Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
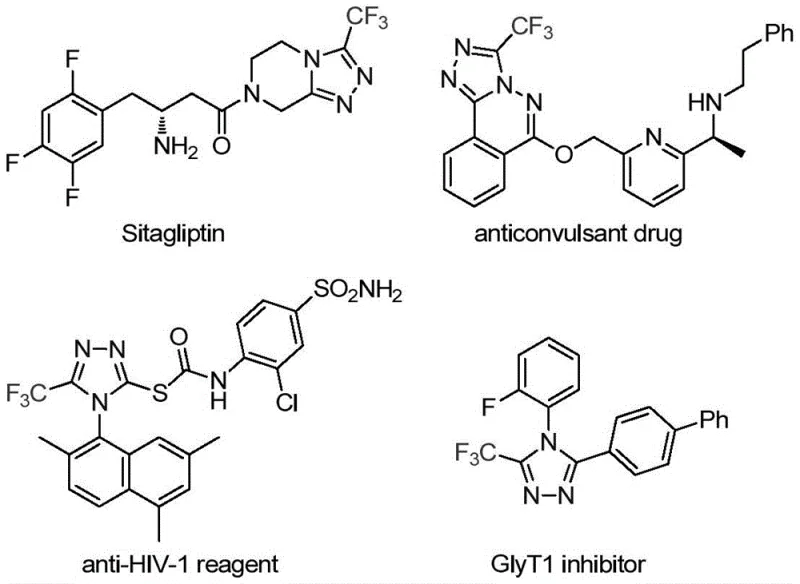
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are critical for enhancing metabolic stability and bioavailability. A groundbreaking development in this domain is detailed in patent CN113307778A, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a significant leap forward for manufacturers aiming to produce reliable pharmaceutical intermediate supplier grades of these valuable scaffolds. The presence of the trifluoromethyl group is not merely structural; it fundamentally alters the physicochemical properties of the parent molecule, offering improved lipophilicity and electronegativity that are essential for modern drug design, as seen in blockbuster medications like Sitagliptin. By leveraging a dual-metal catalytic system, this invention addresses long-standing challenges in heterocyclic synthesis, providing a pathway that is both economically viable and chemically elegant for the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on synthetic routes that are often fraught with operational difficulties and safety concerns. Traditional literature reports predominantly describe the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These classical approaches frequently necessitate the use of hazardous hydrazine derivatives, which pose significant handling risks and generate toxic waste streams that complicate environmental compliance. Furthermore, alternative copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane often suffer from narrow substrate scope and require strictly anhydrous conditions that are difficult to maintain on a large industrial scale. The reliance on unstable intermediates and the generation of stoichiometric amounts of salt byproducts in these older methods result in poor atom economy and inflated production costs, making them less attractive for cost reduction in API manufacturing where margin compression is a constant pressure.
The Novel Approach
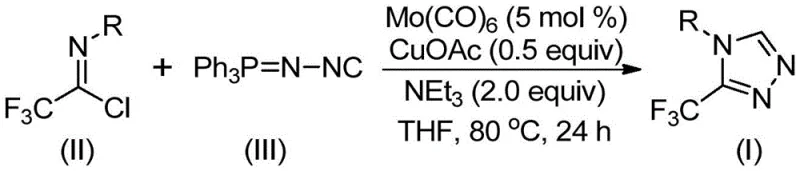
In stark contrast to these legacy techniques, the methodology outlined in the patent introduces a streamlined cycloaddition reaction that utilizes cheap and easily obtainable functionalized isonitriles and trifluoroethylimidoyl chloride as starting materials. This innovative route operates under remarkably mild conditions, typically requiring temperatures between 70°C and 90°C, which drastically reduces energy consumption compared to high-temperature pyrolysis methods. The use of a molybdenum and copper co-catalytic system enables the direct assembly of the triazole ring with high regioselectivity, effectively bypassing the need for pre-functionalized hydrazine intermediates. This shift in synthetic strategy not only simplifies the operational workflow but also broadens the applicability of the method, allowing for the synthesis of various官能团 (functional group) substituted derivatives with impressive efficiency. The ability to scale this reaction to the gram level with consistent results suggests a clear path toward commercial scale-up of complex pharmaceutical intermediates, offering a distinct competitive advantage for producers.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The success of this transformation lies in the synergistic interaction between the molybdenum hexacarbonyl and the cuprous acetate catalysts, which orchestrate a sophisticated [3+2] cycloaddition mechanism. Mechanistically, the molybdenum species is believed to activate the functionalized isonitrile by forming a transient metal-isocyanide complex, thereby increasing the nucleophilicity of the carbon center. Simultaneously, the copper catalyst promotes the activation of the trifluoroethylimidoyl chloride, facilitating the initial attack that leads to the formation of a five-membered ring intermediate. This cooperative catalysis is crucial for overcoming the kinetic barriers associated with triazole ring closure, ensuring that the reaction proceeds smoothly even with sterically hindered substrates. The subsequent elimination of triphenylphosphine oxide, driven by the presence of water or molecular sieves in the system, serves as the thermodynamic driving force that pushes the equilibrium toward the final 3-trifluoromethyl-substituted 1,2,4-triazole product. Understanding this mechanistic nuance is vital for R&D teams looking to optimize reaction parameters for specific substrate classes.
