Revolutionizing Pharmaceutical Intermediates: Scalable One-Pot Synthesis of High-Purity 2-Trifluoromethyl Quinazolinone
The patent CN112480015B introduces a groundbreaking multi-component one-pot synthesis method for producing 2-trifluoromethyl substituted quinazolinone compounds, representing a significant advancement in heterocyclic chemistry for pharmaceutical applications. This innovative approach addresses critical limitations in conventional quinazolinone synthesis by utilizing readily available starting materials under optimized catalytic conditions that enhance both efficiency and scalability. The methodology demonstrates exceptional substrate versatility while maintaining high reaction yields across diverse functional groups, making it particularly valuable for producing complex pharmaceutical intermediates. By eliminating the need for pre-activated substrates and harsh reaction environments, this patent establishes a new benchmark for sustainable manufacturing of fluorinated heterocyclic compounds essential in modern drug development pipelines. The process design inherently supports industrial implementation through its operational simplicity and compatibility with standard manufacturing equipment, positioning it as a transformative solution for pharmaceutical intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for quinazolinone compounds typically suffer from multiple critical limitations that hinder their commercial viability and scalability in pharmaceutical manufacturing environments. Conventional methods often require high-pressure carbon monoxide conditions with ruthenium or platinum catalysts, creating significant safety concerns and specialized equipment requirements that increase capital expenditure substantially. Many existing approaches depend on pre-activated substrates or expensive transition metal catalysts that necessitate complex removal processes, adding multiple purification steps that reduce overall yield and increase production costs. The narrow substrate scope of established methods restricts their applicability to specific molecular architectures, limiting their utility in producing diverse quinazolinone derivatives needed for drug discovery programs. Furthermore, these conventional processes frequently operate under harsh reaction conditions that generate unwanted byproducts, complicating purification and compromising final product purity to levels unacceptable for pharmaceutical applications where stringent quality standards must be met.
The Novel Approach
The patented methodology overcomes these limitations through an elegant multi-component one-pot cascade reaction that utilizes palladium catalysis under moderate temperature conditions without requiring high-pressure carbon monoxide systems. By employing trifluoroethylimidoyl chloride and nitro compounds as readily available starting materials, the process eliminates the need for pre-activated substrates while maintaining excellent functional group tolerance across diverse molecular architectures. The carefully optimized catalytic system featuring palladium chloride with dppp ligand operates efficiently at 120°C in standard organic solvents, avoiding specialized equipment requirements while delivering consistently high yields across a broad substrate scope. This innovative approach significantly simplifies the manufacturing workflow by consolidating multiple synthetic steps into a single reaction vessel, thereby reducing processing time, minimizing solvent usage, and eliminating intermediate purification requirements that typically plague conventional routes. The resulting process demonstrates exceptional scalability potential while maintaining the high purity standards essential for pharmaceutical intermediate production.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The reaction mechanism begins with hexacarbonyl molybdenum-mediated reduction of nitro compounds to amines under thermal conditions, followed by base-promoted intermolecular carbon-nitrogen bond formation between the amine and trifluoroethylimidoyl chloride to generate trifluoroacetamidine intermediates. This critical transformation occurs under mild conditions without requiring expensive catalysts or specialized equipment, establishing a foundation for the subsequent cascade reactions. The palladium catalyst then inserts into the carbon-iodine bond of the intermediate to form a key divalent palladium species that facilitates the carbonylation process through controlled carbon monoxide release from Mo(CO)6 under heating conditions. This step represents a significant innovation as it avoids high-pressure CO systems while maintaining efficient carbonyl group incorporation essential for quinazolinone ring formation.
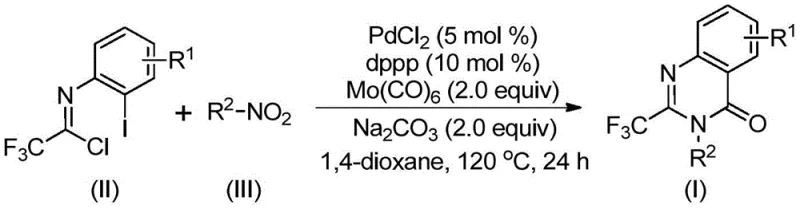
The carbonyl insertion step forms an acylpalladium intermediate that undergoes base-promoted cyclization to create a seven-membered palladacycle, which subsequently undergoes reductive elimination to yield the final quinazolinone product with excellent regioselectivity. This mechanistic pathway demonstrates remarkable efficiency through its self-contained cascade design that minimizes intermediate isolation while maximizing atom economy. The strategic use of carbonate base not only facilitates key bond-forming steps but also helps maintain optimal reaction pH throughout the process, preventing unwanted side reactions that could compromise product purity.


