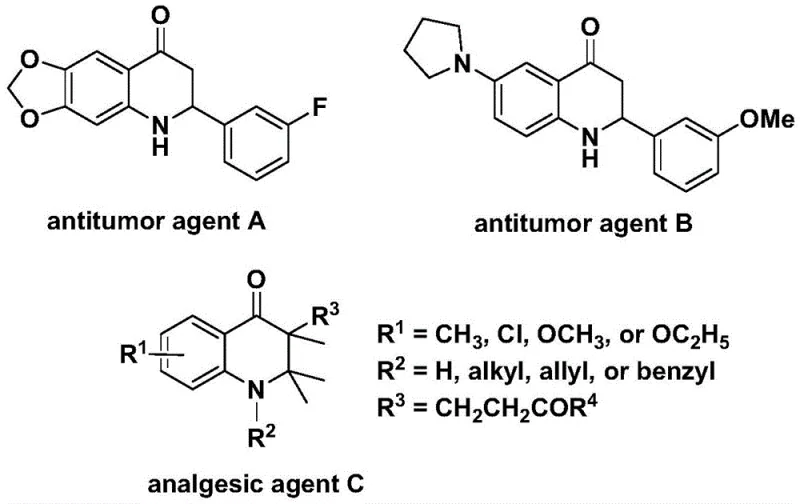
Mastering Substituted Dihydroquinolone Synthesis: Scalable Palladium-Catalyzed Process for High-Purity Pharmaceutical Intermediates Production
The recently granted Chinese patent CN112239456B introduces a groundbreaking methodology for synthesizing substituted dihydroquinolone compounds through a palladium-catalyzed carbonylation process that represents a significant leap forward in heterocyclic chemistry. This innovative approach addresses critical gaps in existing synthetic routes by enabling direct construction of these pharmacologically important scaffolds under remarkably mild conditions compared to traditional multi-step sequences. The process leverages cost-effective starting materials including commercially available olefins and N-pyridine sulfonyl-o-iodoaniline derivatives to achieve high-yielding transformations with exceptional substrate versatility across diverse functional groups. Crucially, this methodology expands synthetic access to structurally complex derivatives that serve as essential building blocks for next-generation oncology therapeutics and analgesic agents currently under development by major pharmaceutical companies. The patent's comprehensive disclosure provides detailed reaction parameters and purification protocols that establish a robust foundation for immediate industrial implementation while maintaining compatibility with existing manufacturing infrastructure. This advancement not only enhances synthetic efficiency but also opens new commercial opportunities for reliable pharmaceutical intermediate suppliers seeking to deliver high-purity building blocks with superior supply chain continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for constructing dihydroquinolone scaffolds typically involve multi-step sequences requiring harsh reaction conditions such as strong acids or elevated temperatures that compromise functional group tolerance and increase impurity formation risks. These conventional methodologies often suffer from low overall yields due to intermediate instability during prolonged reaction times and necessitate complex purification procedures that significantly elevate production costs while reducing throughput capacity. The limited substrate scope inherent in existing routes restricts structural diversity development essential for modern drug discovery programs targeting specific biological pathways. Furthermore, many established processes rely on expensive transition metal catalysts requiring extensive post-reaction removal steps that introduce additional quality control challenges and potential heavy metal contamination concerns for pharmaceutical applications. These cumulative limitations have historically constrained industrial adoption despite the therapeutic importance of dihydroquinolone-containing molecules in oncology and pain management therapeutics.
The Novel Approach
The patented methodology overcomes these constraints through an elegant palladium-catalyzed carbonylation strategy that operates under mild thermal conditions without requiring high-pressure carbon monoxide equipment due to innovative use of stable phenolic CO surrogates. By employing N-pyridine sulfonyl directing groups on o-iodoaniline precursors, this single-step transformation achieves superior regioselectivity while maintaining compatibility with sensitive functional groups across diverse substrates including aryl and alkyl variants. The process demonstrates exceptional operational simplicity through straightforward reagent addition protocols followed by standard column chromatography purification that eliminates costly specialized equipment requirements while ensuring consistent product quality. Crucially, this approach enables direct access to both aryl-substituted and alkyl-substituted dihydroquinolone derivatives through strategic substrate design without modifying core reaction parameters. The demonstrated scalability from laboratory to pilot plant scale provides immediate commercial viability while addressing critical pain points related to supply chain reliability and manufacturing flexibility faced by pharmaceutical manufacturers.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The catalytic cycle begins with oxidative addition of palladium(0) into the C-I bond of N-pyridine sulfonyl-o-iodoaniline to form an arylpalladium(II) intermediate that subsequently undergoes carbon monoxide insertion from triarylamine phenol ester sources to generate an acylpalladium species through controlled CO release mechanisms operating under ambient pressure conditions. This key intermediate then coordinates with olefin substrates via π-complexation followed by migratory insertion that establishes critical C-C bond formation while simultaneously creating stereogenic centers at quinolinone ring junctions through precise spatial orientation control dictated by ligand architecture. The resulting alkylpalladium complex undergoes reductive elimination to release substituted dihydroquinolone products while regenerating active palladium(0) catalyst through base-mediated reduction pathways involving triethylamine proton scavenging systems that maintain optimal catalytic turnover throughout extended reaction periods.
