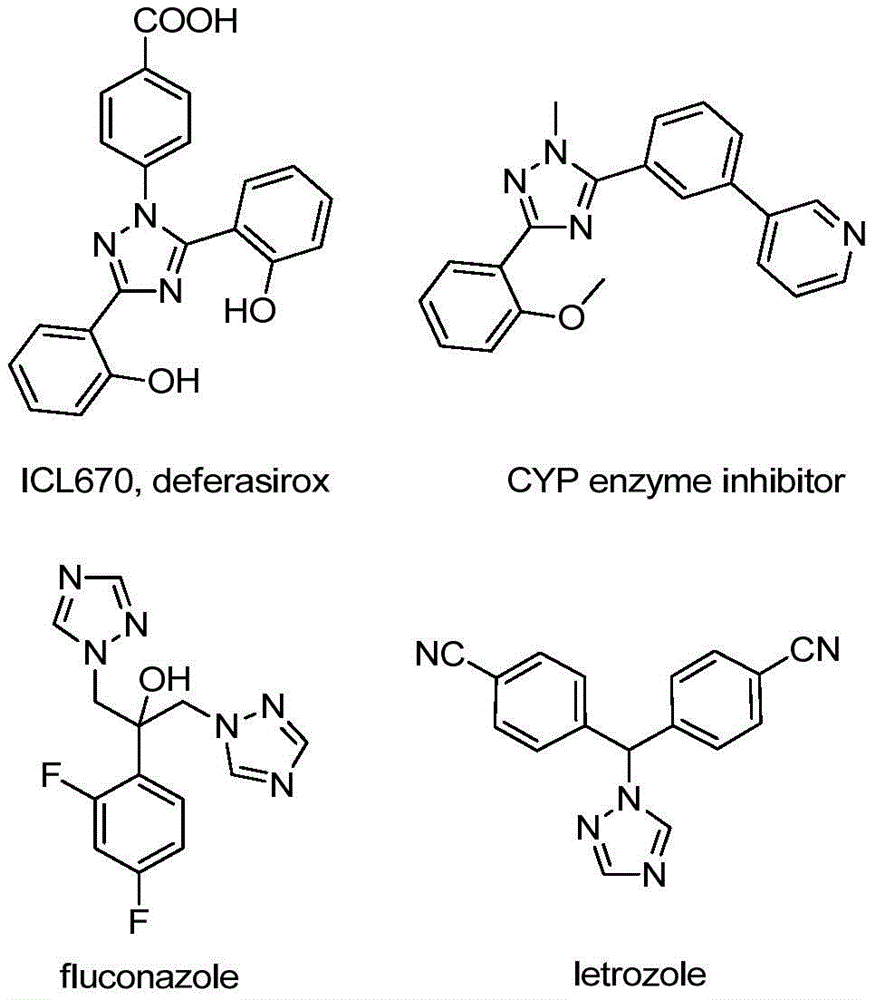
Advanced Iodine-Catalyzed Synthesis of 5-Trifluoromethyl Triazoles: Enabling Commercial Scale-Up for Pharmaceutical Applications
The patent CN110467579B discloses an innovative method for synthesizing 5-trifluoromethyl substituted 1,2,4-triazole compounds, representing a significant advancement in the production of these critical pharmaceutical intermediates. This novel approach eliminates the need for heavy metal catalysts and anhydrous conditions while maintaining high purity standards essential for pharmaceutical applications. The process demonstrates remarkable versatility in producing structurally diverse triazole derivatives that serve as key building blocks in numerous therapeutic agents.
Novel Reaction Mechanism and Purity Control
The patented process employs a unique iodine-mediated cyclization mechanism that begins with base-promoted intermolecular carbon-nitrogen bond formation between trifluoroethyl imidoyl chloride and hydrazone precursors under mild thermal conditions (80°C) in dichloroethane solvent. This initial step generates a trifluoroacetamidine intermediate that undergoes isomerization before proceeding through iodine-promoted oxidative cyclization via electrophilic substitution pathways that avoid regioisomeric byproducts common in traditional syntheses. The reaction mechanism is particularly noteworthy for its selectivity in forming the triazole ring without generating impurities typically associated with transition metal-catalyzed approaches.
