Scalable Palladium-Catalyzed Bis-Carbonylation for High-Purity 1,5-Dihydro-2H-Pyrrole-2-One Intermediates
Introduction to Next-Generation Pyrrolone Synthesis
The structural motif of 1,5-dihydro-2H-pyrrole-2-one represents a privileged scaffold in modern medicinal chemistry, serving as the core backbone for a diverse array of bioactive natural products and synthetic pharmaceuticals. As highlighted in recent patent literature, specifically CN112694430B, the demand for efficient access to these heterocycles is driven by their presence in potent antibacterial agents like althiomycin, effective hypoglycemic drugs such as glimepiride, and anticancer candidates like isomalyngamide A. 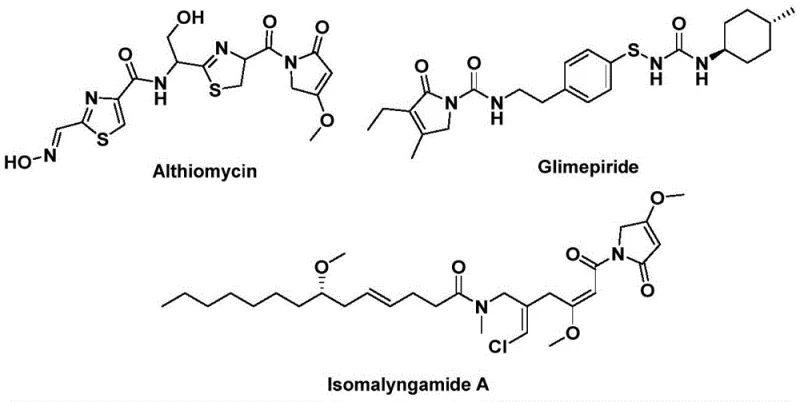 . Traditional synthetic routes to these valuable intermediates often suffer from multi-step sequences, harsh reaction conditions, or poor atom economy, creating bottlenecks for both research and commercial production. The disclosed technology introduces a transformative palladium-catalyzed bis-carbonylation strategy that streamlines the construction of this five-membered lactam ring directly from readily available propargylamines and benzyl chlorides. This report analyzes the technical merits of this innovation, evaluating its potential to redefine supply chains for reliable pharmaceutical intermediate suppliers seeking robust, scalable, and cost-effective manufacturing solutions.
. Traditional synthetic routes to these valuable intermediates often suffer from multi-step sequences, harsh reaction conditions, or poor atom economy, creating bottlenecks for both research and commercial production. The disclosed technology introduces a transformative palladium-catalyzed bis-carbonylation strategy that streamlines the construction of this five-membered lactam ring directly from readily available propargylamines and benzyl chlorides. This report analyzes the technical merits of this innovation, evaluating its potential to redefine supply chains for reliable pharmaceutical intermediate suppliers seeking robust, scalable, and cost-effective manufacturing solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,5-dihydro-2H-pyrrol-2-one skeleton has relied on classical condensation reactions or cyclization strategies that frequently necessitate pre-functionalized starting materials and stoichiometric activating agents. Conventional carbonylation approaches, while direct, have traditionally been plagued by the requirement for high-pressure gaseous carbon monoxide, posing significant safety hazards and requiring specialized high-pressure reactor infrastructure that limits accessibility for many contract manufacturing organizations. Furthermore, existing methods often exhibit limited substrate compatibility, struggling to tolerate sensitive functional groups such as halogens or trifluoromethyl moieties without extensive protection-deprotection sequences. These inefficiencies result in prolonged lead times, increased waste generation, and higher overall production costs, making the commercial scale-up of complex pharmaceutical intermediates challenging for procurement teams aiming to optimize their supply chain resilience.
The Novel Approach
In stark contrast, the novel methodology described in the patent leverages a sophisticated palladium-catalyzed dual carbonylation cascade that operates under remarkably mild and operationally simple conditions. By utilizing a solid carbon monoxide substitute, specifically 1,3,5-tricarboxylic acid phenol ester (TFBen), the process completely circumvents the safety risks and engineering complexities associated with handling toxic CO gas. The reaction proceeds efficiently in acetonitrile at moderate temperatures of 110°C, utilizing inexpensive benzyl chlorides and propargylamines as feedstocks. 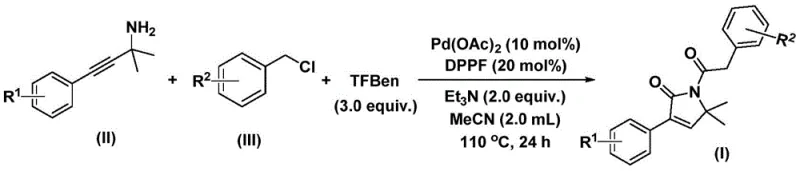 . This one-pot transformation not only simplifies the operational workflow but also demonstrates exceptional functional group tolerance, successfully incorporating diverse substituents including methoxy, fluoro, chloro, bromo, and trifluoromethyl groups with high yields ranging from 70% to 92%. This represents a paradigm shift towards greener, safer, and more economically viable manufacturing protocols for high-purity OLED material and API precursors.
