Revolutionizing Fluorinated Triazole Production: A Cost-Effective DMF-Based Route for Pharmaceutical Intermediates
Revolutionizing Fluorinated Triazole Production: A Cost-Effective DMF-Based Route for Pharmaceutical Intermediates
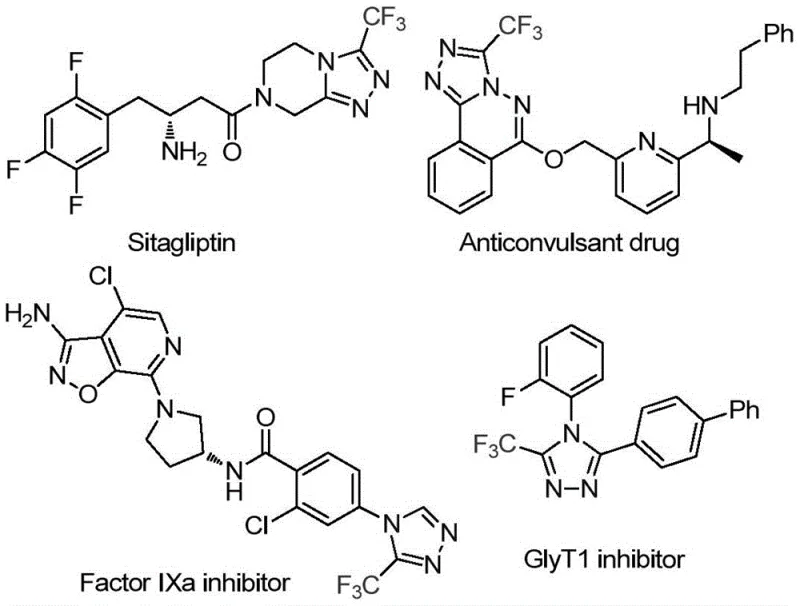
In the rapidly evolving landscape of pharmaceutical process development, the efficient construction of fluorinated heterocycles remains a critical bottleneck for many reliable pharmaceutical intermediate supplier networks. Patent CN114920707B, published in April 2023, introduces a transformative methodology for synthesizing 3-trifluoromethyl substituted 1,2,4-triazole compounds, a structural motif pervasive in high-value bioactive molecules. As illustrated in the initial structural survey, this scaffold is integral to potent therapeutics such as Sitagliptin, Factor IXa inhibitors, and GlyT1 inhibitors, where the trifluoromethyl group enhances metabolic stability and lipophilicity. The disclosed invention leverages a unique tandem cyclization strategy where the ubiquitous solvent N,N-dimethylformamide (DMF) acts not merely as a medium but as an active carbon source. This dual functionality represents a paradigm shift in green chemistry, offering a streamlined pathway that bypasses the need for specialized C1 donors, thereby addressing key pain points regarding cost reduction in API manufacturing and operational simplicity for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of 1,2,4-triazole rings bearing trifluoromethyl groups has relied on multi-step sequences involving harsh reaction conditions and expensive reagents. Conventional routes often necessitate the use of pre-functionalized hydrazines or nitriles, requiring stringent anhydrous and anaerobic environments to prevent side reactions and ensure high yields. These legacy methods frequently employ transition metal catalysts that are not only costly but also introduce significant challenges in downstream purification, particularly concerning residual metal limits in final drug substances. Furthermore, the requirement for specialized carbon sources adds layers of complexity to the supply chain, increasing lead times and inventory costs. The cumulative effect of these factors is a manufacturing process that is fragile, capital-intensive, and difficult to scale without compromising the purity profiles required by regulatory bodies for high-purity heterocyclic building blocks.
