Revolutionizing Pharmaceutical Intermediate Production via Efficient Multi-Component Palladium Catalysis
Introduction to Next-Generation Heterocyclic Synthesis
The landscape of modern pharmaceutical development is increasingly defined by the complexity and specificity of molecular scaffolds required for next-generation therapeutics. In this context, the groundbreaking methodology disclosed in Chinese Patent CN115353511A represents a paradigm shift in the construction of complex heterocyclic architectures, specifically focusing on carbonyl-bridged biheterocyclic compounds. These structures, which often combine indolinone and imidazole motifs, are ubiquitous in natural products and bioactive molecules exhibiting broad-spectrum pharmacological activities. The patent introduces a highly efficient, transition metal palladium-catalyzed multi-component reaction that streamlines the assembly of these valuable cores. By leveraging a cascade reaction sequence, this technology enables the simultaneous formation of multiple chemical bonds in a single operational step, thereby drastically reducing the synthetic burden traditionally associated with building such intricate bis-heterocyclic systems. For research and development teams seeking reliable pharmaceutical intermediate suppliers, this approach offers a robust pathway to access diverse chemical libraries with enhanced speed and precision.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of biheterocyclic compounds has been fraught with significant challenges that hinder both laboratory exploration and industrial scalability. Traditional strategies often rely on the direct coupling of two pre-formed heterocyclic substrates, a process that frequently suffers from low atom economy and poor regioselectivity. Alternatively, oxidative cyclization reactions involving bifunctional nucleophiles and activated methyl-substituted heterocycles often require harsh oxidizing agents and elevated temperatures, which can lead to the decomposition of sensitive functional groups and the generation of complex impurity profiles. Furthermore, conventional carbonylation reactions, while powerful for introducing carbonyl linkers, typically necessitate the use of toxic carbon monoxide gas under high pressure. This requirement imposes severe safety constraints, demanding specialized high-pressure equipment and rigorous safety protocols that significantly inflate capital expenditure and operational risks in manufacturing facilities. These limitations collectively create a bottleneck in the cost reduction in API manufacturing, particularly for complex scaffolds requiring precise carbonyl bridging.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a sophisticated yet operationally simple multi-component coupling strategy that elegantly bypasses the need for hazardous gaseous reagents. As illustrated in the reaction scheme below, the process integrates trifluoroethylimidoyl chloride, propargylamine, and an acrylamide derivative in a single pot, mediated by a palladium catalyst system. 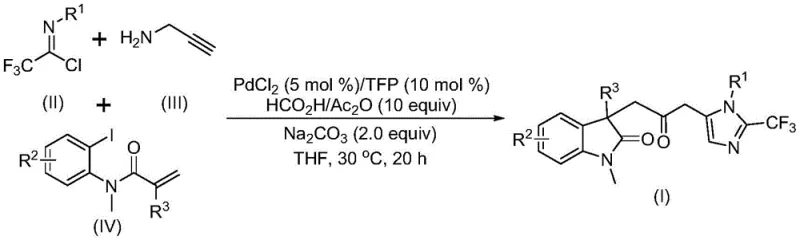 This innovative protocol employs a mixture of formic acid and acetic anhydride as a safe, liquid-phase carbon monoxide substitute, which generates the necessary CO in situ under mild conditions. The reaction proceeds at a remarkably low temperature of 30°C, demonstrating exceptional energy efficiency and minimizing thermal degradation of reactants. This method not only enhances safety by eliminating high-pressure gas handling but also improves the overall process mass intensity by combining multiple synthetic steps into one seamless transformation. For procurement managers, this translates to a streamlined supply chain where complex intermediates can be produced with fewer unit operations and reduced waste generation.
This innovative protocol employs a mixture of formic acid and acetic anhydride as a safe, liquid-phase carbon monoxide substitute, which generates the necessary CO in situ under mild conditions. The reaction proceeds at a remarkably low temperature of 30°C, demonstrating exceptional energy efficiency and minimizing thermal degradation of reactants. This method not only enhances safety by eliminating high-pressure gas handling but also improves the overall process mass intensity by combining multiple synthetic steps into one seamless transformation. For procurement managers, this translates to a streamlined supply chain where complex intermediates can be produced with fewer unit operations and reduced waste generation.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
To fully appreciate the technical sophistication of this synthesis, one must delve into the intricate catalytic cycle that drives the formation of the carbonyl-bridged biheterocyclic core. The mechanism is believed to initiate with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating an aryl-palladium intermediate. This is followed by an intramolecular Heck-type reaction, where the palladium center inserts into the alkene moiety to form a divalent alkyl-palladium species, effectively constructing the indolinone ring system. Subsequently, the carbon monoxide released from the formic acid/acetic anhydride mixture coordinates to the palladium center and undergoes migratory insertion to yield an acyl-palladium intermediate. Concurrently, in a parallel pathway facilitated by the base, the trifluoroethylimidoyl chloride reacts with propargylamine to form a trifluoroacetamidine compound via intermolecular carbon-nitrogen bond formation. The final stage involves the activation of this amidine species by the acyl-palladium intermediate, triggering an intramolecular cyclization that closes the imidazole ring and releases the final product while regenerating the active palladium catalyst.
