Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
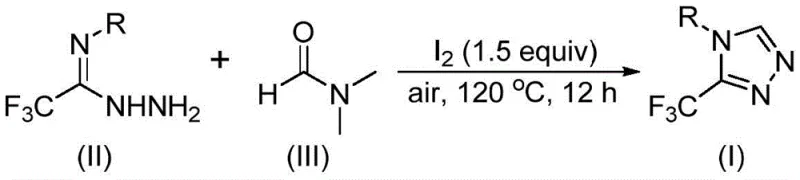
The pharmaceutical and agrochemical industries continuously seek robust methodologies for constructing nitrogen-rich heterocycles, particularly those incorporating fluorine motifs which are known to enhance metabolic stability and bioavailability. Patent CN114920707B introduces a significant advancement in this domain by disclosing a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology addresses critical bottlenecks in traditional heterocycle synthesis by utilizing N,N-dimethylformamide (DMF) not merely as a solvent, but as an active carbon source in an iodine-promoted tandem cyclization. For R&D directors and process chemists, this represents a paradigm shift towards atom-economical and operationally simple transformations that can be seamlessly integrated into the manufacturing of complex bioactive molecules such as Factor IXa inhibitors and GlyT1 inhibitors.
The core innovation lies in the ability to construct the triazole ring and introduce the trifluoromethyl group simultaneously or in a highly convergent manner using readily available starting materials. Unlike legacy processes that often rely on hazardous reagents or multi-step sequences with poor overall yields, this method leverages the ubiquitous solvent DMF to drive the reaction forward. The process operates under mild thermal conditions (110-130°C) and, crucially, does not demand inert gas protection, making it exceptionally attractive for large-scale production environments where simplicity and safety are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with challenges related to reagent availability, reaction complexity, and environmental impact. Traditional routes often necessitate the use of specialized trifluoromethylating agents which can be prohibitively expensive and difficult to handle on a multi-kilogram scale. Furthermore, many existing protocols require stringent anhydrous and anaerobic conditions, demanding specialized equipment like gloveboxes or Schlenk lines that increase capital expenditure and operational overhead. The reliance on transition metal catalysts in some conventional methods also introduces the risk of heavy metal contamination, necessitating costly and time-consuming purification steps to meet the rigorous purity specifications required for pharmaceutical intermediates.
The Novel Approach
The methodology described in patent CN114920707B circumvents these issues by employing a metal-free, iodine-promoted system where DMF acts as the C1 building block. This approach drastically simplifies the reaction setup, allowing it to proceed in an open vessel under air. The use of molecular iodine as a promoter is particularly advantageous due to its low cost and ease of removal compared to precious metal catalysts. By integrating the solvent and reactant roles into a single molecule (DMF), the process reduces the total mass intensity of the reaction and minimizes waste generation. This streamlined workflow not only accelerates the timeline from laboratory discovery to pilot plant validation but also enhances the overall sustainability profile of the manufacturing process.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
From a mechanistic perspective, this transformation is a fascinating example of solvent-participating organic synthesis. The reaction initiates with the activation of DMF by molecular iodine. Depending on the specific pathway, either the formyl group or the N-methyl group of DMF serves as the carbon source. When the formyl group is utilized, it undergoes condensation with the trifluoroethyliminohydrazide to form a hydrazone intermediate, which subsequently cyclizes with the elimination of dimethylamine. Alternatively, when the N-methyl group participates, DMF reacts with iodine to form an amine salt, followed by nucleophilic addition and elimination steps to generate an azadiene intermediate. This intermediate then undergoes intramolecular nucleophilic addition and oxidative aromatization to yield the final 3-trifluoromethyl-1,2,4-triazole scaffold.
Understanding this mechanism is vital for impurity control and process optimization. The dual pathways suggest that reaction conditions, particularly temperature and stoichiometry, must be carefully balanced to favor the desired cyclization over potential side reactions. The patent data indicates that maintaining the reaction temperature between 110°C and 130°C for 10 to 15 hours provides the optimal balance for high conversion rates. The use of 1.5 equivalents of iodine relative to the hydrazide substrate ensures sufficient promotion of the cyclization without leading to excessive iodination of the aromatic rings. This precise control over reaction parameters allows for the consistent production of high-purity intermediates suitable for downstream coupling reactions in API synthesis.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this synthesis effectively, operators must adhere to the specific stoichiometric ratios and thermal profiles outlined in the patent documentation. The process begins with the careful weighing of trifluoroethyliminohydrazide and molecular iodine, which are then suspended in DMF. The mixture is heated to the target temperature range and stirred vigorously to ensure homogeneous heat transfer and mass transfer throughout the reaction bulk. Monitoring the reaction progress via TLC or HPLC is recommended to determine the exact endpoint, although the standard protocol suggests a duration of roughly 12 hours. Following the reaction, the workup procedure involves standard filtration and silica gel treatment, followed by column chromatography to isolate the pure product.