The Novel Approach
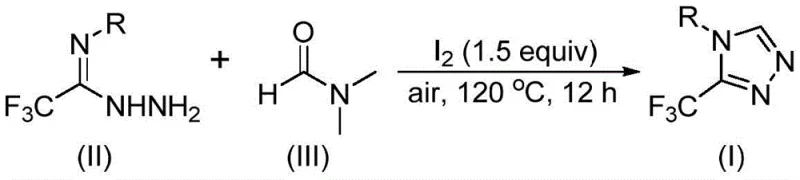
The methodology described in CN114920707B fundamentally disrupts this status quo by utilizing molecular iodine as a promoter and DMF as the sole carbon source in an open-air system. As depicted in the general reaction scheme, the process merges trifluoroethyliminohydrazide with DMF under thermal conditions to directly forge the triazole ring. This approach eliminates the dependency on inert atmospheres, allowing reactions to proceed efficiently in the presence of oxygen and moisture, which drastically reduces the engineering controls needed for production. The use of DMF, a commodity chemical available in bulk quantities globally, ensures that the raw material costs are minimized while maximizing atom economy. By integrating the solvent and reactant roles, the process inherently simplifies the workup procedure, removing the need for complex extraction steps to remove excess carbon donors, thus facilitating a more robust and scalable manufacturing protocol suitable for commercial scale-up of complex fluorinated intermediates.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic elegance of this transformation lies in the activation of DMF by molecular iodine, which generates reactive intermediates capable of engaging with the hydrazide substrate. Detailed analysis suggests a dual-pathway mechanism where either the formyl group or the N-methyl group of DMF can serve as the carbon donor. In the formyl pathway, condensation with the hydrazide forms a hydrazone intermediate, which subsequently undergoes intramolecular cyclization with the elimination of dimethylamine to yield the target triazole. Alternatively, the N-methyl pathway involves the formation of an amine salt via iodine activation, followed by nucleophilic addition and oxidative aromatization. This mechanistic flexibility ensures high conversion rates across a diverse range of substrates, as the system can adapt to the electronic properties of the starting materials. The involvement of iodine as a mild oxidant and activator avoids the use of heavy metals, aligning with modern sustainability goals while maintaining high reaction efficiency at temperatures between 110°C and 130°C.
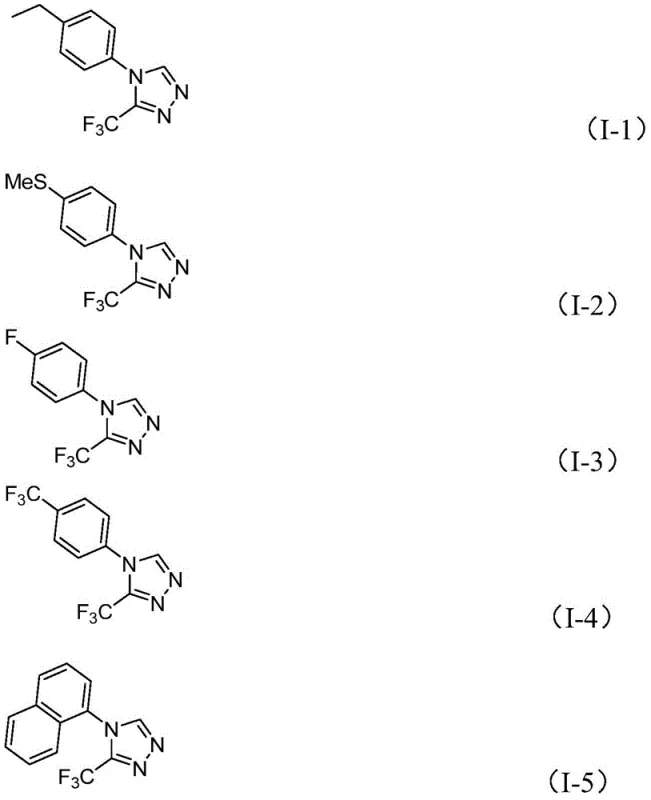
From an impurity control perspective, this mechanism offers distinct advantages for R&D teams focused on quality by design. The absence of transition metals eliminates a major class of genotoxic impurities, simplifying the purification strategy to standard silica gel chromatography or crystallization. The reaction tolerance is remarkably broad, accommodating various substituents on the aromatic ring of the hydrazide precursor. As shown in the substrate scope examples, electron-rich groups like methoxy and methylthio, as well as electron-deficient groups like fluoro and chloro, are all compatible with the reaction conditions. This versatility allows medicinal chemists to rapidly generate libraries of analogues for structure-activity relationship (SAR) studies without needing to re-optimize reaction conditions for each new derivative, thereby accelerating the drug discovery timeline and reducing lead time for high-purity intermediates.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and safety. The process is designed to be operationally simple, utilizing standard glassware and heating equipment without the need for gloveboxes or Schlenk lines. The key to success lies in the precise stoichiometric balance between the hydrazide substrate and the iodine promoter, typically maintained at a ratio of 1:1.5, ensuring complete activation of the DMF solvent. Following the reaction period of 10 to 15 hours, the crude mixture can be processed through straightforward filtration and purification techniques. For detailed operational protocols and safety guidelines, please refer to the standardized synthesis steps provided below.
