Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Molybdenum-Copper Co-Catalysis
Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Molybdenum-Copper Co-Catalysis
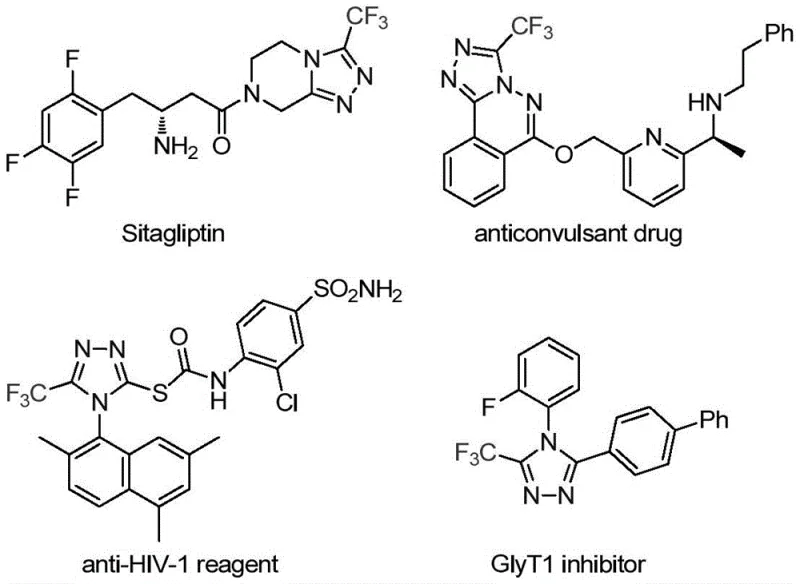
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine atoms which significantly enhance metabolic stability and bioavailability. A groundbreaking development in this sector is detailed in Chinese Patent CN113307778A, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology addresses critical bottlenecks in the synthesis of these valuable scaffolds, which are ubiquitous in modern medicinal chemistry, appearing in blockbuster drugs such as Sitagliptin and various antifungal agents. By leveraging a novel dual-catalyst system involving molybdenum and copper, this process offers a streamlined pathway that bypasses the harsh conditions and safety hazards associated with traditional synthetic routes. For R&D directors and procurement strategists, understanding this methodology is key to securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity building blocks for next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with significant operational challenges and safety concerns that hinder large-scale manufacturing. Traditional literature reports predominantly rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones, processes that often require multiple synthetic steps and generate substantial chemical waste. Furthermore, alternative copper-catalyzed multi-component reactions frequently utilize diazonium salts and trifluorodiazoethane as starting materials; these reagents are notoriously unstable, explosive, and difficult to handle safely in a commercial production environment. The reliance on such hazardous precursors not only escalates the cost of safety infrastructure but also introduces severe supply chain risks, as the availability of these specialized reagents can be inconsistent. Additionally, older methods often suffer from poor atom economy and limited substrate scope, failing to accommodate sensitive functional groups without extensive protection-deprotection sequences.
The Novel Approach
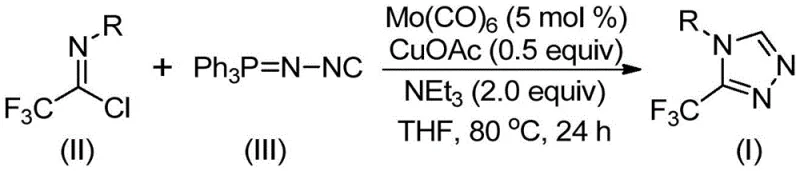
In stark contrast, the methodology described in CN113307778A represents a paradigm shift towards safer and more sustainable chemical manufacturing by utilizing readily available and stable starting materials. This innovative route employs trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as the primary building blocks, eliminating the need for dangerous diazonium species entirely. The reaction proceeds through a direct cycloaddition mechanism co-catalyzed by molybdenum hexacarbonyl and cuprous acetate, operating under remarkably mild thermal conditions ranging from 70°C to 90°C. This gentle thermal profile not only reduces energy consumption but also minimizes the formation of thermal degradation byproducts, thereby simplifying downstream purification. The versatility of this approach allows for the facile introduction of various substituents, including alkyl, alkoxy, and halogen groups, enabling the rapid generation of diverse chemical libraries for drug discovery programs without compromising on yield or purity.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The success of this synthetic strategy lies in the synergistic interaction between the molybdenum and copper catalysts, which orchestrate a complex sequence of bond-forming events with high precision. Mechanistically, the molybdenum hexacarbonyl serves as a crucial activator for the functionalized isonitrile, forming a transient metal-isocyanide complex that enhances the nucleophilicity of the carbon center. Simultaneously, the cuprous acetate facilitates the activation of the trifluoroethylimidoyl chloride, promoting a [3+2] cycloaddition reaction that constructs the five-membered triazole ring intermediate. This cooperative catalysis ensures that the reaction barrier is significantly lowered, allowing the transformation to proceed efficiently at moderate temperatures where traditional thermal cyclizations would fail. Following the ring closure, the system undergoes a spontaneous elimination of triphenylphosphine oxide, driven by the thermodynamic stability of the resulting aromatic triazole core, to deliver the final 3-trifluoromethyl substituted product.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-energy pathways. The concerted nature of the metal-mediated cycloaddition minimizes the generation of free radical species that typically lead to polymerization or non-specific side reactions. Furthermore, the use of triethylamine as a base scavenges the hydrochloric acid byproduct generated during the reaction, preventing acid-catalyzed decomposition of the sensitive imidoyl chloride starting material. The inclusion of molecular sieves in the reaction mixture further aids in maintaining anhydrous conditions, which is critical for preventing the hydrolysis of the imidoyl chloride and ensuring high conversion rates. This rigorous control over the reaction environment results in a clean crude reaction profile, reducing the burden on purification teams and ensuring that the final active pharmaceutical ingredient (API) intermediates meet stringent regulatory specifications for residual metals and organic impurities.