Advanced Palladium-Catalyzed Synthesis of N-Acyl Indoles for Scalable Pharmaceutical Manufacturing
Introduction to Next-Generation Indole Synthesis
The structural motif of the indole ring serves as a cornerstone in modern medicinal chemistry, underpinning the efficacy of countless therapeutic agents ranging from anti-inflammatory drugs like Indomethacin to antiviral compounds such as Delavirdine. As depicted in 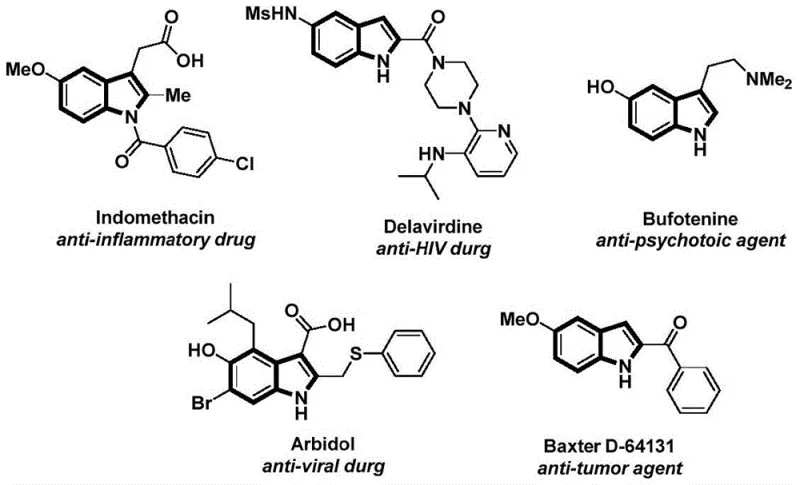 , the versatility of this scaffold is evident in its diverse pharmacological applications. However, the efficient construction of N-acyl indole derivatives has historically presented significant synthetic challenges, often requiring harsh conditions or hazardous reagents. The recent disclosure in patent CN112898192B introduces a transformative methodology that addresses these limitations through a novel palladium-catalyzed carbonylative cyclization strategy. This technology leverages a solid carbon monoxide surrogate to achieve high-efficiency synthesis under remarkably mild conditions, representing a paradigm shift for manufacturers seeking reliable pharmaceutical intermediate supplier capabilities. By integrating this advanced protocol, production facilities can enhance their portfolio of high-purity OLED material and API precursors while adhering to stringent safety and environmental standards.
, the versatility of this scaffold is evident in its diverse pharmacological applications. However, the efficient construction of N-acyl indole derivatives has historically presented significant synthetic challenges, often requiring harsh conditions or hazardous reagents. The recent disclosure in patent CN112898192B introduces a transformative methodology that addresses these limitations through a novel palladium-catalyzed carbonylative cyclization strategy. This technology leverages a solid carbon monoxide surrogate to achieve high-efficiency synthesis under remarkably mild conditions, representing a paradigm shift for manufacturers seeking reliable pharmaceutical intermediate supplier capabilities. By integrating this advanced protocol, production facilities can enhance their portfolio of high-purity OLED material and API precursors while adhering to stringent safety and environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing N-acyl indole frameworks have frequently relied on the direct use of carbon monoxide gas, a reagent that poses severe logistical and safety hurdles in an industrial setting. The handling of pressurized CO cylinders requires specialized infrastructure, rigorous leak detection systems, and extensive operator training, all of which contribute to inflated operational expenditures and increased risk profiles. Furthermore, conventional carbonylation reactions often necessitate elevated temperatures and pressures to drive the equilibrium forward, which can lead to the degradation of sensitive functional groups and the formation of complex impurity profiles that are difficult to purge. These factors collectively hinder the commercial scale-up of complex polymer additives and pharmaceutical intermediates, creating bottlenecks in supply chains that demand rapid turnaround times. Additionally, the reliance on stoichiometric amounts of toxic metal oxidants in older methodologies generates substantial heavy metal waste, complicating downstream processing and environmental compliance efforts.
