Scalable Palladium-Catalyzed Synthesis of N-Acyl Indoles for Advanced Pharmaceutical Manufacturing
Scalable Palladium-Catalyzed Synthesis of N-Acyl Indoles for Advanced Pharmaceutical Manufacturing
The structural motif of indole is ubiquitous in medicinal chemistry, serving as the core scaffold for a vast array of bioactive molecules ranging from anti-inflammatory agents like Indomethacin to potent anti-tumor compounds. As depicted in the reference structures of known pharmaceutical agents, the N-acyl modification of the indole ring is a critical transformation that often dictates the biological activity and pharmacokinetic profile of the final drug candidate. 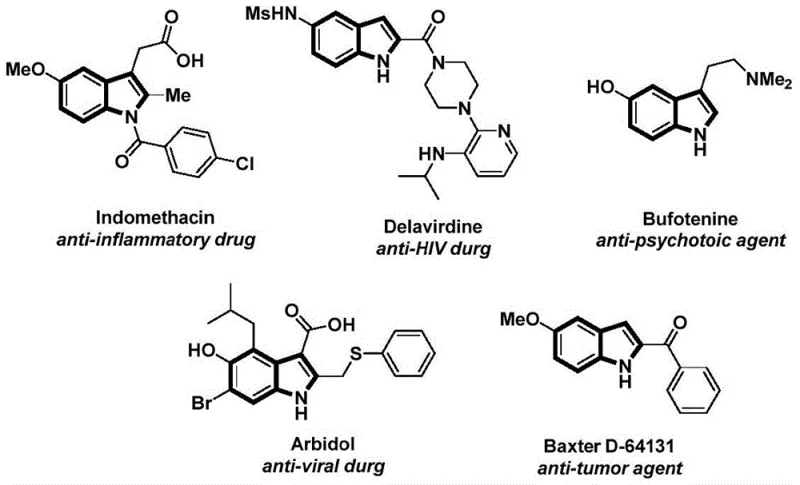 Recent advancements in organic synthesis have sought to streamline the construction of these complex heterocycles. Patent CN112898192B introduces a robust and efficient methodology for the preparation of N-acyl indole compounds, utilizing a palladium-catalyzed carbonylation cyclization strategy. This technical breakthrough addresses long-standing challenges in heterocyclic synthesis by employing readily available starting materials—specifically 2-alkynyl anilines and aryl iodides—and operating under remarkably mild conditions. For industrial stakeholders, this represents a significant opportunity to optimize the supply chain for high-value pharmaceutical intermediates, ensuring both purity and process reliability.
Recent advancements in organic synthesis have sought to streamline the construction of these complex heterocycles. Patent CN112898192B introduces a robust and efficient methodology for the preparation of N-acyl indole compounds, utilizing a palladium-catalyzed carbonylation cyclization strategy. This technical breakthrough addresses long-standing challenges in heterocyclic synthesis by employing readily available starting materials—specifically 2-alkynyl anilines and aryl iodides—and operating under remarkably mild conditions. For industrial stakeholders, this represents a significant opportunity to optimize the supply chain for high-value pharmaceutical intermediates, ensuring both purity and process reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of N-acyl indoles has relied on methods that often suffer from significant operational drawbacks, limiting their utility in large-scale commercial manufacturing. Classical approaches frequently involve the use of gaseous carbon monoxide (CO), which poses severe safety hazards due to its high toxicity and flammability, requiring specialized high-pressure equipment and rigorous safety protocols that inflate capital expenditure. Furthermore, many conventional routes necessitate harsh reaction conditions, such as extreme temperatures or strong acidic/basic environments, which can lead to poor functional group tolerance and the degradation of sensitive substrates. These limitations often result in lower overall yields, complex purification processes to remove toxic metal residues or byproducts, and extended production timelines that hinder rapid scale-up. The reliance on unstable reagents and dangerous gases creates a bottleneck for reliable pharmaceutical intermediate supplier operations, making the search for safer, solid-state alternatives a priority for modern process chemistry.
