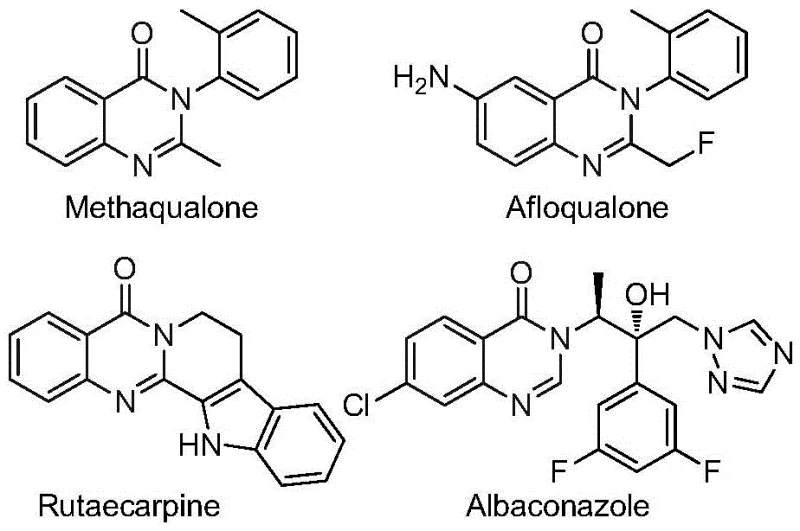
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Manufacturing
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries continuously seek robust synthetic routes for heterocyclic scaffolds that offer both biological potency and metabolic stability. Patent CN112480015B introduces a transformative multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones, a privileged structure found in numerous bioactive molecules. This technology leverages a palladium-catalyzed carbonylation cascade, utilizing inexpensive nitro compounds and trifluoroethylimidoyl chloride as key starting materials. By replacing hazardous high-pressure carbon monoxide gas with solid molybdenum hexacarbonyl, this process significantly enhances operational safety while maintaining high reaction efficiency. For R&D directors and procurement managers, this represents a critical advancement in accessing complex fluorinated intermediates with improved cost structures and supply chain reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing the quinazolinone core often suffer from significant logistical and economic drawbacks that hinder large-scale adoption. Conventional methods frequently rely on ruthenium or platinum catalysts operating under high-pressure carbon monoxide conditions, which necessitate specialized autoclave equipment and stringent safety protocols. Furthermore, many existing routes require pre-activated substrates such as 2-bromoformylaniline or acid anhydrides, which are not only expensive but also generate substantial stoichiometric waste during the activation steps. These factors collectively result in higher production costs, longer lead times for raw material sourcing, and increased environmental burdens due to complex waste streams. Additionally, the substrate scope in older methodologies is often narrow, limiting the ability to introduce diverse functional groups required for modern drug discovery programs.
The Novel Approach
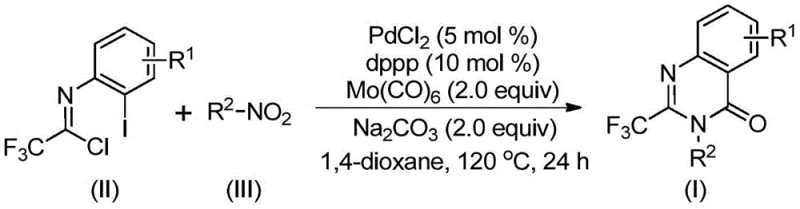
The methodology disclosed in patent CN112480015B overcomes these historical barriers through an elegant multi-component coupling strategy that streamlines the synthesis into a single operational step. By employing nitro compounds as the nitrogen source, the process utilizes some of the most abundant and cost-effective building blocks available in the fine chemical market. The reaction proceeds via a palladium-catalyzed cascade where molybdenum hexacarbonyl serves as a safe, solid carbon monoxide surrogate, eliminating the need for dangerous gas handling infrastructure. This one-pot protocol demonstrates exceptional functional group tolerance, allowing for the incorporation of halogens, alkyl groups, and trifluoromethyl moieties without compromising yield. The result is a highly versatile platform capable of generating diverse libraries of 2-trifluoromethyl quinazolinones with yields reaching up to 96%, providing a superior alternative for both medicinal chemistry and process development.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific analogues. The reaction initiates with the reduction of the nitro compound to the corresponding amine by molybdenum hexacarbonyl, which simultaneously acts as the reducing agent and the carbon monoxide source. Following this reduction, a base-promoted intermolecular carbon-nitrogen bond coupling occurs between the generated amine and trifluoroethylimidoyl chloride, forming a trifluoroacetamidine derivative in situ. The palladium catalyst then inserts into the carbon-iodine bond of the imidoyl chloride moiety, generating a reactive divalent palladium intermediate. As the temperature is maintained at 120°C, the molybdenum complex releases carbon monoxide, which subsequently inserts into the carbon-palladium bond to form an acyl-palladium species.
