Scalable One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Advanced Pharmaceutical Applications
Scalable One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Advanced Pharmaceutical Applications
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activity. Patent CN112480015B introduces a transformative multi-component one-pot methodology for the construction of 2-trifluoromethyl substituted quinazolinones, a privileged structure found in numerous bioactive molecules ranging from antifungal agents to anticancer drugs. This innovation addresses critical bottlenecks in traditional synthesis by leveraging a palladium-catalyzed carbonylation cascade that utilizes readily available nitro compounds and trifluoroethylimidoyl chlorides. The strategic incorporation of the trifluoromethyl group is paramount, as it significantly enhances the metabolic stability, lipophilicity, and bioavailability of the parent molecular framework, making these intermediates highly desirable for the development of next-generation active pharmaceutical ingredients (APIs).
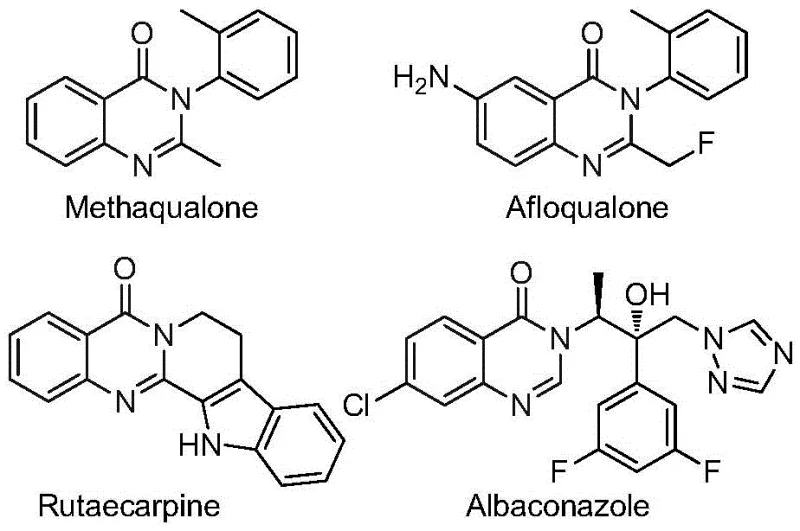
Quinazolinone compounds represent an important class of condensed nitrogen-containing six-membered heterocycles that are widely prevalent in various functional molecules and pharmaceutical agents. As illustrated in the structural diversity of known drugs such as Methaqualone and Afloqualone, this scaffold possesses a broad spectrum of biological properties, including antiviral, anti-inflammatory, and anticonvulsant activities. The introduction of fluorine atoms, specifically in the form of a trifluoromethyl group, serves to markedly improve the physicochemical profile of these molecules. Consequently, establishing a reliable and scalable supply chain for high-purity 2-trifluoromethyl quinazolinone intermediates is of strategic importance for pharmaceutical manufacturers aiming to optimize their drug discovery pipelines and ensure consistent quality in commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone derivatives has been plagued by significant operational challenges and safety concerns that hinder large-scale implementation. Traditional methodologies often rely on the use of high-pressure carbon monoxide gas, which necessitates specialized autoclave equipment and rigorous safety protocols to mitigate the risks associated with toxic gas handling. Furthermore, many existing routes require expensive or pre-activated substrates, such as 2-bromoformylaniline or acid anhydrides, which drastically increase the raw material costs and limit the economic feasibility of the process. Additionally, conventional iron or ruthenium-catalyzed systems frequently suffer from narrow substrate scope and moderate reaction efficiencies, leading to lower overall yields and complex purification workflows that generate substantial chemical waste. These factors collectively create a barrier to entry for cost-effective manufacturing of complex heterocyclic intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN112480015B offers a streamlined, one-pot solution that circumvents the need for hazardous high-pressure conditions by employing molybdenum hexacarbonyl as a safe and controllable carbon monoxide surrogate. This novel approach utilizes cheap and commercially available nitro compounds as starting materials, which undergo an in situ reduction and subsequent cyclization cascade catalyzed by a palladium system. The reaction proceeds efficiently at atmospheric pressure in common organic solvents like 1,4-dioxane, demonstrating exceptional compatibility with a wide array of functional groups including halogens and alkyl chains. By consolidating multiple synthetic steps into a single operational unit, this method not only simplifies the workflow but also significantly enhances the atom economy and overall process safety, making it an ideal candidate for industrial scale-up.
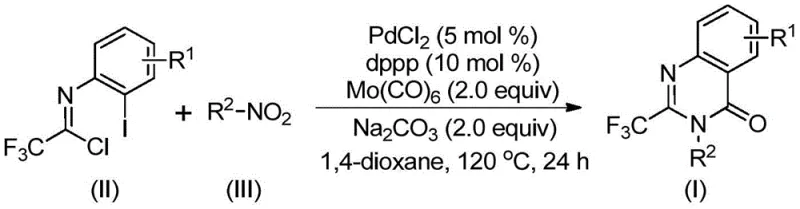
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation involves a sophisticated sequence of organometallic steps initiated by the reduction of the nitro compound to the corresponding amine by molybdenum hexacarbonyl under thermal conditions. Following this reduction, a base-promoted intermolecular coupling occurs between the generated amine and the trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate. The palladium catalyst then inserts into the carbon-iodine bond of the aromatic ring, forming a divalent palladium species that is crucial for the subsequent cyclization. As the reaction temperature is maintained at 120°C, the molybdenum complex releases carbon monoxide, which inserts into the carbon-palladium bond to generate an acyl-palladium intermediate. This key step facilitates the formation of a seven-membered cyclic palladium species through nitrogen coordination, which finally undergoes reductive elimination to release the desired 2-trifluoromethyl substituted quinazolinone product and regenerate the active catalyst.
