Revolutionizing Thioester Production: A Safe and Scalable Nickel-Catalyzed Carbonylation Strategy
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of the pharmaceutical and agrochemical industries, particularly regarding safety and efficiency. A significant breakthrough in this domain is documented in patent CN112239384B, which discloses a robust preparation method for thioester compounds. Thioesters are pivotal structural motifs found in numerous bioactive molecules and natural products, serving as essential acyl donors in complex organic transformations. The traditional reliance on hazardous reagents has long been a bottleneck for industrial scale-up, but this new methodology offers a transformative alternative by utilizing cheap and readily available arylboronic acids and sulfonyl chlorides. By leveraging a transition metal-catalyzed carbonylation strategy, this process eliminates the need for external oxidants or reductants, streamlining the synthetic pathway significantly.
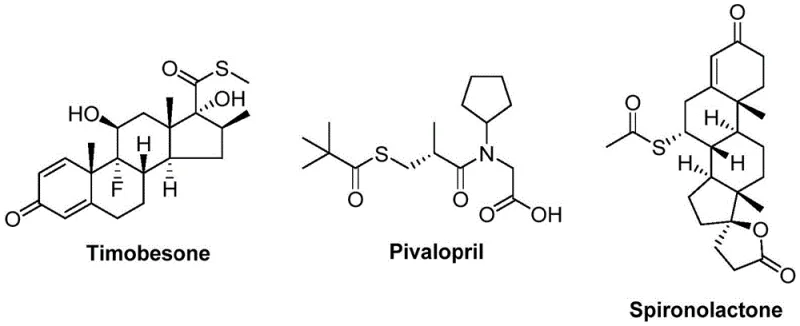
This innovation is particularly relevant for manufacturers seeking a reliable pharmaceutical intermediate supplier who can deliver high-purity compounds without the logistical nightmares associated with volatile sulfur sources. The ability to synthesize various thioester compounds according to actual needs widens the practicability of the method, making it an attractive option for cost reduction in fine chemical manufacturing. As we delve deeper into the technical specifics, it becomes clear that this protocol addresses critical pain points in supply chain continuity and operational safety, positioning it as a superior choice for commercial scale-up of complex polymer additives and drug precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioester compounds has predominantly relied on the direct acylation reaction of mercaptans with carboxylic acids or their derivatives. While chemically straightforward in a laboratory setting, this approach presents severe drawbacks when translated to an industrial environment. The primary limitation lies in the nature of mercaptans themselves; they are notorious for their extremely unpleasant, pervasive odors which pose significant health and safety challenges for plant personnel. Furthermore, sulfur-containing compounds like thiols have a strong tendency to coordinate with transition metals, leading to catalyst poisoning. This deactivation necessitates higher catalyst loadings or more frequent catalyst replacement, driving up operational costs and complicating waste management. Additionally, many conventional methods require harsh reaction conditions or the use of unstable intermediates that compromise the overall yield and purity of the final product.
The Novel Approach
In stark contrast, the method described in the patent utilizes sulfonyl chlorides as the sulfur source, coupled with arylboronic acids in a nickel-catalyzed system. This strategic shift fundamentally alters the safety and efficiency profile of the reaction. Sulfonyl chlorides are stable, odorless solids or liquids that are easy to handle and store, effectively removing the olfactory and toxicity hazards associated with mercaptans. The reaction proceeds through a carbonylation pathway where molybdenum carbonyl serves as a convenient solid source of carbon monoxide, bypassing the need for dangerous high-pressure CO gas cylinders. This not only enhances safety but also simplifies the reactor setup, allowing for more flexible batch processing. The use of a nickel catalyst system, specifically nickel triflate with a bipyridine ligand, ensures high reaction efficiency and broad substrate compatibility, enabling the synthesis of diverse thioester structures with excellent yields.
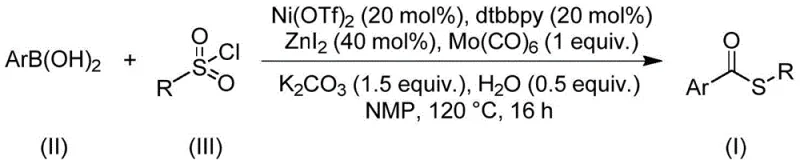
Mechanistic Insights into Nickel-Catalyzed Carbonylation
The core of this synthetic advancement lies in the intricate interplay between the nickel catalyst, the ligand, and the carbonyl source. The reaction initiates with the oxidative addition of the sulfonyl chloride to the low-valent nickel species, generating a nickel-sulfur intermediate. Simultaneously, the arylboronic acid undergoes transmetallation, transferring the aryl group to the metal center. The presence of molybdenum hexacarbonyl is crucial, as it thermally decomposes under the reaction conditions to release carbon monoxide in situ. This CO then inserts into the nickel-carbon or nickel-sulfur bond, forming the requisite acyl-nickel species. The subsequent reductive elimination step releases the desired thioester product and regenerates the active nickel catalyst, closing the catalytic cycle. This mechanism is highly efficient because it avoids the formation of stable off-cycle species that often plague sulfur chemistry.
