Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Pharmaceutical Applications
Introduction to Novel Biheterocyclic Synthesis Technology
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds, which serve as critical cores in numerous bioactive molecules. Patent CN115353511A introduces a groundbreaking multi-component strategy for the efficient preparation of carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs. This innovation addresses significant synthetic challenges by employing a transition metal palladium-catalyzed cascade reaction that operates under remarkably mild conditions. By integrating trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives into a single reaction vessel, the process achieves high atom economy and structural diversity. The technology represents a substantial leap forward for reliable pharmaceutical intermediate supplier networks, offering a pathway to access privileged scaffolds that are otherwise difficult to synthesize using conventional stepwise approaches.
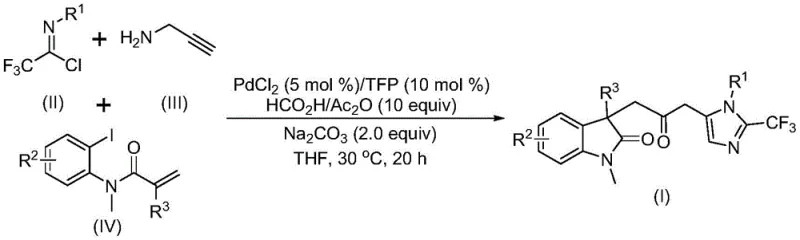
The significance of this invention lies in its ability to construct multiple chemical bonds simultaneously, including carbon-carbon and carbon-nitrogen bonds, alongside the crucial carbonyl bridge. Traditional methods often require harsh conditions or pre-functionalized intermediates that increase cost and waste. In contrast, this patented approach leverages the reactivity of readily available starting materials to drive the formation of the complex biheterocyclic core in a single operational step. For research and development teams focused on drug discovery, this means accelerated access to novel chemical space. The method's compatibility with various functional groups ensures that medicinal chemists can rapidly generate libraries of analogs for structure-activity relationship studies without being hindered by synthetic bottlenecks associated with older technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-bridged biheterocycles has been plagued by significant operational hazards and inefficiencies. Conventional carbonylation reactions typically rely on the direct use of carbon monoxide gas, which is highly toxic and requires specialized high-pressure equipment to handle safely. This necessity not only escalates the capital expenditure for manufacturing facilities but also introduces severe safety risks that complicate regulatory compliance and insurance protocols. Furthermore, traditional routes often involve multi-step sequences where the heterocyclic rings are constructed separately and then coupled, leading to lower overall yields and increased solvent consumption. These legacy processes frequently suffer from poor substrate compatibility, limiting the types of substituents that can be introduced onto the final molecular framework, thereby restricting the chemical diversity available for optimization in drug development programs.
The Novel Approach
The methodology described in the patent data revolutionizes this landscape by replacing hazardous gaseous carbon monoxide with a safe, liquid-phase surrogate system comprising formic acid and acetic anhydride. This in-situ generation of carbon monoxide allows the reaction to proceed at atmospheric pressure and a mild temperature of 30°C, drastically reducing energy consumption and safety overheads. The one-pot nature of the reaction eliminates the need for isolating unstable intermediates, thereby streamlining the workflow and minimizing material loss. For procurement managers, this translates to cost reduction in pharmaceutical intermediate manufacturing by simplifying the supply chain requirements for hazardous gases and reducing the complexity of the production infrastructure. The process is designed to be scalable, having been successfully demonstrated at the gram scale, which provides a clear trajectory for industrial adoption and commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The catalytic cycle underpinning this transformation is a sophisticated interplay of organometallic steps initiated by the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. This is followed by an intramolecular Heck reaction that constructs the indolinone ring system, generating a key alkyl-palladium intermediate. Subsequently, the carbon monoxide released from the formic acid and acetic anhydride mixture inserts into the palladium-carbon bond to form an acyl-palladium species. Concurrently, the trifluoroethylimidoyl chloride reacts with propargylamine in a base-promoted manner to form a trifluoroacetamidine intermediate, which undergoes isomerization. The final stage involves the activation of this amidine by the acyl-palladium complex, triggering an intramolecular cyclization that forge the imidazole ring and completes the carbonyl-bridged biheterocyclic architecture. Understanding this mechanism is vital for R&D directors aiming to optimize reaction parameters for specific substrates.
