Advanced Palladium-Catalyzed Synthesis of Bioactive 1,5-Dihydro-2H-Pyrrole-2-One Scaffolds
Advanced Palladium-Catalyzed Synthesis of Bioactive 1,5-Dihydro-2H-Pyrrole-2-One Scaffolds
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, safer, and cost-effective synthetic routes to complex heterocyclic cores. A significant breakthrough in this domain is detailed in patent CN112694430B, which discloses a novel preparation method for 1,5-dihydro-2H-pyrrole-2-one compounds. This structural motif is not merely an academic curiosity; it serves as the critical pharmacophore in a wide array of bioactive molecules with profound therapeutic potential. For instance, the natural product Althiomycin exhibits significant antibacterial activity, while Glimepiride is a widely prescribed hypoglycemic agent for diabetes management, and Isomalyngamide A has demonstrated potential anticancer properties. The ability to construct this privileged scaffold efficiently is therefore of paramount importance to R&D teams aiming to access these high-value targets.
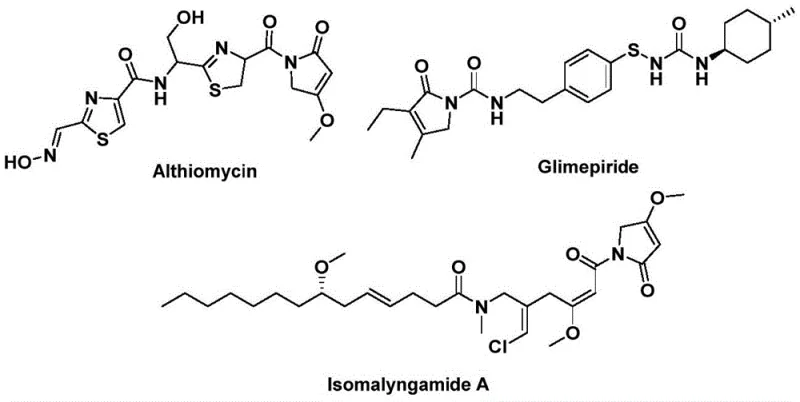
Traditionally, the synthesis of such lactam structures often involves multi-step sequences that can be cumbersome and low-yielding. However, the methodology outlined in CN112694430B leverages a sophisticated palladium-catalyzed bis-carbonylation strategy. By utilizing readily available starting materials such as propargyl amines and benzyl chlorides, coupled with a solid carbon monoxide substitute, this process achieves the construction of the five-membered lactam ring in a single operational step. This represents a paradigm shift for procurement managers and supply chain heads who are constantly seeking to reduce the number of unit operations and minimize the inventory of hazardous gases like carbon monoxide in their facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,5-dihydro-2H-pyrrol-2-one skeletons has relied on classical condensation reactions or multi-step functional group manipulations that often suffer from poor atom economy and harsh reaction conditions. Conventional carbonylation methods, while effective, frequently require the direct use of high-pressure carbon monoxide gas, which poses severe safety risks and necessitates specialized high-pressure equipment that many standard pilot plants lack. Furthermore, traditional routes often struggle with substrate scope, failing to tolerate sensitive functional groups such as halogens or trifluoromethyl moieties without extensive protection-deprotection strategies. These limitations result in prolonged development timelines, increased waste generation, and ultimately, higher costs per kilogram of the final active pharmaceutical ingredient (API) intermediate, creating bottlenecks for reliable pharmaceutical intermediate suppliers trying to scale up production.
The Novel Approach
In stark contrast, the novel approach described in the patent utilizes a palladium-catalyzed bis-carbonylation cascade that elegantly solves these logistical and chemical challenges. By employing 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid CO surrogate, the process completely bypasses the need for gaseous CO handling, thereby drastically simplifying the engineering requirements and enhancing workplace safety. The reaction proceeds smoothly in acetonitrile at moderate temperatures (100-120°C), demonstrating exceptional functional group tolerance across a wide range of electronic environments. This one-pot transformation directly converts simple propargyl amines and benzyl chlorides into the target lactams with high efficiency, effectively collapsing what would traditionally be a multi-step synthesis into a single, streamlined operation. This innovation not only accelerates the speed of discovery for medicinal chemists but also offers a robust platform for cost reduction in API manufacturing by minimizing raw material consumption and waste disposal costs.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors evaluating its feasibility for large-scale production. The catalytic cycle initiates with the oxidative addition of the palladium(0) species, generated in situ from palladium acetate and the DPPF ligand, into the carbon-chlorine bond of the benzyl chloride substrate. This forms a reactive benzyl-palladium intermediate, which is then intercepted by carbon monoxide liberated from the thermal decomposition of the TFBen additive. The insertion of the first CO molecule generates an acyl-palladium species, setting the stage for the subsequent cyclization event.