From an impurity control perspective, this catalytic system offers inherent advantages due to its high selectivity and clean reaction profile. The use of molecular sieves in the reaction mixture plays a dual role: it acts as a desiccant to protect sensitive intermediates from hydrolysis while also sequestering water generated during the phosphine oxide elimination step, preventing side reactions that could lead to hydrolyzed byproducts. Furthermore, the choice of triethylamine as a base ensures that acidic byproducts are neutralized effectively without promoting decomposition of the sensitive triazole ring. The resulting crude reaction mixtures are typically clean enough to be purified via standard silica gel column chromatography, indicating that the formation of polymeric tars or intractable side products is minimal. This high level of purity is essential for meeting the stringent quality specifications required for high-purity OLED material or pharmaceutical intermediate applications, where trace impurities can have detrimental effects on downstream biological activity or device performance.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis involves a straightforward one-pot procedure that minimizes unit operations and solvent exchanges. To initiate the reaction, precise stoichiometric amounts of molybdenum hexacarbonyl, cuprous acetate, triethylamine, and molecular sieves are combined with the trifluoroethylimidoyl chloride and functionalized isonitrile in a suitable organic solvent such as tetrahydrofuran (THF). The reaction mixture is then heated to a controlled temperature range of 70°C to 90°C and maintained under stirring for a period of 18 to 30 hours to ensure full conversion of the starting materials. Following the reaction completion, the workup procedure is notably simple, involving filtration to remove solid catalysts and sieves, followed by adsorption onto silica gel and purification via column chromatography. For a comprehensive understanding of the specific molar ratios and optimization data, the detailed standardized synthesis steps are provided in the guide below.
- Combine molybdenum hexacarbonyl, cuprous acetate, triethylamine, molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The transition from hazardous hydrazine-based chemistry to this stable imidoyl chloride/isocyanide coupling eliminates the need for specialized containment equipment and expensive waste disposal protocols associated with toxic reagents. This fundamental shift in raw material selection translates into substantial cost savings by reducing the overhead costs related to safety compliance and environmental remediation. Moreover, the catalysts employed, specifically cuprous acetate and molybdenum hexacarbonyl, are commodity chemicals that are readily available from multiple global suppliers, mitigating the risk of supply chain disruptions caused by single-source dependencies. The robustness of the reaction conditions further enhances supply chain reliability by reducing the likelihood of batch failures due to minor fluctuations in temperature or moisture content.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive and commercially available starting materials, which avoids the premium pricing often associated with specialized heterocyclic building blocks. By eliminating the need for expensive ligands or precious metal catalysts like palladium or rhodium, the overall catalyst loading cost is drastically simplified, leading to a more favorable cost of goods sold (COGS). Additionally, the high reaction efficiency and yield reported in the patent examples mean that less raw material is wasted, maximizing the output per kilogram of input and driving down the unit cost of production significantly.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents such as trifluoroethylimidoyl chloride and functionalized isonitriles ensures that inventory can be held safely for extended periods without degradation, providing a buffer against market volatility. The simplicity of the post-treatment process, which utilizes standard silica gel chromatography rather than complex crystallization or distillation setups, allows for faster turnaround times in the manufacturing suite. This operational agility enables manufacturers to respond more quickly to fluctuating demand signals from downstream clients, thereby reducing lead time for high-purity pharmaceutical intermediates and strengthening customer relationships through consistent on-time delivery.
- Scalability and Environmental Compliance: The demonstration of this chemistry at the gram level with excellent yields provides a strong foundation for scaling to kilogram and multi-ton quantities without encountering the heat transfer or mixing issues common in exothermic hydrazine reactions. The absence of heavy metal contaminants in the final product simplifies the purification train, reducing the solvent volume required for recrystallization and lowering the overall E-factor of the process. This alignment with green chemistry principles not only satisfies increasingly strict regulatory requirements but also appeals to environmentally conscious partners who prioritize sustainable manufacturing practices in their vendor selection criteria.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled a set of frequently asked questions based on the technical disclosures within the patent documentation. These inquiries address common concerns regarding substrate scope, reaction scalability, and purification requirements, providing clarity on how this method compares to existing state-of-the-art technologies. The answers provided below are derived directly from the experimental data and mechanistic proposals found in the source material, ensuring accuracy and relevance for process development scientists. We encourage stakeholders to review these insights carefully to determine the optimal integration of this synthetic route into their existing pipelines.
Q: What are the key advantages of this Mo/Cu co-catalyzed method over traditional triazole synthesis?
A: Unlike traditional methods that often require harsh conditions or complex multi-step sequences involving hydrazines, this novel approach utilizes readily available trifluoroethylimidoyl chloride and functionalized isonitriles under mild thermal conditions (70-90°C), significantly simplifying the operational workflow and improving safety profiles.
Q: Can this synthesis method tolerate diverse functional groups on the substrate?
A: Yes, the protocol demonstrates excellent substrate tolerance, successfully accommodating various substituents including alkyl, alkoxy, halogen, and nitro groups on the aromatic ring, which allows for the flexible design of diverse molecular libraries for drug discovery.
Q: Is this process suitable for large-scale industrial manufacturing?
A: The method has been validated at the gram level with high efficiency and uses inexpensive catalysts like cuprous acetate and molybdenum hexacarbonyl, indicating strong potential for cost-effective scale-up to kilogram or tonnage production without requiring exotic reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of next-generation therapeutics and functional materials. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify identity and assay. By partnering with us, clients gain access to a supply chain that is not only robust and reliable but also deeply knowledgeable about the nuances of fluorine chemistry and transition metal catalysis.
We invite you to engage with our technical procurement team to discuss how this innovative Mo/Cu co-catalyzed synthesis can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis for an existing route or need to evaluate the feasibility of a new analog series, our experts are ready to provide specific COA data and route feasibility assessments. Let us help you accelerate your development timeline and secure a competitive advantage in the marketplace through superior chemical manufacturing solutions.