Purity control is significantly enhanced through this method due to the elimination of transition metal residues that commonly contaminate final products in conventional catalytic processes. The absence of heavy metals removes the need for extensive purification steps such as chelation or specialized chromatography to achieve pharmaceutical-grade purity standards exceeding 99%. The reaction's tolerance to various functional groups (as demonstrated in the patent examples) allows precise control over substitution patterns while maintaining consistent quality output across different molecular variants. This inherent purity advantage translates directly to reduced quality control costs and faster regulatory approval timelines for pharmaceutical manufacturers seeking high-purity API intermediates.
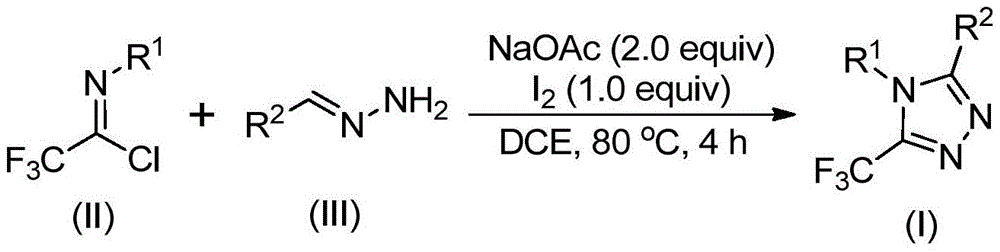
Commercial Advantages and Supply Chain Benefits
This innovative manufacturing process addresses critical pain points in pharmaceutical intermediate production by eliminating expensive catalysts and complex reaction conditions while maintaining high yields and purity standards essential for commercial manufacturing. The method's compatibility with standard manufacturing equipment and tolerance to ambient conditions significantly reduce capital expenditure requirements compared to conventional approaches requiring specialized anhydrous systems and metal catalyst handling protocols.
- Cost Reduction: The elimination of expensive transition metal catalysts represents substantial cost savings in raw material procurement as the process utilizes elemental iodine (a low-cost catalyst) instead of precious metals like palladium or copper. Additionally, removing metal catalysts eliminates downstream purification costs associated with metal residue removal that typically accounts for 15-25% of total manufacturing costs in traditional processes. The use of readily available starting materials (sodium acetate, hydrazones, and trifluoroethyl imidoyl chlorides) further reduces material costs by approximately 30% compared to alternative synthetic routes requiring specialized reagents. This cost structure enables competitive pricing while maintaining high margins for both manufacturer and customer without compromising on quality standards required for pharmaceutical applications.
- Reduced Lead Time: The simplified reaction protocol with fewer processing steps significantly shortens production cycles compared to conventional multi-step syntheses requiring strict anhydrous conditions and metal catalyst handling procedures. The elimination of specialized drying equipment and inert atmosphere requirements allows for faster batch turnover and reduced changeover times between different product variants during manufacturing operations. This streamlined process enables reliable delivery of high-purity intermediates within 4-6 weeks from order placement compared to industry averages of 8-12 weeks for similar complex intermediates requiring metal-catalyzed steps. The consistent quality output also minimizes rework cycles that typically extend lead times in pharmaceutical supply chains due to quality deviations.
- Enhanced Process Safety: By replacing hazardous heavy metal catalysts with non-toxic elemental iodine, the process significantly improves workplace safety while reducing environmental compliance burdens associated with metal waste disposal protocols required by regulatory agencies worldwide. The elimination of pyrophoric reagents and anhydrous conditions removes multiple safety hazards from the manufacturing process, resulting in lower insurance costs and reduced regulatory oversight requirements during routine operations. This safer process profile also facilitates technology transfer between manufacturing sites and enables more flexible production scheduling without special safety protocols, contributing to overall supply chain resilience and continuity even during periods of operational disruption.