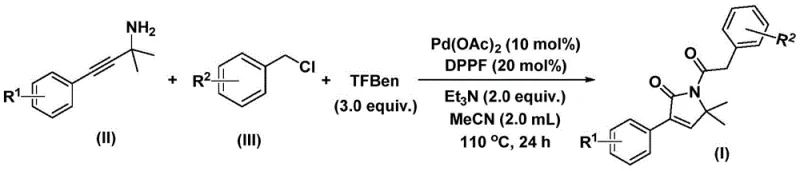
Following the initial carbonylation, the nucleophilic nitrogen of the propargyl amine attacks the acyl-palladium intermediate, leading to the formation of a five-membered ring palladium species. Crucially, the mechanism involves a second insertion of carbon monoxide into this cyclic intermediate, expanding the coordination sphere before a final reductive elimination step releases the 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active palladium catalyst. This dual-carbonylation pathway is highly selective, minimizing the formation of linear amide byproducts or polymerization side reactions that often plague alkyne carbonylations. The use of the bidentate phosphine ligand DPPF is instrumental in stabilizing the palladium center throughout this complex cycle, ensuring high turnover numbers and consistent batch-to-batch reproducibility, which are key metrics for assessing the purity and杂质 profile of the final product.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
The operational simplicity of this protocol makes it highly attractive for immediate adoption in process development laboratories. The procedure typically involves charging a reaction vessel with the palladium catalyst, ligand, base, CO source, and substrates in acetonitrile, followed by heating to 110°C for 24 hours. The detailed standardized synthesis steps, including precise stoichiometric ratios and workup procedures, are provided in the guide below to ensure successful replication of the high yields reported in the patent literature.
- Combine palladium acetate, DPPF ligand, triethylamine, TFBen (CO source), propargyl amine, and benzyl chloride in acetonitrile solvent.
- Heat the reaction mixture to 110°C and stir for 24 hours under inert atmosphere to facilitate bis-carbonylation and cyclization.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 1,5-dihydro-2H-pyrrole-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the substantial simplification of the supply chain for raw materials; benzyl chlorides and propargyl amines are commodity chemicals available from multiple global vendors, reducing the risk of single-source dependency. Furthermore, the elimination of gaseous carbon monoxide removes a significant regulatory and safety burden, allowing for production in facilities that may not be equipped for high-pressure gas handling, thus expanding the pool of eligible contract manufacturing organizations (CMOs).
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the convergence of several factors, primarily the reduction in step count and the use of inexpensive reagents. By achieving the target structure in a single pot, manufacturers eliminate the solvent usage, energy consumption, and labor costs associated with isolating intermediates in multi-step sequences. Additionally, the use of a solid CO source avoids the capital expenditure required for specialized gas containment infrastructure. While specific percentage savings depend on local utility costs, the qualitative reduction in processing time and waste treatment volume translates directly to a lower cost of goods sold (COGS), making the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply continuity. The protocol operates at atmospheric pressure with standard heating, minimizing the risk of equipment failure or batch loss due to pressure excursions. The broad substrate scope means that if a specific substituted benzyl chloride becomes unavailable, alternative analogs can often be sourced or synthesized without re-optimizing the entire process. This flexibility ensures that production schedules remain intact even when facing raw material volatility, providing a stable supply of high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: From an environmental perspective, the atom economy of this bis-carbonylation is superior to traditional methods that might generate stoichiometric amounts of heavy metal waste or toxic byproducts. The reaction produces minimal waste streams, primarily consisting of the spent phenol byproduct from the CO source, which is easier to manage than gaseous emissions. The scalability is further enhanced by the homogeneous nature of the catalytic system, which allows for efficient heat and mass transfer in larger reactors. This alignment with green chemistry principles facilitates smoother regulatory approvals and reduces the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation of this technology, we have compiled a set of questions based on the specific experimental data and scope defined in the patent documentation. These answers reflect the proven capabilities of the method across various substituted derivatives, ensuring clarity for technical teams evaluating the route for their specific projects.
Q: What is the carbon monoxide source used in this synthesis?
A: The process utilizes 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide substitute, which eliminates the need for handling hazardous gaseous CO directly.
Q: What is the typical reaction temperature and time for this transformation?
A: Optimal reaction conditions involve heating the mixture to 110°C for approximately 24 hours to ensure complete conversion and high yield.
Q: Does this method tolerate diverse functional groups on the substrates?
A: Yes, the protocol demonstrates excellent substrate compatibility, tolerating substituents such as methoxy, fluoro, chloro, bromo, trifluoromethyl, and methyl groups on both the amine and benzyl chloride components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed bis-carbonylation technology for the synthesis of complex heterocyclic intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped to handle sensitive catalytic processes with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to guarantee the quality of every batch delivered.
We invite you to leverage our technical expertise to optimize your supply chain for these critical building blocks. Whether you require custom synthesis of specific derivatives or scale-up of the standard protocol, our team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of high-quality intermediates for your next generation of therapeutics.
