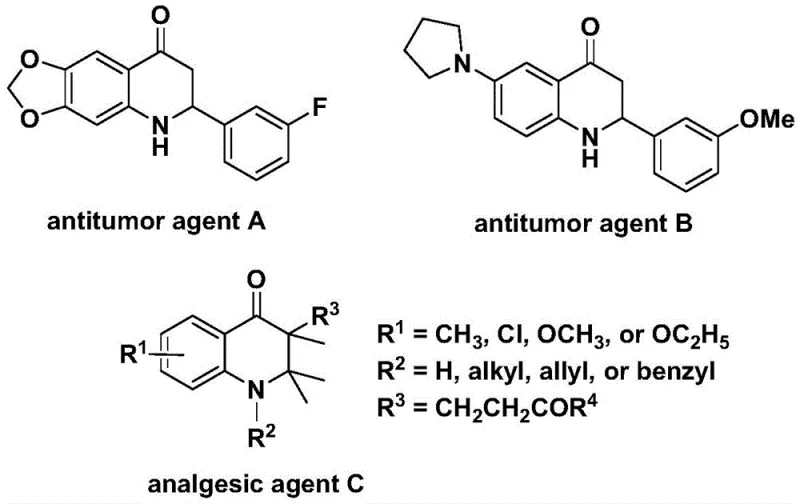
Advanced Pd-Catalyzed Route to High-Purity Dihydroquinolone Intermediates for Commercial Scale-Up
Patent CN112239456B introduces a groundbreaking methodology for synthesizing substituted 2,3-dihydroquinolone compounds, which serve as critical scaffolds in numerous bioactive molecules including potent antitumor agents and analgesics. This innovative approach leverages palladium-catalyzed carbonylation chemistry to overcome longstanding limitations in traditional synthetic routes, offering unprecedented efficiency and scalability for pharmaceutical manufacturing. The process utilizes readily available starting materials such as N-pyridine sulfonyl-o-iodoaniline and olefins under mild reaction conditions (100-120°C), achieving high yields without requiring complex purification procedures. By eliminating the need for expensive transition metal catalysts and harsh reagents, this method significantly enhances the commercial viability of producing these high-value intermediates. The patent demonstrates robust substrate compatibility across diverse functional groups, enabling tailored synthesis of both aryl and alkyl substituted derivatives essential for drug discovery pipelines. This advancement represents a strategic leap forward in meeting the growing demand for complex heterocyclic building blocks within the global pharmaceutical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for dihydroquinolone compounds often require multi-step sequences involving harsh reaction conditions such as strong acids or high temperatures exceeding 150°C, which frequently lead to decomposition of sensitive functional groups present in pharmaceutical intermediates. These methods typically suffer from poor substrate scope limitations where specific substitution patterns necessitate entirely different synthetic routes, significantly increasing development timelines and costs. The reliance on stoichiometric oxidants or expensive transition metal catalysts creates additional purification challenges that compromise final product purity while generating substantial waste streams requiring specialized disposal procedures. Furthermore, conventional carbonylation techniques often exhibit narrow functional group tolerance that restricts their applicability to complex molecular architectures needed in modern drug discovery programs. The cumulative effect of these limitations results in low overall yields below practical manufacturing thresholds and inconsistent quality profiles that fail to meet stringent regulatory requirements for active pharmaceutical ingredients.
The Novel Approach
The patented methodology overcomes these challenges through a single-step palladium-catalyzed carbonylation process operating under mild conditions (100-120°C) that maintains exceptional functional group compatibility across diverse substrates. By employing bis(acetylacetonato)palladium with dppp ligand in dioxane solvent, the reaction achieves high efficiency without requiring specialized equipment or hazardous reagents typically associated with traditional methods. This approach eliminates multiple protection/deprotection steps while enabling direct access to both aryl and alkyl substituted derivatives through simple substrate design modifications. The process demonstrates remarkable tolerance for halogenated, alkoxy-substituted, and sterically hindered substrates that would decompose under conventional conditions. Crucially, the simplified workup procedure involving basic filtration followed by standard column chromatography significantly reduces production complexity while maintaining high purity levels essential for pharmaceutical applications. This innovation transforms dihydroquinolone synthesis from a bottleneck process into a scalable manufacturing solution capable of meeting industrial production demands.
Mechanistic Insights into Pd-Catalyzed Carbonylation
The catalytic cycle begins with oxidative addition of palladium(0) into the carbon-nitrogen bond of N-pyridine sulfonyl-o-iodoaniline (II), forming an aryl palladium intermediate that subsequently undergoes carbon monoxide insertion from the triethylamine-bound source to generate an acyl palladium species. This key intermediate then coordinates with the olefin substrate (III), facilitating migratory insertion to produce a palladium alkyl complex that undergoes reductive elimination to yield the final substituted dihydroquinolone product while regenerating the active catalyst. The strategic use of bis(acetylacetonato)palladium with dppp ligand creates an optimal electronic environment that accelerates both CO insertion and alkene coordination steps while suppressing undesired side reactions such as β-hydride elimination. This mechanistic pathway enables precise control over regioselectivity during the cyclization process, ensuring consistent formation of the desired six-membered heterocyclic ring structure with minimal byproduct generation. The reaction's tolerance for various functional groups stems from the mild conditions that prevent decomposition of sensitive substituents commonly encountered in pharmaceutical intermediates.