From an impurity control perspective, this mechanism offers distinct advantages. The use of zinc iodide and water as additives plays a vital role in facilitating the transmetallation step and stabilizing the catalytic species, thereby minimizing side reactions such as homocoupling of the boronic acid or desulfonylation. The mild basicity provided by potassium carbonate ensures that the reaction medium remains conducive to the catalytic cycle without promoting hydrolysis of the sensitive thioester product. Furthermore, the high functional group tolerance observed in this system means that sensitive moieties like halogens, ethers, and alkyl chains remain intact throughout the process. This selectivity is paramount for pharmaceutical applications where the presence of trace impurities can disqualify a batch, ensuring that the final product meets stringent purity specifications required for downstream drug synthesis.
How to Synthesize Thioester Compound Efficiently
Implementing this synthesis route requires precise adherence to the optimized conditions outlined in the patent to achieve maximum yield and purity. The process involves charging a reaction vessel with specific molar ratios of nickel triflate, dtbbpy ligand, molybdenum carbonyl, zinc iodide, potassium carbonate, and water in an aprotic solvent like N-methylpyrrolidone (NMP). The choice of NMP is critical as it effectively dissolves all inorganic and organic components, creating a homogeneous reaction environment that promotes efficient mass transfer. Once the reagents are mixed, the system is heated to 120°C and maintained for 16 hours. This extended reaction time ensures complete consumption of the starting materials, particularly the sulfonyl chloride, which can sometimes be sluggish. Following the reaction, the workup is remarkably simple, involving filtration to remove inorganic salts followed by standard silica gel chromatography.
- Combine nickel triflate, dtbbpy ligand, molybdenum carbonyl, zinc iodide, potassium carbonate, water, arylboronic acid, and sulfonyl chloride in NMP solvent.
- Heat the reaction mixture to 120°C and maintain stirring for 16 hours to ensure complete conversion.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target thioester compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible operational improvements and risk mitigation. The shift away from malodorous and toxic mercaptans significantly reduces the regulatory burden and safety infrastructure costs associated with handling hazardous sulfur compounds. This change alone can lead to substantial cost savings by eliminating the need for specialized scrubbing systems and reducing insurance premiums related to chemical exposure risks. Moreover, the raw materials employed—arylboronic acids and sulfonyl chlorides—are commodity chemicals available from multiple global suppliers, ensuring a robust and competitive supply chain. This diversity in sourcing prevents bottlenecks and allows for better negotiation leverage on pricing, directly impacting the bottom line.
- Cost Reduction in Manufacturing: The elimination of expensive and specialized sulfur sources like thiols, combined with the use of earth-abundant nickel instead of precious metals like palladium, drastically lowers the raw material cost per kilogram. The process does not require additional oxidants or reductants, further simplifying the bill of materials and reducing waste disposal costs. The high conversion rates achieved under these conditions mean less raw material is wasted, and the simplified post-treatment reduces labor and solvent consumption during purification. These factors collectively contribute to a more economically viable manufacturing process that enhances profit margins without compromising quality.
- Enhanced Supply Chain Reliability: By utilizing stable and commercially available starting materials, the risk of supply disruption due to the volatility or instability of reagents is minimized. Sulfonyl chlorides have a long shelf life and do not require the stringent storage conditions necessary for thiols or pressurized gases. This stability allows manufacturers to maintain optimal inventory levels without fear of degradation, ensuring consistent production schedules. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, providing a buffer against supply chain fluctuations and ensuring reliable delivery to customers.
- Scalability and Environmental Compliance: The use of a solid CO source (Mo(CO)6) removes the significant safety barriers associated with scaling up gas-phase carbonylations, making the transition from pilot plant to commercial production seamless. The reaction generates minimal hazardous waste compared to traditional methods, aligning with increasingly strict environmental regulations and sustainability goals. The simplicity of the workup procedure reduces the volume of organic solvents required for purification, lowering the facility's environmental footprint. This green chemistry profile not only facilitates regulatory approval but also enhances the brand reputation of the manufacturer as a responsible and sustainable partner in the global chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented thioester synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances is essential for optimizing the process parameters and maximizing the yield for specific target molecules.
Q: Why is sulfonyl chloride preferred over mercaptans for thioester synthesis?
A: Sulfonyl chlorides are odorless, stable, and do not poison the catalyst, unlike mercaptans which are malodorous and can deactivate transition metal catalysts.
Q: What is the role of Molybdenum Carbonyl in this reaction?
A: Molybdenum carbonyl acts as a solid carbon monoxide source, eliminating the need for handling hazardous high-pressure CO gas during the carbonylation process.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates wide functional group tolerance, accommodating alkyl, alkoxy, and halogen substituents on both the arylboronic acid and sulfonyl chloride components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to stay competitive in the global market. Our team of expert chemists has thoroughly analyzed the potential of this nickel-catalyzed carbonylation route and is fully equipped to implement it for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is smooth and efficient. Our state-of-the-art facilities are designed to handle complex organometallic reactions safely, and our rigorous QC labs guarantee that every batch meets stringent purity specifications required by the pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced technology for your next product launch. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss your project specifics,索取 specific COA data, and obtain detailed route feasibility assessments. Let us help you optimize your supply chain and reduce manufacturing costs while maintaining the highest standards of quality and safety.