The Novel Approach
The methodology disclosed in the patent offers a transformative solution by replacing hazardous gaseous CO with a solid carbon monoxide substitute, specifically 1,3,5-tricarboxylic acid phenol ester (TFBen). This innovation allows the reaction to proceed in a standard laboratory or plant setting without the need for high-pressure autoclaves, drastically simplifying the engineering requirements. The process operates at a moderate temperature of 60°C in acetonitrile, a common and recoverable solvent, ensuring energy efficiency and ease of handling. By integrating the carbonylation and cyclization steps into a streamlined sequence, this approach achieves high atom economy and reduces waste generation. The use of aryl iodides and 2-alkynyl anilines as starting materials provides a modular platform where diverse substituents can be introduced easily, enabling the rapid generation of compound libraries for drug discovery while maintaining a pathway suitable for cost reduction in API manufacturing.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The core of this synthetic strategy lies in a sophisticated palladium-catalyzed cascade that elegantly constructs the indole skeleton and installs the acyl group in a controlled manner. The reaction initiates with the oxidative addition of the palladium(0) catalyst into the carbon-iodine bond of the aryl iodide substrate, generating a reactive aryl-palladium(II) intermediate. Subsequently, carbon monoxide, which is slowly and safely released in situ from the decomposition of TFBen, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the amino group of the 2-alkynyl aniline, followed by reductive elimination to yield an amide precursor. 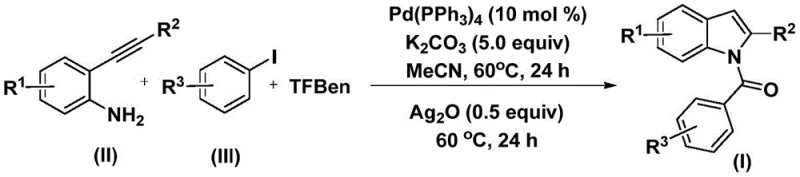 The final and crucial step involves the addition of silver oxide (Ag2O), which acts as a promoter to facilitate the intramolecular cyclization of the amide, closing the five-membered pyrrole ring to furnish the target N-acyl indole product. This mechanistic pathway ensures high regioselectivity and minimizes the formation of unwanted isomers.
The final and crucial step involves the addition of silver oxide (Ag2O), which acts as a promoter to facilitate the intramolecular cyclization of the amide, closing the five-membered pyrrole ring to furnish the target N-acyl indole product. This mechanistic pathway ensures high regioselectivity and minimizes the formation of unwanted isomers.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity OLED material or pharmaceutical grades. The mild conditions prevent the polymerization of the alkyne moiety, a common side reaction in harsher cyclization protocols. Additionally, the use of TFBen ensures a steady, low concentration of CO, which prevents the formation of bis-carbonylated byproducts that can occur when CO pressure is too high. The compatibility with various functional groups, including halogens like fluorine and chlorine, as well as electron-donating groups like methoxy and methyl, is preserved throughout the catalytic cycle. Experimental data from the patent indicates that substrates bearing electron-withdrawing groups on the aryl iodide component generally afford higher yields, suggesting that the oxidative addition step is not the rate-limiting factor, but rather the subsequent insertion and cyclization steps are well-balanced. This deep understanding of the reaction kinetics allows for precise optimization of stoichiometry to maximize throughput.
How to Synthesize N-Acyl Indole Efficiently
To implement this synthesis effectively, operators must adhere to a precise two-stage heating protocol that leverages the unique reactivity of the silver oxide promoter. The process begins by combining the palladium catalyst, typically tetrakis(triphenylphosphine)palladium, with potassium carbonate as the base, the solid CO source TFBen, the 2-alkynyl aniline, and the aryl iodide in acetonitrile. This mixture is heated at 60°C for 24 hours to allow the initial carbonylation and amide bond formation to reach completion. Following this first stage, silver oxide is introduced directly into the reaction vessel, and the heating is continued at 60°C for an additional 24 hours to drive the cyclization to completion. Detailed standardized synthesis steps see the guide below.
- Combine palladium catalyst, potassium carbonate, TFBen (CO source), 2-alkynyl aniline, and aryl iodide in acetonitrile.