Beyond the primary catalytic cycle, the robustness of this method lies in its exceptional impurity control mechanisms, which are critical for producing high-purity OLED material or pharmaceutical intermediates. The mild reaction temperature of 30°C plays a pivotal role in suppressing side reactions such as polymerization of the acrylamide or hydrolysis of the imidoyl chloride, which are common pitfalls in more aggressive conditions. Furthermore, the use of trifurylphosphine as a ligand stabilizes the palladium center, preventing the formation of palladium black and ensuring consistent catalytic turnover throughout the 12 to 20-hour reaction window. The compatibility with a wide range of substituents, including electron-withdrawing groups like nitro and trifluoromethyl, as well as electron-donating groups like methoxy and alkyl, underscores the versatility of the electronic environment tolerated by the catalyst. 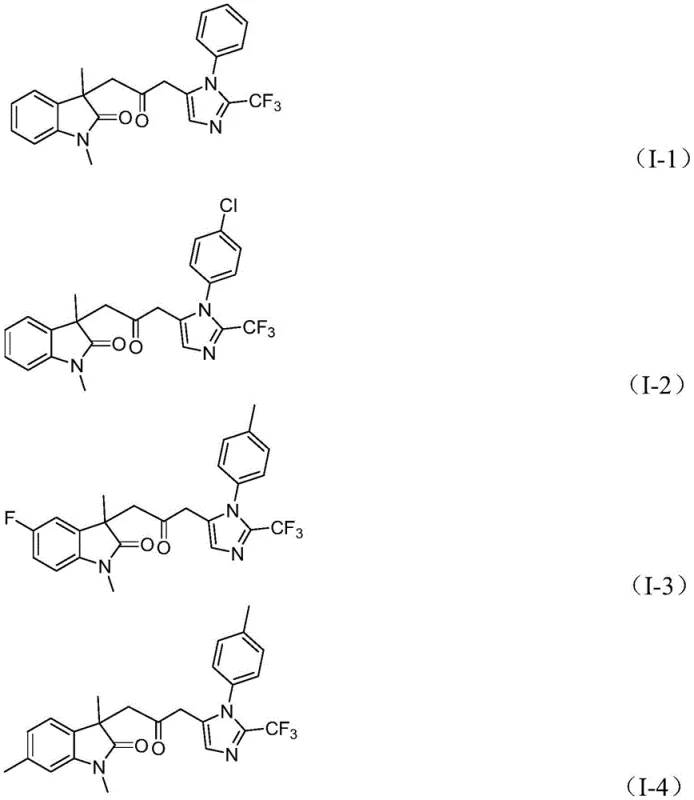 This broad substrate scope ensures that the process can be adapted for the commercial scale-up of complex polymer additives or drug candidates without extensive re-optimization, providing a reliable foundation for diverse chemical synthesis projects.
This broad substrate scope ensures that the process can be adapted for the commercial scale-up of complex polymer additives or drug candidates without extensive re-optimization, providing a reliable foundation for diverse chemical synthesis projects.
How to Synthesize Carbonyl-Bridged Biheterocycles Efficiently
Implementing this advanced synthesis route requires careful attention to reagent quality and reaction parameters to maximize yield and purity. The protocol is designed to be user-friendly, utilizing commercially available starting materials such as propargylamine and acryloyl chloride derivatives, which reduces the barrier to entry for laboratories adopting this technology. The procedure involves a straightforward mixing of the palladium catalyst, ligand, base, and substrates in an aprotic solvent like tetrahydrofuran, followed by the controlled addition of the CO source. Detailed standard operating procedures regarding stoichiometry, addition rates, and workup protocols are essential for reproducibility. For a comprehensive guide on executing this synthesis with optimal results, please refer to the standardized steps outlined below.
- Prepare the catalytic system by mixing palladium chloride, trifurylphosphine ligand, and sodium carbonate base in an organic solvent such as THF.
