Advanced Mo-Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial API Production
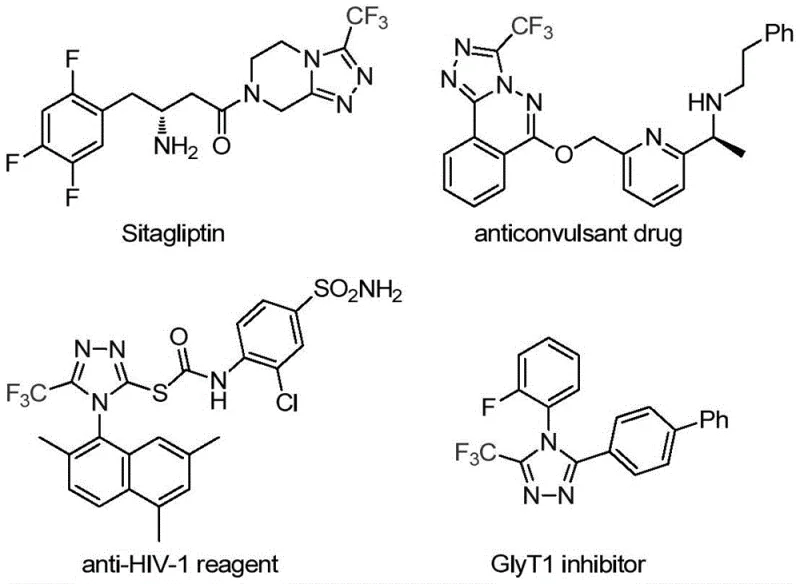
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are pivotal for enhancing metabolic stability and bioavailability in modern drug candidates. Patent CN113307778A introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in the synthesis of these valuable scaffolds. As illustrated in the structural diversity of bioactive molecules below, the 1,2,4-triazole core is a fundamental building block found in numerous approved drugs such as Sitagliptin, underscoring the immense commercial value of mastering its functionalization.
This novel methodology leverages a synergistic molybdenum and copper co-catalytic system to facilitate the cycloaddition of trifluoroethylimidoyl chloride with functionalized isonitriles. By operating under mild thermal conditions between 70°C and 90°C, the process circumvents the safety hazards and energy intensity associated with conventional high-temperature cyclizations. For procurement managers and supply chain directors, this represents a significant opportunity for cost reduction in API manufacturing, as the protocol utilizes readily accessible reagents and avoids the need for exotic or prohibitively expensive transition metal complexes that often plague fine chemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on methodologies that present substantial operational and safety challenges for large-scale production. Traditional routes frequently involve the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones, processes that often necessitate the handling of unstable hydrazine derivatives and generate significant quantities of hazardous waste. Furthermore, alternative copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane introduce severe safety risks due to the explosive nature of diazo compounds, requiring specialized equipment and rigorous safety protocols that inflate capital expenditure. These legacy methods often suffer from narrow substrate scope and poor atom economy, leading to lower overall yields and complicating the purification of the final active pharmaceutical ingredient, thereby increasing the cost of goods sold for downstream manufacturers.
The Novel Approach
In stark contrast, the invention disclosed in CN113307778A offers a streamlined, one-pot synthetic strategy that dramatically simplifies the production workflow while enhancing safety profiles. By employing trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as the primary building blocks, the reaction proceeds through a highly efficient [3+2] cycloaddition pathway mediated by molybdenum hexacarbonyl and cuprous acetate. This approach eliminates the need for dangerous diazo reagents and harsh hydrazine conditions, replacing them with stable, commercially available starting materials that can be sourced reliably from global chemical suppliers. The reaction conditions are remarkably forgiving, tolerating a wide array of functional groups on the aromatic ring, which allows medicinal chemists to rapidly generate diverse libraries of analogues without redesigning the entire synthetic route, thus accelerating the drug discovery timeline.
Mechanistic Insights into Mo-Cu Co-Catalyzed Cycloaddition
The mechanistic elegance of this transformation lies in the cooperative interplay between the molybdenum and copper species, which activates the relatively inert isonitrile functionality towards nucleophilic attack. As depicted in the general reaction scheme below, molybdenum hexacarbonyl acts as a potent metal activator that coordinates with the functionalized isonitrile to form a reactive metal-carbene-like complex, lowering the activation energy for the subsequent ring closure. Simultaneously, the cuprous acetate promoter facilitates the initial addition of the activated isonitrile to the electrophilic carbon of the trifluoroethylimidoyl chloride, driving the formation of the five-membered triazole ring intermediate with high regioselectivity.