From an impurity control perspective, the use of a well-defined palladium catalyst system combined with a solid CO source allows for precise regulation of the reaction kinetics, minimizing the formation of side products often associated with uncontrolled gas-phase carbonylations. The choice of 1,3-bis(diphenylphosphino)propane (dppp) as the ligand plays a critical role in stabilizing the palladium center and promoting the specific reductive elimination pathway required for ring closure. Furthermore, the basic conditions provided by sodium carbonate ensure the efficient neutralization of hydrochloric acid byproducts generated during the amidine formation, preventing catalyst deactivation. This robust mechanistic design ensures high selectivity and purity, which are essential parameters for meeting the stringent quality standards required for pharmaceutical intermediate manufacturing.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed for simplicity and reproducibility, requiring standard laboratory equipment without the need for specialized high-pressure reactors. The process begins by charging a reaction vessel with the palladium catalyst, ligand, base, and carbon monoxide source, followed by the addition of the organic solvent and substrates. The mixture is then heated to the optimal temperature range to drive the cascade reaction to completion. Detailed standardized operating procedures regarding specific stoichiometric ratios, solvent volumes, and workup protocols are essential for achieving the reported high yields and purity levels. For comprehensive step-by-step instructions tailored to specific substrate variations, please refer to the technical guide below.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in 1,4-dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours under inert atmosphere.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The primary advantage lies in the utilization of nitro compounds and trifluoroethylimidoyl chlorides as starting materials, both of which are commodity chemicals available in bulk quantities from multiple global suppliers. This abundance ensures a stable supply chain with minimal risk of raw material shortages or price volatility, which is a common issue with specialized pre-activated substrates used in older methodologies. Moreover, the elimination of high-pressure carbon monoxide gas removes the requirement for expensive containment infrastructure and specialized safety training, resulting in substantial capital expenditure savings and reduced operational overheads for manufacturing facilities.
- Cost Reduction in Manufacturing: The transition to a one-pot protocol significantly reduces processing time and labor costs by eliminating intermediate isolation and purification steps. By avoiding the use of high-pressure equipment, facilities can utilize standard glass-lined or stainless steel reactors, thereby lowering depreciation costs and maintenance requirements. The high reaction efficiency and broad substrate tolerance mean that fewer batches are rejected due to poor conversion, optimizing resource utilization and maximizing output per unit of time. Additionally, the use of a solid CO surrogate simplifies waste management compared to handling toxic gas streams, further contributing to overall cost efficiency in the production of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved as the key reagents are not proprietary or restricted substances, allowing for multi-vendor qualification and competitive pricing negotiations. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the need for extensive rework or reprocessing that can delay delivery schedules. This reliability is crucial for maintaining continuous production lines for downstream API synthesis, ensuring that downstream customers receive their orders on time without interruption. The scalability of the method from gram to kilogram scales without significant loss in efficiency further guarantees that supply can be rapidly ramped up to meet market demand fluctuations.
- Scalability and Environmental Compliance: The process aligns well with green chemistry principles by improving atom economy and reducing the generation of hazardous waste associated with traditional activation steps. The ability to run the reaction in common solvents like dioxane simplifies solvent recovery and recycling processes, minimizing the environmental footprint of the manufacturing operation. Scaling this process to multi-kilogram or tonne levels is straightforward since it does not rely on complex engineering controls for high-pressure gases, making it easier to obtain regulatory approvals for new production lines. This ease of scale-up ensures that the technology remains viable and compliant with increasingly stringent environmental regulations governing chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity on process capabilities and limitations. Understanding these details is vital for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the safety advantages of using Mo(CO)6 over high-pressure CO gas?
A: Using solid Mo(CO)6 as a carbon monoxide surrogate eliminates the need for specialized high-pressure autoclaves and hazardous gas handling infrastructure, significantly reducing operational risks and capital expenditure.
Q: Is this synthesis method compatible with diverse functional groups?
A: Yes, the protocol demonstrates excellent substrate tolerance, successfully accommodating halogens, alkyl groups, and trifluoromethyl substituents on both the imidoyl chloride and nitro compound starting materials.
Q: What is the typical yield range for this transformation?
A: Experimental data indicates robust performance with isolated yields ranging from 69% to 96% across various substituted derivatives, ensuring efficient material throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global clients. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and potency. Our capability to adapt complex catalytic processes like the one described in CN112480015B allows us to offer customized solutions that balance cost, speed, and quality effectively.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall procurement expenses. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a seamless transition from laboratory research to commercial supply.