Impurity control in this system is inherently managed by the mild reaction conditions and the specificity of the palladium catalyst. The use of THF as the preferred solvent ensures that all reactants remain in solution, promoting homogeneous catalysis and preventing side reactions associated with phase transfer issues. The stoichiometry is carefully balanced, with propargylamine and acrylamide used in slight excess relative to the trifluoroethylimidoyl chloride to drive the reaction to completion without generating excessive byproducts. The base, sodium carbonate, plays a dual role in neutralizing acidic byproducts and facilitating the deprotonation steps necessary for cyclization. This precise control over the reaction environment results in high-purity products that require minimal downstream purification, aligning with the stringent quality standards expected of a high-purity pharmaceutical intermediate supplier.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Executing this synthesis requires careful attention to the order of addition and the quality of reagents to ensure reproducibility and high yield. The protocol begins with the preparation of the catalytic system in an inert atmosphere to prevent oxidation of the palladium species. The reaction mixture is then heated gently to the specified temperature, allowing the in-situ generation of carbon monoxide to synchronize with the catalytic cycle. Detailed standard operating procedures regarding workup and purification are essential to isolate the final product with the required purity profile for biological testing. The following guide outlines the critical steps derived from the patent examples to assist technical teams in implementing this chemistry effectively.
- Combine palladium chloride, trifurylphosphine ligand, sodium carbonate, and the formic acid/acetic anhydride mixture in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 12 to 20 hours, followed by filtration, silica gel treatment, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers compelling advantages that directly impact the bottom line and operational resilience of chemical manufacturing operations. The elimination of toxic carbon monoxide gas removes a major logistical hurdle, simplifying the storage and handling requirements within the facility. This shift significantly reduces the regulatory burden and safety training costs associated with hazardous gas management. Furthermore, the use of commercially available and inexpensive starting materials such as propargylamine and acrylamide derivatives ensures a stable and cost-effective supply chain. The mild reaction conditions also extend the lifespan of reactor equipment by reducing corrosion and thermal stress, leading to lower maintenance costs and higher asset utilization rates over time.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by removing the need for specialized high-pressure reactors and gas handling infrastructure. The use of a simple formic acid and acetic anhydride mixture as the carbon source is significantly cheaper and safer than purchasing and transporting compressed carbon monoxide. Additionally, the one-pot nature of the reaction reduces solvent usage and labor hours associated with intermediate isolation and purification steps. These factors combine to lower the overall cost of goods sold, making the final biheterocyclic compounds more competitive in the global market while maintaining healthy profit margins for manufacturers.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this synthesis is straightforward, as the key reagents are commodity chemicals available from multiple global suppliers. This diversification mitigates the risk of supply disruptions that can occur with specialized or proprietary reagents. The robustness of the reaction conditions means that production schedules are less likely to be impacted by equipment failures or environmental constraints. For supply chain heads, this reliability translates to consistent lead times and the ability to meet fluctuating demand from pharmaceutical clients without the need for excessive safety stock, thereby optimizing working capital.
- Scalability and Environmental Compliance: The methodology has been validated for scalability, moving seamlessly from milligram to gram scales with consistent performance. The absence of toxic gas emissions aligns with increasingly stringent environmental regulations, reducing the need for complex scrubbing systems. Waste generation is minimized through high atom economy and the use of recyclable solvents like THF. This environmental friendliness enhances the corporate sustainability profile of manufacturers adopting this technology, appealing to eco-conscious partners and investors who prioritize green chemistry principles in their supply chain decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing manufacturing workflows or research pipelines.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: This method utilizes a formic acid and acetic anhydride mixture to generate carbon monoxide in-situ, eliminating the need for handling toxic, high-pressure carbon monoxide gas cylinders typically required in conventional carbonylation reactions.
Q: What are the optimal reaction conditions for maximizing yield in this multi-component system?
A: The patent specifies using THF as the preferred solvent at a mild temperature of 30°C for a duration of 12 to 20 hours, utilizing a PdCl2/TFP catalyst system with sodium carbonate as the base.
Q: Does this process support diverse substrate scopes for drug discovery?
A: Yes, the methodology demonstrates excellent functional group tolerance, accommodating various substituents on the aryl rings including halogens, alkyl groups, alkoxy groups, and trifluoromethyl groups without significant loss in efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality, leveraging deep expertise in palladium-catalyzed transformations. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of carbonyl-bridged biheterocyclic compounds meets the highest international standards. Our commitment to quality and safety makes us the preferred partner for global pharmaceutical companies seeking reliable sources of complex intermediates.
We invite you to collaborate with us to explore the full potential of this innovative synthesis route for your specific drug development projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can accelerate your timeline to market while optimizing your production costs.