- Prepare the reaction mixture by adding molecular iodine (1.5 equivalents) and trifluoroethyliminohydrazide into an organic solvent system primarily composed of DMF.
- Heat the reaction mixture to a temperature range of 110-130°C under an air atmosphere and maintain stirring for 10 to 15 hours to ensure complete conversion.
- Upon completion, perform post-treatment involving filtration, washing, and silica gel mixing, followed by column chromatography purification to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary driver for cost reduction lies in the substitution of expensive, specialized reagents with commodity chemicals. DMF is one of the most widely produced and cheapest polar aprotic solvents available globally, ensuring a stable and predictable supply chain. By eliminating the need for exotic trifluoromethyl sources or precious metal catalysts, the raw material costs are significantly lowered, directly improving the gross margin of the final intermediate.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the dual function of DMF, which removes the need to purchase separate C1 synthons. Additionally, the absence of transition metals eliminates the expensive downstream processing steps associated with metal scavenging and residual metal testing. This simplification of the purification train reduces solvent consumption and labor hours, leading to substantial operational savings. The use of molecular iodine, a low-cost halogen promoter, further contributes to a favorable cost structure compared to palladium or copper-catalyzed alternatives.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available starting materials. Trifluoroethyliminohydrazide derivatives can be synthesized from common aromatic amines and trifluoroacetic acid, both of which are produced at massive scales by the global chemical industry. This reduces the risk of supply disruptions caused by reliance on niche suppliers. Furthermore, the robustness of the reaction conditions—specifically the tolerance to air and moisture—means that the process is less sensitive to variations in utility quality or operator technique, ensuring consistent batch-to-batch reliability.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the lack of sensitive handling requirements. The ability to run the reaction under air atmosphere removes the engineering complexity and safety hazards associated with large-scale inert gas purging. From an environmental standpoint, the atom economy is improved by incorporating the solvent into the product, thereby reducing the volume of chemical waste generated per kilogram of product. This aligns with modern green chemistry principles and facilitates easier regulatory compliance regarding waste disposal and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the technology's capabilities and limitations for potential partners and licensees.
Q: What is the primary advantage of using DMF in this synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source (C1 synthon), eliminating the need for separate, expensive carbon donors and simplifying the reaction setup significantly.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, one of the key operational advantages of this patented method is that it proceeds efficiently under standard air atmosphere without the need for rigorous anhydrous or oxygen-free environments.
Q: What types of substituents are tolerated on the aromatic ring?
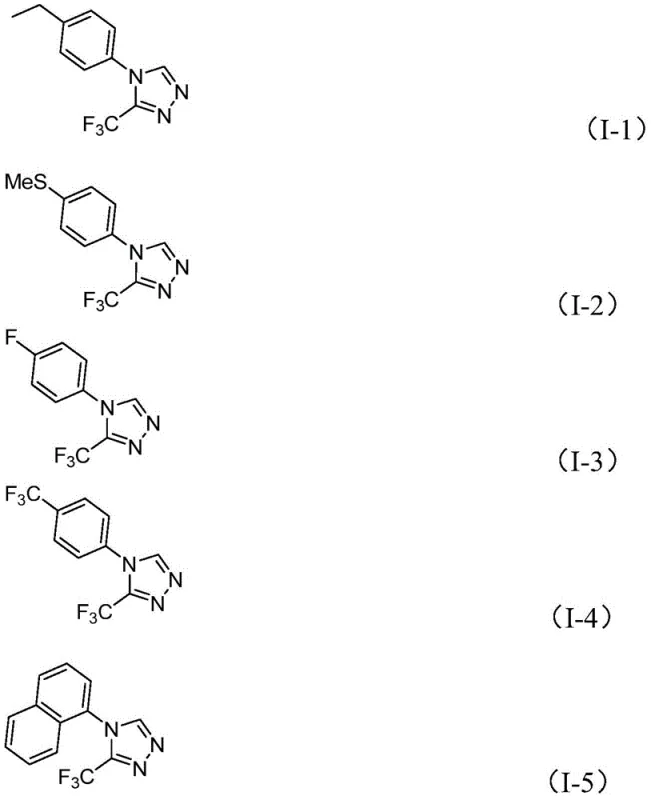
A: The method demonstrates broad substrate scope, successfully accommodating various substituents including alkyl, alkoxy, alkylthio, halogens (fluorine, chlorine), and trifluoromethyl groups at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move smoothly from clinical trials to market launch. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify identity and assay.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fluorine chemistry can accelerate your supply chain and reduce your overall manufacturing costs.