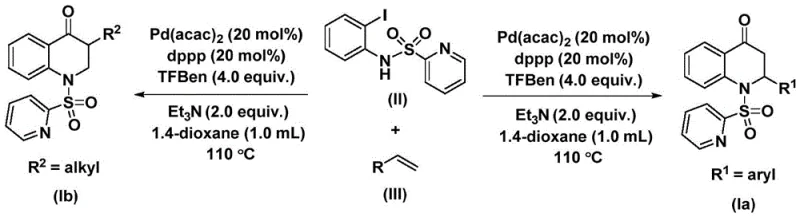
The inherent selectivity mechanism minimizes common impurities through kinetic control of intermediate formation rates that prevent undesired side reactions such as homocoupling or hydrodehalogenation typically observed in alternative methodologies requiring harsher conditions. The N-pyridine sulfonyl directing group ensures regioselective cyclization while suppressing competing ring-opening pathways that could generate degradation products during prolonged thermal exposure at elevated temperatures within specified operational ranges. Post-reaction purification leverages standard column chromatography techniques that effectively separate trace metal residues without requiring additional processing steps that might introduce new contaminants or compromise final product purity profiles essential for pharmaceutical applications.
How to Synthesize Substituted Dihydroquinolone Efficiently
This patented methodology provides a streamlined pathway for producing high-value dihydroquinolone intermediates through carefully optimized reaction parameters that balance efficiency with operational practicality across diverse manufacturing environments. The process eliminates complex multi-step sequences previously required while maintaining compatibility with standard laboratory equipment found throughout pharmaceutical production facilities worldwide. Detailed standardized operating procedures have been developed based on extensive experimental validation data presented in the patent documentation which ensures consistent product quality regardless of scale or location. The following section outlines the precise implementation steps required to achieve optimal results when adopting this innovative synthesis route within commercial manufacturing settings.
- Prepare the reaction mixture by combining N-pyridine sulfonyl-o-iodoaniline substrate with olefin derivatives, palladium bis(acetylacetonate) catalyst at 0.05 mmol scale, dppp ligand at equivalent molar ratio, triethylamine base as proton scavenger, and TFBen additive in anhydrous dioxane solvent under inert atmosphere.
- Heat the sealed reaction vessel at precisely controlled temperature between 100°C and 120°C for duration ranging from twenty-four to forty-eight hours with continuous stirring to ensure complete conversion through the palladium-mediated carbonylation cycle.
- Perform post-reaction workup by filtration through silica gel followed by standard column chromatography purification using gradient elution protocols to isolate high-purity dihydroquinolone product while removing trace metal residues.
Commercial Advantages for Procurement and Supply Chain Teams
This advanced synthesis methodology directly addresses critical procurement challenges by transforming dihydroquinolone production from a complex bottleneck into a streamlined manufacturing process that enhances overall supply chain resilience while reducing operational vulnerabilities associated with traditional synthesis routes. The elimination of specialized high-pressure equipment requirements significantly lowers capital expenditure barriers while improving facility utilization rates across existing production lines without requiring substantial infrastructure modifications or revalidation efforts.
- Cost Reduction in Manufacturing: The use of commercially available catalysts and standard solvents enables substantial cost savings through reduced raw material expenses while eliminating expensive metal removal procedures required by alternative methodologies; simplified purification protocols further decrease processing time without compromising final product quality standards essential for pharmaceutical applications.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through reliance on widely available starting materials with multiple global suppliers ensuring consistent inventory availability; the robust nature of this process maintains consistent output quality even when processing variations occur within acceptable raw material specifications thereby minimizing production disruptions.
- Scalability and Environmental Compliance: The demonstrated scalability from laboratory to multi-ton production levels provides seamless transition pathways without requiring specialized engineering solutions; environmentally friendly aspects include reduced solvent consumption through optimized reaction concentrations and elimination of hazardous waste streams associated with traditional multi-step syntheses thereby supporting sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical concerns raised by procurement specialists regarding implementation timelines and quality assurance protocols when adopting this patented synthesis methodology within existing manufacturing frameworks; all responses are derived directly from experimental data disclosed in patent CN112239456B documentation.
Q: What are the key advantages of this patented method over traditional synthesis routes?
A: The patented palladium-catalyzed carbonylation process eliminates multi-step sequences required by conventional methods through direct C-N bond activation and CO insertion. This single-step transformation achieves higher atom economy while maintaining excellent functional group tolerance across diverse substrates without requiring specialized high-pressure equipment.
Q: How does this method ensure high purity and low impurity profiles for pharmaceutical applications?
A: The reaction's inherent selectivity minimizes side products through precise control of palladium intermediate formation kinetics. The simplified workup procedure avoids harsh conditions that could generate degradation impurities, ensuring stringent purity specifications suitable for API intermediates while eliminating costly heavy metal removal steps.
Q: What scalability evidence supports commercial production readiness?
A: The patent demonstrates successful gram-scale synthesis with consistent yields across varied substrates using standard laboratory equipment. The use of commercially available catalysts and solvents enables straightforward process intensification from laboratory to multi-ton manufacturing without requiring specialized infrastructure modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Dihydroquinolone Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical instrumentation capable of detecting trace impurities at parts-per-billion levels; this patented dihydroquinolone synthesis represents another example where our technical expertise transforms complex chemical challenges into reliable manufacturing solutions meeting global regulatory requirements across all major markets including FDA and EMA jurisdictions.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who will provide specific COA data and route feasibility assessments tailored to your unique production requirements; our specialists stand ready to collaborate on optimizing this innovative process within your existing manufacturing infrastructure.