Impurity control is achieved through precise stoichiometric balance between reactants and catalysts, with the molar ratio of palladium chloride to dppp ligand (0.05:0.1) ensuring optimal catalytic activity without promoting side reactions. The use of sodium carbonate as base provides controlled deprotonation without causing decomposition of sensitive intermediates, while the choice of dioxane as solvent offers ideal solubility characteristics for all reaction components without participating in unwanted side reactions. This carefully engineered system maintains consistent performance across diverse substrate combinations, as evidenced by the broad range of successfully synthesized derivatives with varying R¹ and R² substituents while maintaining high product purity essential for pharmaceutical applications.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
This patented methodology represents a significant advancement in quinazolinone synthesis by integrating multiple reaction steps into a single operational sequence that dramatically simplifies manufacturing workflows while maintaining exceptional product quality standards required by pharmaceutical clients. The process leverages commercially available starting materials and standard laboratory equipment to achieve high-yielding transformations under precisely controlled conditions that ensure reproducibility at scale. By eliminating intermediate isolation steps and complex purification requirements, this approach reduces both processing time and resource consumption while delivering products meeting stringent pharmaceutical purity specifications. The following standardized procedure details the critical parameters necessary for successful implementation of this innovative synthesis route.
- Prepare the reaction mixture by combining trifluoroethylimidoyl chloride, nitro compounds, palladium catalyst, ligand, and carbonate base in dioxane solvent under inert atmosphere.
- Heat the reaction mixture to precisely controlled 120°C and maintain for optimized duration between 16 to 30 hours to complete the carbonylation cascade reaction.
- Perform post-reaction processing including filtration, silica gel mixing, and column chromatography purification to obtain high-purity 2-trifluoromethyl quinazolinone compounds.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial commercial benefits by addressing critical pain points in pharmaceutical intermediate supply chains through its elegant process design that prioritizes operational efficiency without compromising product quality or regulatory compliance requirements. The strategic selection of readily available starting materials combined with simplified processing steps creates significant opportunities for cost optimization while enhancing supply chain resilience through reduced dependency on specialized components or complex manufacturing infrastructure.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and high-pressure carbon monoxide systems significantly reduces capital equipment requirements while avoiding costly metal removal processes that typically add multiple purification steps to conventional routes. The use of commercially available starting materials at optimal stoichiometric ratios minimizes raw material waste and improves overall process economics through enhanced atom efficiency without requiring specialized handling procedures or expensive safety infrastructure.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials with multiple global suppliers mitigates single-source dependency risks while enabling flexible sourcing strategies that maintain consistent production schedules even during market fluctuations. The simplified process design with fewer critical control points reduces vulnerability to supply chain disruptions by minimizing specialized component requirements and enabling rapid scale-up from laboratory to commercial production without significant re-engineering efforts.
- Scalability and Environmental Compliance: The one-pot methodology demonstrates exceptional scalability potential from laboratory to commercial production volumes while maintaining consistent product quality through its inherently robust reaction design that requires minimal process adjustments during scale-up. The elimination of hazardous reagents and reduction in solvent usage compared to conventional multi-step routes significantly lowers environmental impact while simplifying waste management procedures, aligning with increasingly stringent regulatory requirements for sustainable manufacturing practices in the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement teams regarding implementation of this patented synthesis methodology in commercial manufacturing environments. These responses are based on detailed analysis of the patent's technical disclosures and experimental validation data provided in the original filing.
Q: How does this one-pot method improve upon conventional synthesis routes for quinazolinones?
A: The novel multi-component one-pot approach eliminates the need for pre-activated substrates and harsh reaction conditions required by conventional methods, while maintaining high substrate compatibility and yield across diverse functional groups without requiring expensive metal removal steps.
Q: What are the key advantages of using palladium catalysis in this synthesis?
A: Palladium catalysis enables efficient carbonylation cascade reactions under moderate conditions (120°C), avoiding high-pressure CO requirements while providing excellent functional group tolerance and eliminating the need for costly transition metal purification processes.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The reaction design minimizes side products through precise catalyst control and optimized stoichiometry, while the straightforward purification process delivers compounds meeting stringent pharmaceutical purity specifications without complex additional processing steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring seamless transition from laboratory development to full-scale manufacturing while maintaining stringent purity specifications required by global regulatory authorities. With rigorous QC labs equipped to handle complex analytical requirements for fluorinated heterocyclic compounds, we provide comprehensive quality assurance throughout the production process to deliver products meeting exacting pharmaceutical standards consistently across all batch sizes.
Leverage our technical expertise through a Customized Cost-Saving Analysis tailored to your specific manufacturing needs and receive detailed COA data along with route feasibility assessments from our technical procurement team to optimize your supply chain strategy for high-value quinazolinone intermediates.