- Add the three key components: trifluoroethylimidoyl chloride, propargylamine, and the acrylamide derivative into the reaction vessel under inert atmosphere.
- Introduce the carbon monoxide substitute mixture of formic acid and acetic anhydride, then stir the reaction at 30°C for 12 to 20 hours.
- Upon completion, filter the mixture, concentrate, and purify the crude product via silica gel column chromatography to obtain the target biheterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this multi-component synthesis technology offers profound advantages that directly address the pain points of modern chemical procurement and supply chain management. By shifting from multi-step linear syntheses to a convergent one-pot process, manufacturers can significantly reduce the number of isolation and purification stages, which are often the most time-consuming and costly parts of production. This consolidation of steps leads to a drastic simplification of the manufacturing workflow, allowing for faster turnaround times and reduced labor costs. Moreover, the elimination of toxic carbon monoxide gas removes the need for specialized high-pressure reactors and the associated regulatory compliance burdens, thereby lowering the capital investment required for facility upgrades. These factors collectively contribute to substantial cost savings in fine chemical intermediates manufacturing, making high-value biheterocyclic scaffolds more economically accessible for downstream applications.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the use of inexpensive and readily available raw materials, such as propargylamine and simple acrylamides, which are commodity chemicals with stable global supply chains. The replacement of high-pressure CO gas with a liquid formic acid/acetic anhydride system eliminates the logistical costs and safety premiums associated with handling hazardous gases. Additionally, the high reaction efficiency and yields reported in the patent data mean that less raw material is wasted, improving the overall atom economy and reducing the cost of goods sold. The mild conditions also result in lower energy consumption for heating and cooling, further enhancing the operational expenditure profile of the manufacturing process.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on generic, off-the-shelf reagents rather than bespoke, hard-to-source intermediates. Since the key building blocks like trifluoroethylimidoyl chloride can be rapidly synthesized from common amines and acids, the risk of supply disruption due to single-source dependency is minimized. The robustness of the reaction conditions, which tolerate moisture and air better than many sensitive organometallic processes, ensures consistent production quality even in varying manufacturing environments. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, enabling companies to respond more agilely to market demands and clinical trial timelines without compromising on quality standards.
- Scalability and Environmental Compliance: The scalability of this method is evidenced by its successful demonstration at the gram scale, with a clear pathway for expansion to kilogram and ton-level production. The use of tetrahydrofuran, a common industrial solvent, facilitates easy solvent recovery and recycling, aligning with green chemistry principles. By avoiding toxic gases and minimizing waste generation through high-yield transformations, the process simplifies wastewater treatment and exhaust gas management, ensuring compliance with increasingly stringent environmental regulations. This environmental compatibility not only mitigates regulatory risk but also enhances the corporate sustainability profile of manufacturers adopting this technology, making it an attractive option for eco-conscious partners in the global supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed multi-component synthesis. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, aiming to clarify the practical aspects of adopting this methodology. Understanding these details is essential for R&D directors evaluating the feasibility of integrating this route into their existing pipelines and for procurement specialists assessing the long-term viability of the supply source.
Q: What is the source of carbon monoxide in this synthesis?
A: Instead of using toxic high-pressure carbon monoxide gas, this method utilizes a safe in-situ generation system comprising a mixture of formic acid and acetic anhydride, which releases CO under the reaction conditions.
Q: What are the optimal reaction conditions for this transformation?
A: The reaction proceeds efficiently at a mild temperature of 30°C in tetrahydrofuran (THF) solvent, typically requiring 12 to 20 hours to reach completion with high conversion rates.
Q: Does this method tolerate diverse functional groups?
A: Yes, the protocol demonstrates excellent substrate compatibility, tolerating various substituents such as halogens, alkyl groups, alkoxy, nitro, and trifluoromethyl groups on the aromatic rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
As the demand for complex heterocyclic scaffolds continues to surge in the pharmaceutical and agrochemical sectors, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM becomes a strategic imperative. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle sensitive palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards. We understand that consistency is key in drug development, and our robust quality management systems are designed to deliver high-purity pharmaceutical intermediates with minimal batch-to-batch variation.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, identifying opportunities to optimize your supply chain and reduce overall production costs. We encourage you to contact our technical procurement team to request specific COA data for similar compounds and to discuss route feasibility assessments for your proprietary candidates. By choosing NINGBO INNO PHARMCHEM, you gain not just a supplier, but a strategic partner committed to accelerating your path to market through innovation and operational excellence.
