Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Commercial API Manufacturing
Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Commercial API Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which enhance metabolic stability and lipophilicity. Patent CN110467579B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds that addresses many of the limitations found in traditional synthetic routes. This technology leverages a non-metallic iodine-promoted cyclization strategy, utilizing inexpensive hydrazones and trifluoroethylimidoyl chlorides as key building blocks. The significance of this chemical scaffold cannot be overstated, as 1,2,4-triazole derivatives serve as the core structural motif in numerous high-value active pharmaceutical ingredients (APIs) and functional materials, ranging from antifungal agents like fluconazole to aromatase inhibitors such as letrozole, as illustrated in the diverse bioactive molecules shown below.
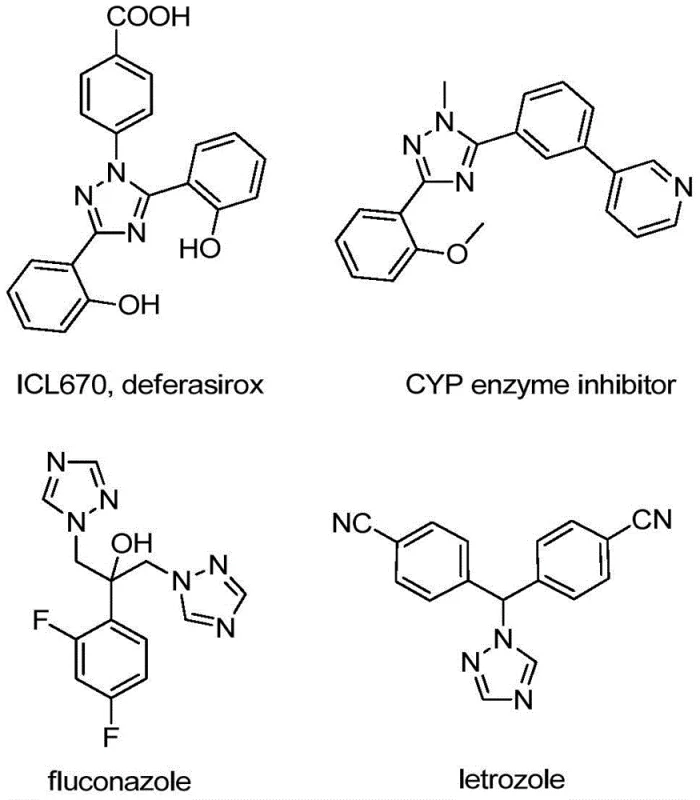
For R&D directors and process chemists, the ability to access these privileged structures without relying on complex multistep sequences or hazardous reagents represents a significant strategic advantage. The disclosed method operates under relatively mild thermal conditions and avoids the stringent requirement for anhydrous or oxygen-free environments, which are often costly to maintain in large-scale reactors. By shifting the paradigm from pre-functionalized heterocycle trifluoromethylation to a direct construction of the triazole ring from acyclic precursors, this innovation opens new avenues for the rapid diversification of chemical libraries. This report analyzes the technical merits of this approach and its profound implications for supply chain optimization and cost reduction in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a trifluoromethyl group into a 1,2,4-triazole ring has been a challenging transformation that often dictates the overall feasibility of a drug development program. Conventional literature reports typically describe two primary strategies, both of which suffer from distinct operational drawbacks that hinder industrial adoption. The first approach involves the direct trifluoromethylation of a pre-synthesized 1,2,4-triazole core; however, this necessitates the use of specialized and often expensive trifluoromethylating reagents that can be difficult to source in bulk quantities. Furthermore, these reagents frequently exhibit poor atom economy and generate significant amounts of fluorinated waste, complicating environmental compliance and waste disposal protocols. The second mainstream method relies on reacting trifluoromethyl-containing synthons, such as trifluorodiazoethane, with coupling substrates, but these reactions often require rigorous exclusion of moisture and oxygen, demanding specialized equipment and increasing capital expenditure.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN110467579B introduces a streamlined, one-pot synthesis that fundamentally simplifies the manufacturing workflow. By employing trifluoroethylimidoyl chloride and hydrazones as the foundational starting materials, the process bypasses the need for pre-formed heterocycles entirely. The reaction is promoted by elemental iodine, a commodity chemical that is vastly cheaper and more accessible than transition metal catalysts like palladium or copper, which are traditionally used in cross-coupling reactions. This novel approach not only eliminates the risk of heavy metal contamination—a critical quality attribute for pharmaceutical intermediates—but also tolerates a wide range of functional groups on the aromatic rings. The operational simplicity allows the reaction to proceed efficiently in common organic solvents like dichloroethane (DCE) at moderate temperatures, thereby reducing energy consumption and enhancing the safety profile of the manufacturing process.
Mechanistic Insights into Iodine-Promoted Cyclization
Understanding the mechanistic pathway of this transformation is crucial for process optimization and impurity control. The reaction is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and the hydrazone, leading to the generation of a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization to align the reactive centers for cyclization. The addition of elemental iodine then triggers a base-promoted oxidative iodination, creating an iodinated intermediate that is primed for the final ring-closing step. This sequence culminates in an intramolecular electrophilic substitution and subsequent aromatization to yield the stable 5-trifluoromethyl-1,2,4-triazole core. The general reaction scheme depicting this efficient transformation is presented below, highlighting the convergence of the two key precursors into the target heterocycle.