. This one-pot transformation not only simplifies the operational workflow but also demonstrates exceptional functional group tolerance, successfully incorporating diverse substituents including methoxy, fluoro, chloro, bromo, and trifluoromethyl groups with high yields ranging from 70% to 92%. This represents a paradigm shift towards greener, safer, and more economically viable manufacturing protocols for high-purity OLED material and API precursors.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The efficacy of this synthetic route is underpinned by a well-defined catalytic cycle initiated by the oxidative addition of the benzyl chloride substrate to the active palladium(0) species generated in situ from palladium acetate and the DPPF ligand. This step forms a crucial benzyl-palladium intermediate, which subsequently undergoes migratory insertion of carbon monoxide liberated from the phenol ester surrogate to generate an acyl-palladium species. The mechanistic elegance continues as the propargylamine component coordinates and inserts into the acyl-palladium bond, triggering an intramolecular cyclization event that constructs the five-membered nitrogen-containing ring. A second molecule of carbon monoxide is then inserted into the resulting organopalladium intermediate, expanding the coordination sphere before final reductive elimination releases the desired 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active catalyst. This intricate dance of bond formations ensures high regioselectivity and minimizes the formation of side products, a critical factor for R&D directors focused on impurity profile control.
From an impurity control perspective, the use of a solid CO source provides a steady, controlled release of carbon monoxide, preventing the local concentration spikes that often lead to oligomerization or alternative carbonylation pathways in gas-phase reactions. The choice of the bidentate phosphine ligand DPPF stabilizes the palladium center throughout the multiple insertion steps, preventing catalyst decomposition and ensuring consistent turnover numbers. Furthermore, the reaction conditions are sufficiently robust to tolerate minor variations in reagent quality, reducing the burden on raw material qualification. The post-reaction workup, involving simple filtration and silica gel treatment, effectively removes palladium residues and organic byproducts, facilitating the isolation of the target compound with the stringent purity specifications required for downstream pharmaceutical applications. This level of mechanistic control translates directly into batch-to-batch consistency, a key metric for supply chain reliability.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
The implementation of this synthesis protocol is designed for seamless integration into existing laboratory and pilot plant workflows, requiring only standard heating and stirring equipment without the need for specialized high-pressure autoclaves. The procedure begins with the precise charging of the palladium catalyst system, the solid CO surrogate, and the amine and chloride substrates into a reaction vessel containing acetonitrile, followed by heating to 110°C for a duration of 24 hours. This standardized approach minimizes operator variability and ensures reproducible outcomes across different scales of production. For detailed operational parameters and specific stoichiometric ratios optimized for various substrates, please refer to the standardized synthesis guide below.
- Charge a reaction vessel with palladium acetate catalyst, DPPF ligand, solid carbon monoxide substitute (TFBen), triethylamine base, propargylamine substrate, and benzyl chloride derivative in acetonitrile solvent.
- Heat the reaction mixture to 110°C and maintain stirring for 24 hours under inert atmosphere to facilitate the bis-carbonylation and cyclization cascade.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 1,5-dihydro-2H-pyrrole-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers tangible strategic advantages that extend beyond mere chemical novelty. The shift from hazardous gaseous reagents to stable solid surrogates drastically reduces the capital expenditure required for facility upgrades, allowing for faster deployment of production lines. Moreover, the reliance on commodity chemicals like benzyl chlorides and propargylamines ensures a stable and diversified supply base, mitigating the risks associated with single-source dependencies on exotic reagents. The high reaction efficiency and simplified purification process contribute to a significantly reduced cost of goods sold (COGS), enabling more competitive pricing strategies in the global marketplace.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide infrastructure results in substantial capital savings and lower operational maintenance costs, while the use of cheap, commercially available starting materials drives down raw material expenses. The high yields observed across a broad substrate scope mean less waste and higher throughput per batch, directly improving the economic viability of large-scale production runs without compromising quality standards.
- Enhanced Supply Chain Reliability: By utilizing widely sourced benzyl chlorides and avoiding specialized gas supplies, manufacturers can secure a more resilient supply chain that is less susceptible to logistical disruptions. The robustness of the reaction conditions allows for flexible scheduling and rapid scale-up from gram to kilogram quantities, ensuring that critical project timelines are met consistently even in volatile market conditions.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional multi-step syntheses, aligning with increasingly strict environmental regulations and sustainability goals. The straightforward workup procedure reduces solvent consumption and energy usage during purification, supporting green chemistry initiatives while maintaining the high purity levels necessary for regulatory approval in pharmaceutical applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed carbonylation technology. These insights are derived directly from the experimental data and scope limitations defined in the patent documentation, providing a realistic overview of the process capabilities for potential partners and licensees.
Q: Does this synthesis require hazardous gaseous carbon monoxide?
A: No, the process utilizes a solid carbon monoxide substitute (1,3,5-tricarboxylic acid phenol ester), which significantly enhances operational safety and eliminates the need for high-pressure CO gas infrastructure.
Q: What is the substrate scope for the benzyl chloride component?
A: The method demonstrates excellent functional group tolerance, accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as halogens (F, Cl, Br) and trifluoromethyl moieties.
Q: How does the purification process impact scalability?
A: The post-treatment involves simple filtration followed by standard silica gel column chromatography, avoiding complex crystallization steps or difficult heavy metal scavenging procedures often associated with palladium catalysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development pipelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging cutting-edge technologies like the palladium-catalyzed bis-carbonylation described herein, we empower our clients to bring life-saving therapies to market faster and more cost-effectively.
We invite you to collaborate with us to explore how this innovative synthesis route can be tailored to your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis, specific COA data, and comprehensive route feasibility assessments. Let us be your partner in navigating the complexities of modern chemical manufacturing and securing a competitive edge in the global pharmaceutical landscape.