The final stages of the catalytic cycle involve the intramolecular nucleophilic attack of the nitrogen atom on the acyl-palladium center, facilitated by the base, leading to the formation of a seven-membered cyclic palladium intermediate. Reductive elimination from this species releases the final 2-trifluoromethyl substituted quinazolinone product and regenerates the active palladium catalyst. This intricate dance of organometallic steps ensures high atom economy and minimizes the formation of side products. The compatibility of this mechanism with various substituents on the aromatic ring, such as electron-withdrawing halogens or electron-donating alkyl groups, underscores the robustness of the catalytic system. For quality control purposes, monitoring the consumption of the nitro starting material and the evolution of the amidine intermediate can provide valuable insights into reaction progress and potential bottlenecks.

How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The protocol involves charging a reaction vessel with palladium chloride, the dppp ligand, sodium carbonate, and molybdenum hexacarbonyl in an anhydrous organic solvent such as 1,4-dioxane. The specific molar ratios are critical, typically employing a slight excess of the nitro compound relative to the imidoyl chloride to drive the reaction to completion. Once the reagents are mixed, the system is heated to 120°C for a duration ranging from 16 to 30 hours. Post-reaction workup is straightforward, involving filtration to remove metal salts followed by silica gel treatment and column chromatography.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in 1,4-dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours under inert atmosphere to facilitate the carbonylation cascade.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that directly address the pain points of procurement managers and supply chain directors. The shift from specialized, high-cost substrates to commodity nitro compounds fundamentally alters the cost basis of the final intermediate. By removing the dependency on high-pressure gas infrastructure, the process becomes accessible to a wider range of manufacturing facilities, thereby increasing supply redundancy and reducing geopolitical supply risks. The simplified one-pot nature of the reaction reduces the number of unit operations, which translates to lower energy consumption and reduced labor costs per kilogram of product. Furthermore, the high yields reported across a broad substrate scope minimize the loss of valuable fluorinated starting materials, ensuring that raw material investments are maximized.
- Cost Reduction in Manufacturing: The elimination of expensive pre-activated substrates and the use of commodity nitro compounds significantly lowers the direct material costs associated with production. Additionally, the replacement of high-pressure carbon monoxide gas with solid molybdenum hexacarbonyl removes the need for costly safety certifications and specialized reactor vessels, leading to substantial capital expenditure savings. The streamlined one-pot process reduces solvent usage and waste disposal fees, further enhancing the overall economic viability of the manufacturing route.
- Enhanced Supply Chain Reliability: Nitro compounds and trifluoroethylimidoyl chlorides are widely available from multiple global suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling manufacturers to respond quickly to fluctuating demand without lengthy requalification processes. This flexibility ensures a steady flow of high-purity intermediates to downstream API synthesis lines, preventing production delays caused by material shortages.
- Scalability and Environmental Compliance: The process has been demonstrated to scale effectively from milligram to gram levels with consistent performance, indicating strong potential for multi-kilogram and ton-scale production. The use of standard organic solvents and the absence of toxic gas emissions simplify environmental compliance and waste treatment protocols. This alignment with green chemistry principles not only reduces regulatory burdens but also enhances the sustainability profile of the final pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic method. These answers are derived directly from the experimental data and mechanistic proposals outlined in the patent documentation. Understanding these nuances is essential for process chemists planning to adopt this technology for their own pipelines.
Q: What are the advantages of using nitro compounds over traditional substrates in this synthesis?
A: Nitro compounds are significantly cheaper and more readily available than pre-activated substrates like 2-bromoformylaniline. This method eliminates the need for harsh high-pressure carbon monoxide conditions, enhancing operational safety and reducing raw material costs.
Q: How does the Mo(CO)6 substitute function in this reaction mechanism?
A: Molybdenum hexacarbonyl acts as a solid carbon monoxide surrogate. Under heating at 120°C, it releases CO in situ, which inserts into the palladium-carbon bond, facilitating the carbonylation step without requiring dangerous high-pressure gas cylinders.
Q: Is this synthetic route scalable for industrial production?
A: Yes, the patent explicitly demonstrates that the method can be expanded to the gram level with high efficiency. The use of standard organic solvents like dioxane and commercially available catalysts supports straightforward scale-up for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient synthetic routes like the one described in patent CN112480015B for accelerating drug development timelines. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with rigorous safety standards, and our stringent purity specifications guarantee that every batch meets the highest quality benchmarks. With our rigorous QC labs and deep expertise in fluorinated chemistry, we are uniquely positioned to support your long-term supply needs for complex pharmaceutical intermediates.
We invite you to leverage our technical capabilities to optimize your supply chain and reduce time-to-market for your critical projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing excellence can drive value for your organization.