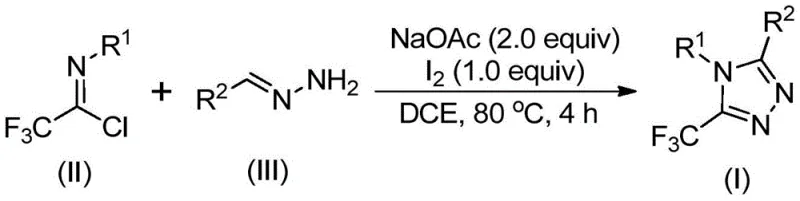
From a purity perspective, this mechanism offers distinct advantages regarding the impurity profile. Because the cyclization is driven by the intrinsic reactivity of the iodinated intermediate rather than a sensitive metal-ligand complex, the formation of side products associated with catalyst decomposition is virtually eliminated. The use of sodium acetate as a mild base further ensures that acid-sensitive functional groups on the substrate remain intact, preserving the integrity of complex molecular architectures. For quality control teams, this translates to a cleaner crude reaction mixture, which significantly reduces the burden on downstream purification units such as column chromatography or recrystallization. The robustness of this mechanistic pathway ensures consistent batch-to-batch reproducibility, a key metric for any reliable pharmaceutical intermediate supplier aiming to support long-term commercial campaigns.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale discovery and pilot-plant production. The protocol involves a straightforward addition of reagents followed by a controlled heating sequence, minimizing the need for complex dosing strategies or cryogenic conditions. Operators simply combine the trifluoroethylimidoyl chloride, hydrazone, and sodium acetate in the chosen solvent, heat the mixture to facilitate the initial condensation, and then introduce the iodine promoter to drive the reaction to completion. Detailed standardized operating procedures and specific stoichiometric ratios for optimal yield are critical for successful implementation. The comprehensive step-by-step synthesis guide is provided below for technical reference.
- Combine sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane (DCE) within a reaction vessel.
- Heat the reaction mixture to approximately 80°C and maintain this temperature for 2 to 4 hours to facilitate the initial condensation and cyclization steps.
- Add elemental iodine to the system and continue heating for an additional 1 to 2 hours to promote oxidative aromatization, followed by standard filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary driver for value creation lies in the drastic simplification of the raw material supply chain. By utilizing hydrazones and imidoyl chlorides, which are derived from widely available aldehydes and amines, manufacturers can mitigate the risks associated with sourcing exotic or single-source reagents. This diversification of the supply base enhances resilience against market volatility and ensures continuity of supply even during global disruptions. Furthermore, the elimination of heavy metal catalysts removes the necessity for expensive scavenging resins and extensive metal testing, directly impacting the cost of goods sold (COGS) and accelerating the release of finished batches.
- Cost Reduction in Manufacturing: The economic impact of switching to this iodine-promoted method is substantial, primarily due to the replacement of precious metal catalysts with commodity chemicals. Elemental iodine and sodium acetate are available in bulk at a fraction of the cost of palladium or rhodium complexes, leading to significant raw material savings. Additionally, the avoidance of strict anhydrous conditions reduces the energy load required for solvent drying and reactor purging, further lowering utility costs. The simplified workup procedure, which often requires only filtration and standard purification, minimizes labor hours and solvent consumption, contributing to a leaner and more cost-efficient manufacturing process overall.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as aromatic amines and aldehydes ensures a stable and predictable supply chain. Unlike specialized trifluoromethylating agents that may have long lead times or limited suppliers, the precursors for this method are produced by multiple chemical vendors globally. This redundancy allows procurement teams to negotiate better pricing and secure backup sources, effectively de-risking the production schedule. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, ensuring consistent output even when sourcing from different suppliers.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram and eventually to tonnage is facilitated by the absence of hazardous reagents and the use of standard organic solvents. The reaction does not generate toxic heavy metal waste streams, simplifying wastewater treatment and aligning with increasingly stringent environmental regulations. This green chemistry profile not only reduces disposal costs but also enhances the corporate sustainability metrics of the manufacturing site. The ability to run the reaction at moderate temperatures (80°C) without high-pressure equipment further lowers the barrier for scale-up, allowing for rapid capacity expansion to meet market demand.
Frequently Asked Questions (FAQ)
To assist technical stakeholders in evaluating the feasibility of this technology for their specific applications, we have compiled a set of frequently asked questions based on the patent data and practical experience. These inquiries address common concerns regarding catalyst residues, substrate compatibility, and process safety, providing clarity on how this method integrates into existing workflows. Understanding these nuances is essential for making informed decisions about process adoption and vendor selection. The following section provides detailed answers derived directly from the technical specifications of the patented method.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process utilizes elemental iodine as a promoter instead of toxic or expensive heavy metal catalysts, significantly simplifying downstream purification and reducing heavy metal residue risks in the final API.
Q: What is the substrate scope for this trifluoromethyl triazole synthesis?
A: The method demonstrates broad functional group tolerance, successfully accommodating various substituted aryl groups, heteroaryl groups, and alkenyl groups on both the imidoyl chloride and hydrazone starting materials.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (80°C), do not require strict anhydrous or anaerobic environments, and utilize commercially available raw materials, making it highly amenable to kilogram and ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly move from pilot trials to full-scale supply. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,4-triazole intermediate meets the highest industry standards for pharmaceutical and agrochemical applications. We are committed to delivering high-purity intermediates that enable our clients to accelerate their drug development timelines with confidence.
We invite you to engage with our technical team to explore how this innovative synthesis route can be tailored to your specific project needs. By leveraging our process optimization expertise, we can provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of adopting this metal-free methodology for your supply chain. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss custom route feasibility assessments for your proprietary molecules. Let us partner with you to drive efficiency and innovation in your chemical manufacturing operations.
