Revolutionizing Pharmaceutical Intermediate Production Scalable Synthesis of Substituted Dihydroquinolones via Palladium-Catalyzed Carbonylation
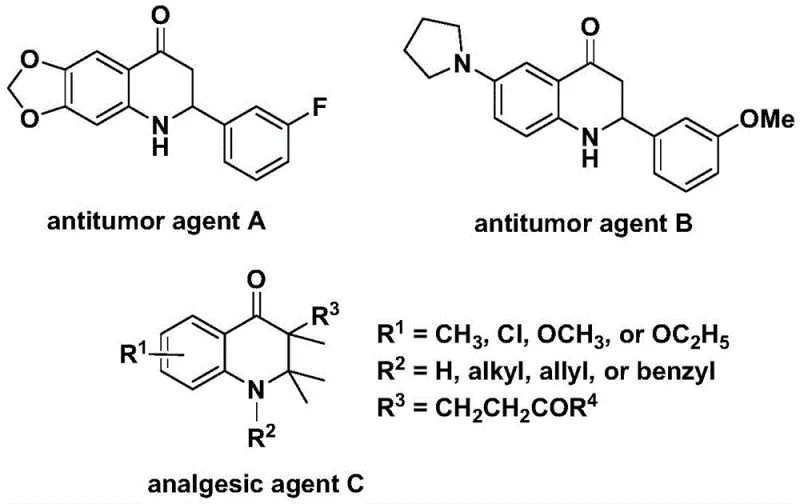
The groundbreaking patent CN112239456B introduces a novel palladium-catalyzed carbonylation methodology for synthesizing substituted 2,3-dihydroquinolone compounds—a critical structural motif prevalent in numerous bioactive pharmaceuticals. This innovative approach addresses longstanding synthetic challenges by enabling direct construction of the dihydroquinolone core through a streamlined catalytic cycle that operates under mild conditions compared to conventional multi-step sequences. The methodology demonstrates exceptional versatility in producing both aryl-substituted and alkyl-substituted variants through strategic substrate design, thereby expanding access to diverse molecular architectures essential for drug discovery pipelines. Crucially, the process leverages commercially available starting materials including N-pyridine sulfonyl-o-iodoaniline and various olefins, establishing a foundation for cost-effective manufacturing of high-value pharmaceutical intermediates. This patent represents a significant advancement in heterocyclic chemistry with direct implications for accelerating development of oncology therapeutics and analgesic agents where these scaffolds exhibit proven biological activity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for dihydroquinolone compounds typically involve multi-step sequences requiring harsh reaction conditions such as strong acids or high-pressure carbon monoxide systems that pose significant safety hazards and operational complexities. These methods often suffer from poor functional group tolerance leading to extensive protection-deprotection strategies that dramatically increase production costs and reduce overall efficiency. The substrate scope is frequently limited to specific substitution patterns, preventing flexible access to diverse molecular variants needed for structure-activity relationship studies in drug development. Additionally, conventional approaches generate substantial waste streams due to stoichiometric reagents and require complex purification procedures that compromise yield and purity—particularly problematic when targeting pharmaceutical-grade intermediates requiring stringent quality specifications. The cumulative effect is extended development timelines and elevated manufacturing costs that hinder commercial viability for many promising therapeutic candidates.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylation process that constructs the dihydroquinolone core in a single operation under controlled thermal conditions between 100°C and 120°C. By utilizing N-pyridine sulfonyl-o-iodoaniline as a versatile starting material combined with readily available olefins, the process achieves remarkable substrate flexibility while maintaining high reaction efficiency across diverse functional groups. The catalytic system employing Pd(acac)₂ with dppp ligand enables precise control over regioselectivity without requiring specialized equipment or hazardous reagents. This streamlined approach eliminates multiple intermediate steps while delivering excellent yields of both aryl-substituted and alkyl-substituted products through simple variations in olefin components. The methodology's compatibility with standard laboratory equipment facilitates immediate adoption by pharmaceutical manufacturers seeking more efficient routes to these critical building blocks.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The catalytic cycle initiates with oxidative addition of palladium into the carbon-nitrogen bond of N-pyridine sulfonyl-o-iodoaniline forming an aryl palladium intermediate that subsequently undergoes carbon monoxide insertion from the TFBen source to generate an acyl palladium species. This key intermediate then coordinates with the olefin substrate followed by migratory insertion that establishes the critical carbon-carbon bond necessary for ring closure. The resulting alkyl palladium complex undergoes reductive elimination to yield the substituted dihydroquinolone product while regenerating the active palladium catalyst for subsequent cycles. This mechanism operates through well-defined organometallic steps that avoid common side reactions such as homocoupling or β-hydride elimination due to the carefully optimized ligand system and reaction conditions. The precise control over each transformation step ensures high regioselectivity and minimizes formation of undesired byproducts that would complicate purification processes.