- Heat the mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide formation.
- Add silver oxide to the reaction mixture and continue heating at 60°C for another 24 hours to induce cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits regarding cost stability and operational flexibility. The shift away from gaseous carbon monoxide eliminates the logistical complexities and regulatory burdens associated with storing and transporting hazardous compressed gases, thereby reducing insurance costs and facility compliance overheads. Furthermore, the reliance on commercially available and inexpensive reagents such as aryl iodides and simple aniline derivatives ensures a robust supply chain that is less susceptible to market volatility compared to exotic or custom-synthesized precursors. The ability to run the reaction at atmospheric pressure in standard glass-lined reactors significantly lowers the barrier to entry for contract manufacturing organizations (CMOs), allowing for rapid technology transfer and scale-up without massive capital investment in high-pressure infrastructure.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive high-pressure equipment and the use of TFBen as a safe, solid CO surrogate. By avoiding the need for specialized autoclaves and the associated safety monitoring systems, manufacturers can achieve substantial cost savings in CAPEX and OPEX. Additionally, the high reaction efficiency and good yields reported across a wide range of substrates mean that less raw material is wasted, directly improving the cost of goods sold (COGS). The simplified post-treatment procedure, which involves basic filtration and column chromatography, further reduces labor and solvent consumption costs compared to multi-step traditional syntheses that require extensive workups.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including 2-alkynyl anilines and aryl iodides, are commodity chemicals that can be sourced from multiple global suppliers, mitigating the risk of single-source dependency. The stability of the solid CO source (TFBen) allows for long-term storage without degradation, ensuring that production schedules are not disrupted by reagent shelf-life issues. This reliability is crucial for maintaining continuous production lines for critical pharmaceutical intermediates, ensuring that downstream API synthesis is never halted due to raw material shortages. The mild reaction conditions also reduce wear and tear on reactor vessels, extending equipment lifespan and minimizing unplanned maintenance downtime.
- Scalability and Environmental Compliance: From an environmental standpoint, this method aligns with green chemistry principles by utilizing a safer carbon source and generating fewer hazardous byproducts. The absence of high-pressure CO gas significantly reduces the risk of accidental release, enhancing workplace safety and simplifying environmental permitting processes. The reaction's high atom economy and the potential for solvent recovery (acetonitrile) contribute to a reduced environmental footprint, helping companies meet increasingly stringent sustainability targets. The scalability of the process is evidenced by its straightforward execution, which can be easily adapted from gram-scale laboratory synthesis to multi-kilogram pilot plant runs without encountering the heat transfer or mixing issues often associated with exothermic high-pressure reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this N-acyl indole synthesis protocol. These answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity for R&D teams evaluating this technology for adoption. Understanding these nuances is essential for optimizing reaction parameters and predicting outcomes for novel substrate combinations.
Q: What is the primary advantage of using TFBen in this synthesis?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) serves as a solid, safe, and easy-to-handle carbon monoxide substitute, eliminating the need for hazardous high-pressure CO gas cylinders while maintaining high reaction efficiency.
Q: What is the role of Silver Oxide (Ag2O) in the reaction mechanism?
A: Silver oxide acts as a critical promoter in the second stage of the reaction, facilitating the intramolecular cyclization of the intermediate amide to form the final N-acyl indole structure under mild thermal conditions.
Q: Does this method support diverse functional groups on the substrates?
A: Yes, the protocol demonstrates excellent substrate compatibility, tolerating various substituents such as halogens (F, Cl, Br), alkyl groups (Me, tBu), and alkoxy groups (OMe) on both the aniline and aryl iodide components without significant yield loss.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and potency. By leveraging advanced catalytic technologies like the palladium-mediated carbonylation described in CN112898192B, we can offer clients a competitive edge through superior process economics and consistent quality.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis for an existing supply chain or need to evaluate the feasibility of a new target molecule, our experts are ready to provide specific COA data and route feasibility assessments. Contact us today to explore how our manufacturing capabilities can support your pipeline and accelerate your time to market.
