aqueous workups or multiple extraction cycles, reduces labor hours and solvent consumption, compounding the savings throughout the production lifecycle.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for maintaining production schedules, and this methodology enhances reliability by relying on stable, shelf-stable starting materials. Unlike diazonium salts or diazo compounds which require immediate preparation and careful handling due to instability, the imidoyl chlorides and isonitriles used here are robust solids or liquids that can be stocked in inventory without significant degradation risks. This stability allows for better demand forecasting and bulk purchasing strategies, shielding the manufacturing operation from short-term market fluctuations. Moreover, the scalability of the reaction from gram-scale discovery to multi-kilogram production has been explicitly validated, ensuring that the transition from R&D to commercial manufacturing does not encounter unforeseen kinetic or thermodynamic barriers that often plague scale-up campaigns.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental footprint of a synthetic route becomes a critical selection criterion. This Mo/Cu co-catalyzed process generates minimal hazardous waste, primarily consisting of spent catalyst residues and solvent, both of which are manageable through standard waste treatment protocols. The absence of heavy metal contaminants like mercury or lead simplifies the purification of the final API, reducing the burden on analytical QC labs to demonstrate compliance with ICH Q3D guidelines for elemental impurities. The high atom economy of the cycloaddition reaction ensures that the majority of the input mass ends up in the desired product, aligning with sustainability goals and reducing the volume of effluent that requires treatment. This eco-friendly profile not only mitigates regulatory risk but also enhances the corporate social responsibility standing of the manufacturing entity.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of adopting this technology, we have compiled answers to common inquiries regarding the reaction parameters and substrate compatibility. These insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy and relevance for process development scientists. Understanding the nuances of catalyst loading and solvent effects is essential for troubleshooting and optimizing the reaction for specific novel substrates that may fall outside the initial scope. We encourage partners to review these details closely when designing their own experimental matrices.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies using THF as the preferred organic solvent at a temperature range of 70-90°C, typically optimized at 80°C for 24 hours, utilizing Mo(CO)6 and CuOAc as the catalytic system.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates excellent substrate tolerance, successfully synthesizing derivatives with methyl, methoxy, fluoro, chloro, nitro, and naphthyl substituents with yields ranging from moderate to excellent.
Q: Is this process suitable for large-scale manufacturing?
A: The invention explicitly states that the method can be expanded to gram-level reactions and beyond, utilizing cheap and commercially available starting materials, making it highly viable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of new therapeutic agents depends on the availability of high-quality, consistently supplied intermediates. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN113307778A and is fully equipped to translate this academic innovation into a robust industrial process. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are designed to handle fluorinated chemistry safely and efficiently, adhering to stringent purity specifications and rigorous QC labs protocols to guarantee that every batch meets the exacting standards required for clinical and commercial applications.
We invite you to collaborate with us to leverage this advanced synthetic route for your upcoming projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We are ready to provide specific COA data and route feasibility assessments to demonstrate how this Mo/Cu catalyzed method can accelerate your development timeline while optimizing your budget. Contact us today to discuss how we can support your supply chain with reliable, high-performance pharmaceutical intermediates.