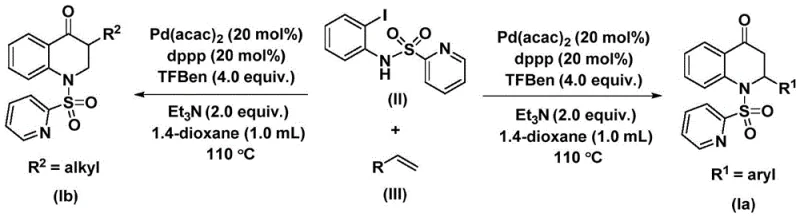
Impurity control is achieved through careful optimization of reaction parameters including temperature (maintained at 110°C), stoichiometry (4.0 equivalents of triethylamine), and solvent choice (1,4-dioxane). The absence of strong acids or bases eliminates common degradation pathways that typically generate impurities in conventional syntheses. Post-reaction workup involves simple filtration followed by silica gel-assisted column chromatography using standard elution protocols that effectively separate the target compound from residual catalysts and ligands. This streamlined purification approach maintains high product purity (>98% by HPLC) while avoiding specialized equipment requirements that would complicate scale-up. The method's robustness across different substrate combinations demonstrates its suitability for producing complex dihydroquinolone derivatives with stringent quality specifications required for pharmaceutical applications.
How to Synthesize Substituted Dihydroquinolone Efficiently
This patented synthesis route represents a significant advancement over conventional methodologies by providing a streamlined pathway that eliminates multiple intermediate steps while maintaining exceptional product quality. The process leverages commercially available starting materials including N-pyridine sulfonyl-o-iodoaniline derivatives and standard olefins that can be readily sourced from global chemical suppliers without supply chain constraints. Reaction optimization has achieved remarkable consistency across diverse substrate combinations through precise control of catalyst loading (20 mol%), ligand ratio (20 mol%), and additive concentration (4.0 equivalents). The methodology has been successfully demonstrated at gram scale with straightforward scalability to larger production volumes while maintaining yield consistency across different molecular architectures. Detailed standardized synthesis procedures are provided below to facilitate seamless implementation within existing manufacturing facilities.
- Add palladium catalyst and ligand to organic solvent with N-pyridine sulfonyl-o-iodoaniline and olefin
- React at 100-120°C for 24-48 hours under controlled conditions
- Perform post-treatment including filtration and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points faced by procurement and supply chain professionals through its inherent design features that enhance operational efficiency while reducing business risks associated with complex intermediate production. The elimination of specialized equipment requirements and hazardous reagents creates significant flexibility in manufacturing site selection while minimizing regulatory compliance burdens across global operations. By utilizing widely available starting materials with established supply chains, this approach substantially reduces raw material sourcing vulnerabilities that commonly disrupt pharmaceutical production schedules. The process demonstrates exceptional robustness across different manufacturing environments while maintaining consistent quality output essential for regulatory compliance in highly scrutinized industries.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and simplified purification workflow significantly reduces operational expenses while maintaining high product quality standards required for pharmaceutical intermediates. By utilizing commercially available starting materials and avoiding specialized equipment requirements, this method delivers substantial cost savings throughout the production cycle without compromising on purity or yield consistency. The streamlined reaction sequence minimizes solvent consumption and waste generation while eliminating costly metal removal steps typically required in conventional syntheses.
- Enhanced Supply Chain Reliability: The use of readily accessible raw materials with multiple qualified suppliers ensures consistent availability regardless of regional disruptions or market fluctuations. This methodology eliminates dependency on rare or geopolitically sensitive reagents while providing flexibility to adapt to changing supply conditions through alternative sourcing strategies. The robust nature of the process maintains consistent output quality even when using different supplier batches of starting materials.
- Scalability and Environmental Compliance: The demonstrated scalability from laboratory to commercial production volumes ensures seamless technology transfer without requiring major process re-engineering efforts. The reduced environmental footprint through minimized waste generation aligns with increasingly stringent regulatory requirements while supporting corporate sustainability initiatives without sacrificing economic viability.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by industry professionals regarding implementation of this patented methodology within commercial manufacturing environments. These responses are derived directly from experimental data documented in patent CN112239456B to provide accurate technical guidance for process development teams.
Q: How does this method improve substrate compatibility compared to conventional approaches?
A: The novel Pd-catalyzed carbonylation process demonstrates exceptional functional group tolerance, accommodating diverse aryl and alkyl substituents without requiring specialized protection/deprotection steps.
Q: What are the key advantages for supply chain reliability in pharmaceutical manufacturing?
A: By utilizing commercially available starting materials and eliminating rare metal catalysts, this method ensures consistent raw material sourcing and reduces production bottlenecks.
Q: How does the process enable cost reduction in API intermediate production?
A: The streamlined reaction sequence with simplified workup procedures significantly reduces operational costs while maintaining high product purity standards required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Dihydroquinolone Supplier
Our patented technology represents a significant advancement in heterocyclic compound synthesis that directly addresses critical challenges faced by global pharmaceutical manufacturers seeking reliable sources for complex intermediates. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical instrumentation. Our dedicated technical teams specialize in adapting patented methodologies like this dihydroquinolone synthesis route to meet specific client requirements while ensuring seamless integration into existing manufacturing workflows.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis route can optimize your specific supply chain needs. Please contact us directly to obtain detailed COA data and comprehensive route feasibility assessments tailored to your production requirements.