The Novel Approach
In stark contrast to these legacy techniques, the methodology outlined in CN112898192B employs 1,3,5-tricarboxylic acid phenol ester (TFBen) as a safe, solid-state carbon monoxide surrogate, effectively decoupling the synthesis from the dangers associated with gaseous CO. This innovative approach allows the reaction to proceed at a moderate temperature of 60°C in acetonitrile, significantly reducing energy consumption and thermal stress on the molecular architecture. The process utilizes a dual-step sequence where the initial carbonylation is followed by a silver oxide-mediated cyclization, ensuring high conversion rates and excellent regioselectivity. As illustrated in the general reaction scheme 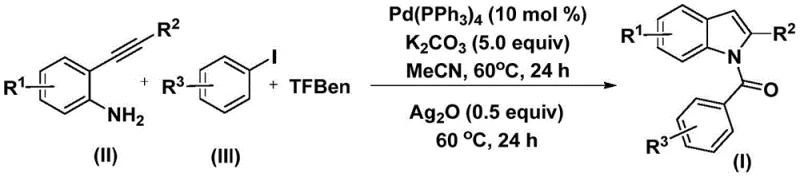 , this one-pot strategy streamlines the workflow by minimizing isolation steps between the coupling and cyclization phases. The result is a robust, operationally simple protocol that offers substantial cost savings in electronic chemical manufacturing by reducing waste generation and eliminating the need for high-pressure reactors.
, this one-pot strategy streamlines the workflow by minimizing isolation steps between the coupling and cyclization phases. The result is a robust, operationally simple protocol that offers substantial cost savings in electronic chemical manufacturing by reducing waste generation and eliminating the need for high-pressure reactors.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The mechanistic pathway of this transformation is a sophisticated orchestration of organometallic steps initiated by the oxidative addition of the palladium catalyst into the aryl iodide bond. This critical activation step generates an aryl-palladium intermediate that is primed for the subsequent insertion of carbon monoxide released in situ from the thermal decomposition of TFBen. The insertion of CO into the palladium-carbon bond forms an acyl-palladium species, which then undergoes nucleophilic attack by the amine group of the 2-alkynyl aniline substrate. This sequence constructs the amide linkage with high fidelity, setting the stage for the final ring-closing event. The presence of silver oxide in the second stage acts as a crucial promoter, facilitating the intramolecular cyclization of the alkyne moiety to form the indole core while regenerating the active catalytic species. This mechanistic elegance ensures that the reaction tolerates a wide array of substituents, including halogens, alkyl groups, and electron-donating methoxy functionalities, without compromising the integrity of the final product.
From an impurity control perspective, the use of a solid CO surrogate provides a distinct advantage by maintaining a low, steady concentration of carbon monoxide throughout the reaction vessel, thereby suppressing side reactions such as homocoupling of the aryl iodide or over-carbonylation. The mild reaction conditions further mitigate the risk of thermal decomposition of the starting materials or the product, leading to a cleaner crude reaction profile that simplifies downstream purification. The compatibility of this system with various functional groups means that manufacturers can produce high-purity pharmaceutical intermediates with minimal chromatographic burden. By understanding these mechanistic nuances, process chemists can fine-tune parameters such as catalyst loading and base equivalents to optimize yield and purity, ensuring that the final material meets the rigorous specifications required for clinical and commercial applications.
How to Synthesize N-Acyl Indole Compounds Efficiently
The execution of this synthesis protocol is designed for reproducibility and ease of handling, making it an ideal candidate for technology transfer from laboratory bench to pilot plant. The procedure involves charging a reaction vessel with the palladium catalyst, potassium carbonate base, the solid CO source TFBen, the 2-alkynyl aniline, and the aryl iodide in acetonitrile. The mixture is heated to 60°C for 24 hours to allow the carbonylation and amidation to reach completion. Subsequently, silver oxide is introduced to the reaction mixture, and heating is continued for an additional 24 hours to drive the cyclization to full conversion. This standardized approach minimizes variability and ensures consistent batch-to-batch quality, which is paramount for maintaining supply chain reliability.