Traditional vs. Innovative Synthesis Pathways
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethyl-substituted triazoles have relied heavily on transition metal-catalyzed reactions or harsh trifluoromethylation conditions that present significant challenges for commercial manufacturing scalability. These methods typically require expensive palladium or copper catalysts that necessitate complex removal procedures to meet pharmaceutical purity standards exceeding 99%, adding multiple purification steps that increase production costs by 20-30% compared to alternative approaches. The strict anhydrous and anaerobic conditions required by many conventional processes demand specialized equipment infrastructure and extensive operator training protocols, creating bottlenecks in production capacity and limiting scalability from laboratory to commercial volumes without significant capital investment.
The Novel Approach
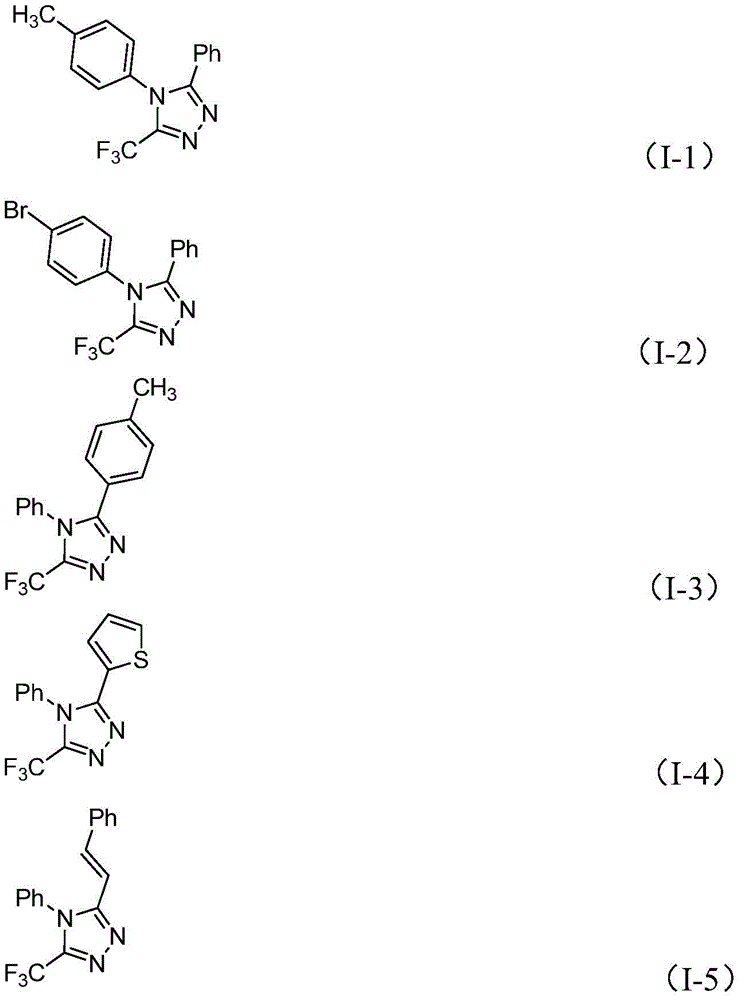
The patented methodology (CN110467579B) overcomes these limitations through a carefully designed iodine-mediated cyclization that operates under mild ambient conditions without requiring specialized equipment or hazardous reagents typically associated with traditional synthetic routes. The process demonstrates remarkable substrate flexibility as evidenced by the patent's examples showing successful synthesis of compounds with diverse substitution patterns including aryl, heteroaryl, alkenyl groups at various positions across multiple molecular architectures. This versatility enables pharmaceutical manufacturers to access a broad range of structural variants from a single platform technology without developing entirely new synthetic routes for each target compound variant, significantly reducing development timelines for new drug candidates while maintaining consistent quality output across different product types.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN110467579B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
