Advanced Palladium-Catalyzed Carbonylation for High-Purity N-Acyl Indole Intermediates
Introduction to Next-Generation Indole Synthesis
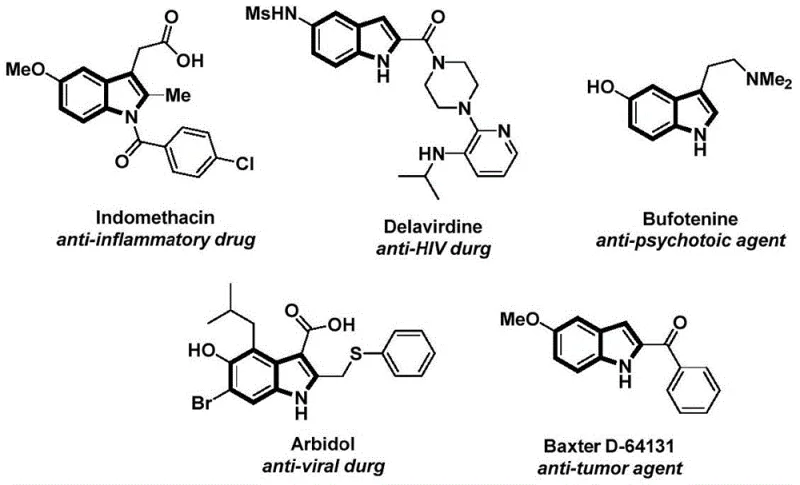
The structural motif of the indole ring serves as a foundational backbone in modern medicinal chemistry, appearing in a vast array of bioactive molecules ranging from anti-inflammatory agents like Indomethacin to potent anti-tumor compounds. As detailed in the recent patent literature, specifically CN112898192B, there is a critical industry shift towards more efficient, one-pot synthetic strategies that can construct these complex heterocycles with high atom economy. This patent discloses a robust preparation method for N-acyl indole compounds, leveraging a sophisticated palladium-catalyzed carbonylation cascade. The significance of this technology lies not just in its chemical elegance, but in its potential to streamline the supply chain for high-value pharmaceutical intermediates. By utilizing readily available 2-alkynyl anilines and aryl iodides, this process bypasses traditional multi-step limitations, offering a direct route to structurally diverse indole derivatives that are essential for drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indoles has often relied on methodologies that present significant operational and safety challenges for large-scale manufacturing. Traditional carbonylation reactions frequently require the use of high-pressure carbon monoxide gas, which poses severe toxicity risks and necessitates expensive, specialized reactor infrastructure to ensure containment and safety compliance. Furthermore, many classical routes involve multi-step sequences where the indole core is constructed first, followed by a separate acylation step, leading to cumulative yield losses and increased waste generation. These conventional approaches often suffer from poor substrate compatibility, particularly when sensitive functional groups are present on the aromatic rings, limiting the chemical space accessible to process chemists. The reliance on harsh conditions or unstable reagents in older methods creates bottlenecks in production scheduling and increases the overall cost of goods sold (COGS) due to complex purification requirements and lower throughput.
The Novel Approach
In stark contrast, the methodology outlined in patent CN112898192B introduces a transformative one-pot strategy that elegantly merges carbonylation and cyclization into a single operational sequence. This novel approach utilizes phenol 1,3,5-tricarboxylate (TFBen) as a safe, solid surrogate for carbon monoxide, effectively mitigating the hazards associated with gaseous CO while maintaining high reaction efficiency. The process operates under relatively mild thermal conditions, typically around 60°C, which preserves the integrity of sensitive functional groups and reduces energy consumption. By employing a dual-catalytic system involving palladium for the initial coupling and silver oxide for the subsequent cyclization, the method achieves excellent yields across a broad range of substrates. This streamlined workflow not only simplifies the post-treatment process—often requiring only filtration and standard column chromatography—but also drastically reduces the time-to-market for new indole-based candidates by collapsing multiple synthetic steps into a single vessel operation.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The chemical elegance of this synthesis lies in its well-defined catalytic cycle, which orchestrates the formation of two distinct bonds (C-N and C-C/C-O) in a sequential manner. The reaction initiates with the oxidative addition of the palladium(0) catalyst into the carbon-iodine bond of the aryl iodide substrate, generating a reactive aryl-palladium intermediate. Crucially, the carbon monoxide required for the acyl group is released in situ from the thermal decomposition of the TFBen additive. This generated CO immediately inserts into the aryl-palladium bond to form an acyl-palladium species. Subsequently, the nucleophilic attack by the amino group of the 2-alkynyl aniline onto this acyl-palladium intermediate leads to the formation of an amide linkage, regenerating the palladium catalyst for the next cycle. This precise orchestration ensures that the carbonyl group is installed with high regioselectivity before the ring closure occurs.
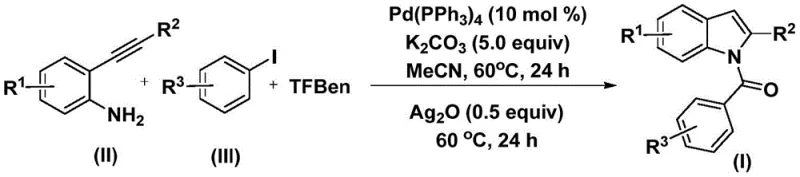
Following the formation of the amide intermediate, the reaction proceeds to the cyclization phase mediated by silver oxide. The addition of Ag2O in the second stage of the reaction is pivotal; it acts as a promoter that facilitates the intramolecular cyclization of the alkyne moiety with the adjacent nitrogen center. This step effectively closes the five-membered pyrrole ring characteristic of the indole scaffold. The use of silver oxide helps to drive the equilibrium towards the product and ensures high conversion rates even with sterically hindered substrates. The mechanistic pathway allows for significant flexibility in the substitution patterns, as evidenced by the tolerance of various electron-donating and electron-withdrawing groups on both the aniline and the aryl iodide components. This robustness is essential for process chemists who need to access diverse analog libraries without redesigning the entire synthetic route for each new derivative.