- Combine palladium catalyst, potassium carbonate, 1,3,5-tricarboxylic acid phenol ester (TFBen), 2-alkynyl aniline, and aryl iodide in an organic solvent such as acetonitrile.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and coupling steps.
- Add silver oxide to the mixture and continue heating at 60°C for another 24 hours to complete the cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers compelling strategic benefits that extend beyond mere chemical efficiency. The elimination of gaseous carbon monoxide from the process inventory drastically simplifies regulatory compliance and reduces the insurance premiums associated with storing hazardous materials. This shift not only enhances the safety culture within the manufacturing facility but also removes a significant bottleneck in the sourcing of raw materials, as solid surrogates are easier to transport and store than compressed gases. Furthermore, the use of commercially available reagents such as tetrakis(triphenylphosphine)palladium and silver oxide ensures a stable supply chain, reducing the risk of production delays caused by specialty reagent shortages. The ability to operate at atmospheric pressure and moderate temperatures also lowers the barrier to entry for contract manufacturing organizations, enabling faster scale-up timelines.
- Cost Reduction in Manufacturing: The transition to a solid CO surrogate system eliminates the capital expenditure required for high-pressure autoclaves and specialized gas handling infrastructure, leading to significant overhead reductions. By avoiding the use of toxic gases, the facility also saves on the costs associated with specialized ventilation systems and continuous gas monitoring equipment. The high atom economy of the reaction, combined with the efficient use of catalysts, ensures that raw material costs are optimized, providing a competitive edge in pricing for high-volume orders. Additionally, the simplified workup procedure reduces solvent consumption and labor hours, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against variations in substrate electronics means that a single protocol can be applied to a diverse library of N-acyl indole derivatives, simplifying inventory management and production scheduling. The reliance on stable, shelf-stable reagents mitigates the risk of supply disruptions that often plague processes dependent on unstable or perishable intermediates. This flexibility allows manufacturers to respond rapidly to fluctuating market demands for specific API intermediates, ensuring that customers receive their orders on time without compromising on quality. The scalability of the process from gram to kilogram scales has been demonstrated, providing confidence in the ability to meet large-volume contractual obligations.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of volatile organic solvents other than acetonitrile align perfectly with green chemistry principles, reducing the environmental footprint of the manufacturing process. The generation of less hazardous waste streams simplifies effluent treatment and lowers disposal costs, which is increasingly important in regions with strict environmental regulations. The process is inherently safer for operators, reducing the likelihood of workplace accidents and associated downtime. This commitment to sustainability and safety enhances the corporate reputation of the manufacturer, making them a preferred partner for multinational corporations with rigorous vendor audit standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-acyl indole synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the process capabilities. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this method into their existing production workflows. We encourage potential partners to review these points carefully to appreciate the full value proposition of this advanced synthetic route.
Q: What is the primary advantage of using TFBen over carbon monoxide gas?
A: Using 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide surrogate eliminates the need for handling toxic, high-pressure CO gas, significantly enhancing operational safety and simplifying equipment requirements for scale-up.
Q: What represents the typical yield range for this synthesis method?
A: According to the patent data, the reaction demonstrates robust efficiency with isolated yields ranging from 44% to 82% across various substrates, indicating good tolerance for different functional groups.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method utilizes commercially available reagents, operates at mild temperatures (60°C), and avoids hazardous gases, making it highly amenable to commercial scale-up and continuous manufacturing processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications and advanced materials. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from concept to reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch. By leveraging the efficiencies of the palladium-catalyzed carbonylation technology, we can offer our clients a reliable source of complex indole derivatives that are both cost-effective and environmentally responsible.
We invite you to collaborate with us to explore how this innovative synthesis method can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to serve as your long-term strategic partner in the fine chemical industry.