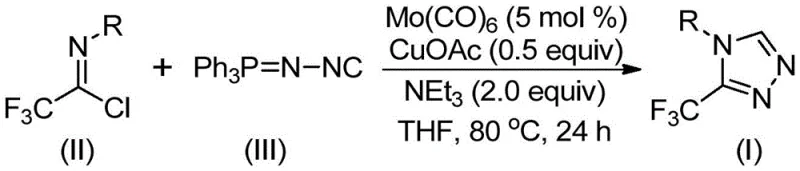
Following the cycloaddition event, the intermediate undergoes a spontaneous elimination of triphenylphosphine oxide, driven by the thermodynamic stability of the P=O bond and assisted by trace water within the reaction system, to yield the final 3-trifluoromethyl-substituted 1,2,4-triazole product. This mechanism is particularly advantageous for impurity control, as the mild reaction temperature of 80°C prevents the thermal decomposition of sensitive functional groups that might occur under more vigorous conditions. The use of triethylamine as a base further ensures the neutralization of acidic byproducts, maintaining a homogeneous reaction environment that minimizes the formation of polymeric side products, thereby simplifying the downstream purification process and ensuring high-purity output suitable for stringent pharmaceutical applications.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this high-efficiency synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and environmental controls to maximize yield and reproducibility. The patent specifies a preferred molar ratio where the functionalized isonitrile is used in slight excess relative to the trifluoroethylimidoyl chloride to drive the equilibrium forward, typically employing 5 mol % of the molybdenum catalyst and 0.5 equivalents of the copper promoter. The choice of solvent is critical, with tetrahydrofuran (THF) identified as the optimal medium due to its ability to effectively dissolve both the organic substrates and the metal catalysts while maintaining stability at the required reaction temperature of 80°C over a 24-hour period.
- Charge a reaction vessel with molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile (NIITP) in an organic solvent such as THF.
- Heat the reaction mixture to a temperature range of 70-90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target 3-trifluoromethyl-substituted 1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain stakeholders, the transition to this novel catalytic system offers profound logistical and economic benefits that extend far beyond simple yield improvements. The reliance on commodity chemicals such as cuprous acetate, triethylamine, and molybdenum hexacarbonyl means that raw material procurement is not subject to the volatility often seen with bespoke or single-source reagents. This stability in the supply base ensures consistent production schedules and reduces the risk of manufacturing delays caused by raw material shortages, a critical factor for maintaining the continuity of supply for life-saving medications.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like diazonium salts directly translates to lower raw material costs and reduced expenditure on safety infrastructure. Furthermore, the high atom economy of the cycloaddition reaction minimizes waste generation, which significantly lowers the costs associated with waste disposal and environmental compliance. The simplified workup procedure, involving basic filtration and standard column chromatography, reduces the consumption of solvents and silica gel, contributing to a leaner and more cost-effective manufacturing process overall.
- Enhanced Supply Chain Reliability: Since the starting materials, including the trifluoroethylimidoyl chloride and functionalized isonitrile, are either commercially available or easily synthesized from bulk commodities, the supply chain is inherently more robust against disruptions. The method's tolerance for various substituents on the aromatic ring allows for the flexible sourcing of different aniline precursors, providing procurement teams with multiple vendor options to mitigate geopolitical or logistical risks. This flexibility ensures that production can continue uninterrupted even if specific feedstock supplies face temporary constraints.
- Scalability and Environmental Compliance: The patent explicitly notes the successful expansion of this reaction to gram-level scales, indicating a clear pathway for kilogram and tonne-scale production without the need for complex reactor modifications. The absence of heavy metal contaminants that are difficult to remove, combined with the use of relatively benign solvents like THF which can be recycled, aligns perfectly with modern green chemistry principles. This facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site, enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route, derived directly from the experimental data and specifications provided in the patent documentation. Understanding these nuances is essential for process chemists aiming to adapt this technology for specific commercial applications.
Q: What are the key advantages of the Mo-Cu co-catalyzed method over traditional triazole synthesis?
A: Unlike traditional methods that often require harsh conditions or hazardous hydrazine derivatives, this patented process utilizes mild temperatures (70-90°C) and commercially available starting materials. The dual catalytic system significantly improves reaction efficiency and substrate tolerance, allowing for the synthesis of diverse 4-substituted derivatives with high purity.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the patent explicitly demonstrates that the method can be expanded to gram-level reactions with simple post-treatment procedures. The use of inexpensive catalysts like cuprous acetate and robust reaction conditions supports potential scale-up for commercial manufacturing of complex heterocyclic intermediates.
Q: What is the substrate scope for the R group in this triazole synthesis?
A: The method exhibits broad substrate tolerance. The R group can be phenethyl or various substituted aryl groups, including those with methyl, methoxy, fluoro, or chloro substituents at ortho, meta, or para positions. This flexibility allows for the tailored synthesis of specific analogues required for drug discovery pipelines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality heterocyclic intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest international standards.
We invite you to collaborate with us to leverage this cutting-edge Mo-Cu co-catalyzed technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain resilience and drive down your overall manufacturing costs.