How to Synthesize N-Acyl Indole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry and timing of reagent addition to maximize yield and purity. The protocol described in the patent emphasizes a two-stage temperature profile where the initial carbonylation is allowed to proceed to completion before the cyclization promoter is introduced. This sequential addition prevents side reactions and ensures that the amide intermediate is fully formed prior to ring closure. For R&D teams looking to replicate or scale this chemistry, the use of anhydrous acetonitrile as the solvent is preferred to maintain catalyst activity and solubility of the organic substrates. The detailed standardized synthesis steps, including specific molar ratios and work-up procedures, are provided in the technical guide below to ensure reproducibility and safety during execution.
- Combine palladium catalyst (Pd(PPh3)4), potassium carbonate, solid CO source (TFBen), 2-alkynyl aniline, and aryl iodide in acetonitrile solvent.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide bond formation.
- Add silver oxide (Ag2O) to the mixture and continue heating at 60°C for another 24 hours to induce cyclization, followed by filtration and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers compelling advantages that directly address the pain points of procurement managers and supply chain directors in the fine chemical sector. The shift from hazardous gaseous reagents to stable solid surrogates fundamentally alters the risk profile of the manufacturing process, allowing for production in facilities that may not be equipped for high-pressure gas handling. This flexibility expands the pool of potential contract manufacturing organizations (CMOs) capable of producing these intermediates, thereby enhancing supply chain resilience and reducing dependency on specialized vendors. Furthermore, the high substrate compatibility means that a single production line can be utilized to manufacture a wide variety of N-acyl indole derivatives simply by swapping out the starting aryl iodides and alkynes, maximizing asset utilization and minimizing changeover times.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the operational workflow and the elimination of expensive safety infrastructure. By replacing high-pressure CO gas with TFBen, manufacturers avoid the capital expenditure associated with gas storage and delivery systems, as well as the ongoing costs of safety monitoring and regulatory compliance for toxic gases. Additionally, the one-pot nature of the reaction reduces solvent usage, labor hours, and waste disposal costs associated with isolating intermediates between steps. The use of commercially available palladium catalysts and standard bases like potassium carbonate ensures that raw material costs remain competitive, while the high yields reported (often exceeding 70-80%) minimize the cost per kilogram of the final active pharmaceutical ingredient (API) intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the use of commodity chemicals as starting materials. Both 2-alkynyl anilines and aryl iodides are widely available from global chemical suppliers, reducing the risk of raw material shortages that can plague proprietary or exotic reagent-dependent syntheses. The mild reaction conditions (60°C) and the use of common solvents like acetonitrile further ensure that the process can be easily transferred between different manufacturing sites globally without requiring unique equipment modifications. This universality allows for a diversified sourcing strategy, enabling companies to qualify multiple suppliers for the same intermediate, thus mitigating the risk of production stoppages due to geopolitical or logistical disruptions.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden inefficiencies, but this carbonylation-cyclization route is inherently designed for scalability. The reaction generates minimal byproducts, and the post-treatment involves straightforward filtration and chromatography, which are unit operations easily adapted from gram to ton scale. From an environmental standpoint, the avoidance of toxic CO gas and the use of a solid CO source align with green chemistry principles, potentially simplifying the permitting process for new manufacturing lines. The high atom economy of the reaction means less waste is generated per unit of product, lowering the environmental footprint and associated disposal fees, which is increasingly critical for meeting corporate sustainability goals and regulatory standards in major markets like the EU and North America.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled a set of frequently asked questions based on the experimental data and technical specifications provided in the patent documentation. These answers address common concerns regarding reaction scope, safety protocols, and purification methods. Understanding these technical nuances is vital for assessing the feasibility of integrating this synthesis route into existing production portfolios or new drug development programs. The following insights are derived directly from the empirical results observed during the optimization of the palladium-catalyzed system.
Q: What are the key advantages of using TFBen over gaseous CO in this synthesis?
A: Using phenol 1,3,5-tricarboxylate (TFBen) as a solid carbon monoxide substitute eliminates the safety hazards and specialized equipment required for handling toxic CO gas, significantly simplifying the operational protocol and enhancing workplace safety.
Q: How does the silver oxide additive influence the reaction outcome?
A: Silver oxide acts as a crucial promoter for the final cyclization step, facilitating the intramolecular closure to form the indole core with high efficiency and ensuring the conversion of the intermediate amide into the desired N-acyl indole structure.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method utilizes commercially available starting materials, mild reaction temperatures (60°C), and standard organic solvents like acetonitrile, making it highly scalable and compatible with existing fine chemical manufacturing infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of robust and scalable synthetic routes for high-value heterocyclic intermediates. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN112898192B and is fully prepared to leverage this advanced palladium-catalyzed methodology for your custom synthesis needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-acyl indole intermediate we deliver meets the exacting standards required for downstream API synthesis.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. By partnering with our technical team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us help you accelerate your drug development timeline with reliable, high-quality chemical solutions.