- Combine trifluoroethyliminohydrazide and molecular iodine in DMF solvent within a reaction vessel.
- Heat the mixture to 110-130°C under an air atmosphere for 10-15 hours to facilitate cyclization.
- Perform post-treatment including filtration and column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this DMF-based synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary driver of value is the drastic simplification of the raw material portfolio. By replacing specialized carbon donors with DMF, a solvent that is already present in almost every chemical facility for cleaning and processing, companies can significantly reduce inventory complexity and purchasing overhead. This consolidation of materials leads to substantial cost savings in logistics and storage, as there is no longer a need to source and manage hazardous or expensive C1 reagents. Furthermore, the elimination of inert gas requirements reduces the consumption of nitrogen or argon, lowering utility costs and freeing up reactor capacity that was previously tied up in purging cycles.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the removal of costly catalysts and the utilization of commodity solvents as reactants. Traditional methods often rely on precious metal catalysts or exotic reagents that drive up the Cost of Goods Sold (COGS). In contrast, this iodine-promoted method uses inexpensive, non-toxic promoters and avoids the generation of heavy metal waste, which in turn reduces waste disposal fees. The streamlined workflow also decreases labor hours per batch, as operators spend less time setting up complex anhydrous conditions and more time on productive throughput. These factors combine to create a leaner manufacturing process that improves overall margin potential for high-volume API production.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available starting materials. DMF and molecular iodine are produced on a massive industrial scale globally, meaning that supply disruptions are highly unlikely compared to niche reagents used in conventional triazole synthesis. This availability ensures consistent production schedules and minimizes the risk of stockouts that could delay clinical trials or commercial launches. Additionally, the robustness of the reaction under air atmosphere means that manufacturing can be transferred between different sites or contract manufacturing organizations (CMOs) with minimal technology transfer friction, as the process does not require specialized infrastructure that might be absent in certain regions.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the benign nature of the reagents and the absence of exothermic hazards associated with strong oxidizers or pyrophoric catalysts. The reaction operates at moderate temperatures that are easily managed in standard jacketed reactors, reducing the energy footprint of the process. From an environmental standpoint, the method aligns with green chemistry principles by improving atom economy and reducing the E-factor (mass of waste per mass of product). The lack of heavy metal residues simplifies wastewater treatment and ensures compliance with increasingly stringent environmental regulations, avoiding potential fines and reputational damage associated with toxic effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and claims within CN114920707B, providing clarity for technical teams evaluating this route for their specific projects. Understanding these nuances is essential for effective process integration and risk assessment.
Q: What is the primary advantage of using DMF in this synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source (C1 synthon), eliminating the need for separate, expensive carbon donors and simplifying the workup process significantly.
Q: Does this reaction require strict anhydrous or anaerobic conditions?
A: No, one of the key innovations of patent CN114920707B is that the reaction proceeds efficiently under standard air atmosphere without the need for rigorous drying or inert gas protection, reducing operational complexity.
Q: What is the substrate scope for the R group in this triazole synthesis?
A: The method demonstrates broad compatibility with various substituted aryl groups, including those with electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups like halogens and trifluoromethyl.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient fluorination technologies in modern drug development. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative academic discoveries like the DMF-based triazole synthesis are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of intermediate meets the exacting standards required by global regulatory agencies. Our commitment to quality ensures that your supply chain remains uninterrupted and compliant.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your development timeline and your bottom line.