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and atmospheric control to maximize the benefits of the patented protocol. The process is designed to be operationally simple, utilizing standard Schlenk techniques or sealed vessel reactors to maintain the necessary inert environment for the metal catalysts. Operators should note that the molar ratio of the functionalized isonitrile to the trifluoroethylimidoyl chloride is preferably maintained at an excess, typically around 1.5 equivalents, to drive the equilibrium towards complete consumption of the more valuable or difficult-to-remove halide species. The reaction is typically conducted in tetrahydrofuran (THF), which provides optimal solubility for all components and stabilizes the catalytic species, although other aprotic solvents like acetonitrile or DMF can be employed depending on specific substrate solubility requirements.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 70-90°C for 18-30 hours, then filter and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Mo/Cu co-catalyzed methodology translates into tangible strategic advantages regarding cost stability and sourcing reliability. The shift away from exotic or hazardous reagents like diazonium salts to commodity chemicals such as trifluoroethylimidoyl chloride and isonitriles significantly de-risks the supply chain, as these materials are produced by multiple global vendors with established logistics networks. This diversification of the supplier base prevents single-source bottlenecks and ensures business continuity even during periods of market volatility. Moreover, the use of earth-abundant transition metals like copper and molybdenum, rather than expensive precious metals like palladium or rhodium, drastically reduces the raw material cost basis, allowing for more competitive pricing models in long-term supply agreements.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the elimination of costly purification steps associated with removing heavy metal residues from precious metal catalysts. Since copper and molybdenum are less regulated and easier to remove to ppm levels compared to noble metals, the downstream processing costs are significantly lowered, directly improving the gross margin for the final intermediate. Additionally, the high reaction efficiency and yields reported in the patent, often exceeding 90% for optimized substrates, mean that less raw material is wasted, further enhancing the overall cost-effectiveness of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more predictable production schedule, as the process is less susceptible to batch-to-batch variability caused by sensitive reagents. The ability to run the reaction at moderate temperatures (70-90°C) reduces the strain on heating and cooling infrastructure, allowing for higher throughput in existing reactor trains without requiring capital-intensive upgrades. This operational flexibility ensures that delivery timelines can be met consistently, a critical factor for pharmaceutical clients managing tight clinical trial schedules or commercial launch windows.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) standpoint, this method aligns perfectly with green chemistry principles by avoiding the generation of toxic nitrogen gas byproducts typical of diazonium chemistry. The simplified workup procedure, which involves basic filtration and chromatography, reduces the volume of solvent waste and aqueous effluent generated per kilogram of product. This reduced environmental footprint not only lowers waste disposal costs but also facilitates smoother regulatory approvals in jurisdictions with strict environmental compliance mandates, making the supply chain more resilient to regulatory changes.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the scope and limitations of the patented synthesis. These insights are derived directly from the experimental data and embodiments provided in the intellectual property documentation, ensuring accuracy and relevance for process development scientists. Understanding these nuances is essential for designing effective synthetic routes that leverage the full potential of this catalytic system while mitigating potential risks associated with scale-up.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies reacting at 70-90°C for 18-30 hours in THF solvent, utilizing a catalytic system of Mo(CO)6 and CuOAc with triethylamine as the base.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates broad substrate tolerance, successfully synthesizing derivatives with methyl, methoxy, fluoro, chloro, and nitro substituents on the aromatic ring.
Q: Is the purification process complex for large-scale production?
A: The post-treatment is straightforward, involving simple filtration followed by standard silica gel column chromatography, making it suitable for scale-up operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality heterocyclic intermediates play in the development of life-saving medications, and we are committed to bridging the gap between academic innovation and industrial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results observed in the laboratory can be seamlessly translated to manufacturing scales. We operate state-of-the-art rigorous QC labs equipped with advanced analytical instrumentation to guarantee stringent purity specifications for every batch of 3-trifluoromethyl-1,2,4-triazole we produce, giving our partners confidence in the consistency and quality of their supply.
We invite forward-thinking organizations to collaborate with us to explore how this advanced catalytic technology can optimize their specific manufacturing workflows. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current bill of materials, alongside specific COA data and route feasibility assessments for your target molecules. Let us partner with you to accelerate your drug development timeline while achieving superior economic and operational outcomes through the power of cutting-edge chemical synthesis.