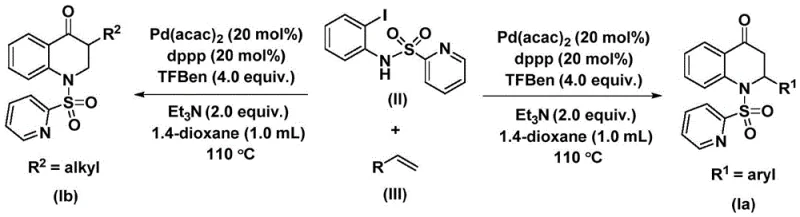
Impurity control is inherently achieved through the selective nature of the catalytic pathway which favors formation of the desired dihydroquinolone structure while suppressing alternative reaction channels. The use of triethylamine as a base effectively neutralizes acidic byproducts that could otherwise promote decomposition pathways or epimerization at chiral centers. The solvent system comprising dioxane provides optimal polarity for solubilizing both organic substrates and inorganic additives while maintaining catalyst stability throughout the extended reaction period. Post-reaction processing through standard column chromatography efficiently removes trace metal residues and unreacted starting materials without requiring specialized purification techniques—ensuring final products consistently meet pharmaceutical industry purity standards without additional processing steps that would increase manufacturing complexity.
How to Synthesize Substituted Dihydroquinolones Efficiently
This section details the standardized synthesis protocol derived from patent CN112239456B that enables reliable production of high-purity substituted dihydroquinolone intermediates at commercial scale. The methodology represents a significant advancement over conventional approaches by eliminating hazardous reagents while maintaining excellent functional group tolerance across diverse substrate combinations. By leveraging commercially available starting materials and standard laboratory equipment, this process delivers consistent results with minimal operator training requirements—making it ideal for implementation in both research laboratories and manufacturing facilities. The following step-by-step guide provides comprehensive instructions for executing this synthesis with optimal efficiency and yield.
- Combine palladium bis(acetylacetonate) catalyst, dppp ligand, triethylamine base, TFBen additive, N-pyridine sulfonyl-o-iodoaniline substrate, and olefin in dioxane solvent under inert atmosphere with precise stoichiometric ratios.
- Heat the reaction mixture to controlled temperatures between 100°C and 120°C while maintaining vigorous stirring for durations of 24 to 48 hours to ensure complete conversion of starting materials.
- Execute post-reaction processing through filtration to remove catalyst residues followed by silica gel mixing and column chromatography purification to isolate high-purity substituted dihydroquinolone products.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points associated with traditional manufacturing approaches for complex heterocyclic intermediates. The process eliminates dependency on specialized equipment or hazardous reagents while utilizing readily available starting materials that enhance supply chain resilience through multiple sourcing options. By streamlining production into a single catalytic operation with simplified post-processing requirements, manufacturers can achieve significant reductions in production cycle times without compromising product quality—directly supporting just-in-time inventory strategies essential for modern pharmaceutical supply chains.
- Cost Reduction in Manufacturing: The elimination of multi-step sequences reduces both capital expenditure requirements and operational costs through decreased solvent consumption and energy usage per production batch. The use of commercially available catalysts and ligands avoids expensive proprietary reagents while maintaining high reaction efficiency across diverse substrates—resulting in substantial cost savings without requiring significant process revalidation when scaling production volumes.
- Enhanced Supply Chain Reliability: Sourcing flexibility is significantly improved through reliance on widely available starting materials including standard olefins and commercially synthesized N-pyridine sulfonyl-o-iodoaniline derivatives that can be obtained from multiple global suppliers. This diversification minimizes single-source dependencies while enabling rapid response to demand fluctuations through straightforward scale-up procedures that maintain consistent product quality across different production volumes.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production without requiring fundamental modifications to reaction parameters or equipment specifications—enabling seamless transition from development to manufacturing phases. The simplified waste stream profile resulting from reduced solvent usage and elimination of hazardous reagents significantly lowers environmental impact while facilitating compliance with increasingly stringent regulatory requirements for sustainable chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by industry professionals regarding implementation of this patented synthesis methodology for pharmaceutical intermediate production. These responses are derived directly from experimental data presented in patent CN112239456B and reflect practical insights gained during process development and scale-up activities.
Q: Why is this palladium-catalyzed method superior to conventional approaches for synthesizing dihydroquinolone intermediates?
A: The patented process eliminates multi-step sequences required in traditional syntheses by directly constructing the dihydroquinolone core through carbonylative cyclization. This approach avoids harsh reaction conditions like strong acids or high-pressure CO systems while maintaining excellent functional group tolerance across diverse substrates.
Q: How does the substrate design flexibility enable tailored production of diverse pharmaceutical intermediates?
A: The modular design allows strategic substitution at both aryl and alkyl positions through simple variations in olefin components. This enables precise customization of molecular properties such as solubility and binding affinity while maintaining consistent reaction efficiency across different structural variants.
Q: What are the key factors ensuring high purity and scalability in commercial manufacturing?
A: The process achieves inherent impurity control through selective catalytic pathways that minimize side reactions. Combined with straightforward post-treatment procedures and commercially available starting materials, this enables seamless transition from laboratory scale to industrial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Dihydroquinolone Supplier
We specialize in delivering high-purity substituted dihydroquinolone intermediates through our proprietary implementation of this patented methodology, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities. Our CDMO expertise ensures seamless technology transfer from laboratory development to full-scale manufacturing with comprehensive process validation that meets global regulatory requirements for pharmaceutical intermediates—providing clients with reliable access to these critical building blocks without operational disruption or quality compromise.
Contact our technical procurement team today to request specific COA data and route feasibility assessments for your development programs. We offer a Customized Cost-Saving Analysis demonstrating how our patented synthesis approach can optimize your supply chain while ensuring consistent quality delivery timelines for your most demanding pharmaceutical intermediate requirements.
