Scalable Palladium-Catalyzed Synthesis of Bioactive 1,5-Dihydro-2H-Pyrrole-2-One Scaffolds for Global Pharma
Introduction to Next-Generation Pyrrolone Synthesis
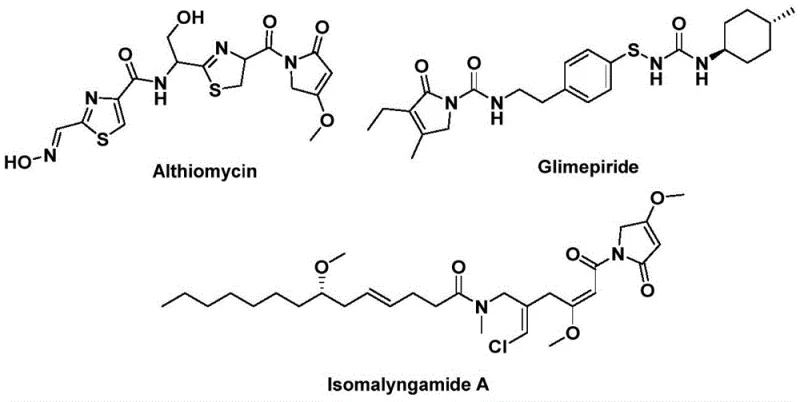
The 1,5-dihydro-2H-pyrrole-2-one structural motif serves as a critical pharmacophore in a diverse array of bioactive natural products and therapeutic agents, ranging from the potent antibacterial agent Althiomycin to the antidiabetic drug Glimepiride and the anticancer candidate Isomalyngamide A. Despite its biological significance, the efficient construction of this five-membered lactam ring has historically presented substantial synthetic challenges, often requiring multi-step sequences or hazardous reagents. Addressing these industrial pain points, the recent technological disclosure in patent CN112694430B introduces a transformative palladium-catalyzed bis-carbonylation strategy. This novel methodology leverages readily available propargyl amines and benzyl chlorides to construct the core scaffold in a single operational step, marking a significant leap forward for reliable pharmaceutical intermediate suppliers seeking to optimize their production pipelines. By replacing gaseous carbon monoxide with a solid surrogate, this process not only enhances safety profiles but also streamlines the manufacturing workflow, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of carbonyl-containing heterocycles like pyrrolones has relied heavily on direct carbonylation reactions utilizing carbon monoxide gas. While chemically effective, this approach imposes severe logistical and safety constraints on large-scale manufacturing facilities. The requirement for high-pressure autoclaves to handle toxic CO gas necessitates expensive infrastructure investments and rigorous safety protocols, which inherently drive up the capital expenditure (CAPEX) and operational expenditure (OPEX) for producers. Furthermore, the handling of gaseous reagents often leads to mass transfer limitations, resulting in inconsistent reaction rates and potential batch-to-batch variability. For procurement teams, this translates to higher costs and longer lead times, as specialized equipment and certified personnel are mandatory. Additionally, conventional routes often suffer from poor atom economy or require pre-functionalized starting materials that are costly and difficult to source globally, creating bottlenecks in the supply chain reliability for key drug intermediates.
The Novel Approach
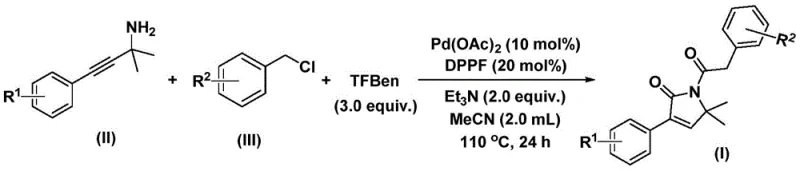
The innovative process detailed in CN112694430B circumvents these traditional hurdles by employing a solid carbon monoxide substitute, specifically 1,3,5-tricarboxylic acid phenol ester (TFBen), within a palladium-catalyzed system. This shift from gas to solid reagents fundamentally alters the risk profile of the synthesis, allowing the reaction to proceed in standard glassware or stainless steel reactors without the need for high-pressure containment. The reaction operates efficiently at moderate temperatures of 110 °C in acetonitrile, utilizing a robust catalyst system comprising palladium acetate and DPPP ligand. This one-pot transformation directly couples propargyl amines with benzyl chlorides, achieving high conversion rates and excellent yields, such as the 92% isolated yield observed in optimized examples. For cost reduction in API manufacturing, this method is unparalleled; it eliminates the need for CO gas infrastructure, reduces purification steps due to high selectivity, and utilizes cheap, commodity-grade starting materials that are universally available, thereby securing a more resilient and cost-effective supply chain for downstream drug manufacturers.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The mechanistic elegance of this transformation lies in its sequential insertion steps, which are meticulously controlled by the palladium catalyst to ensure regioselectivity and high yield. The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzyl-palladium intermediate. This step is crucial as it activates the electrophile for subsequent carbonylation. Following this activation, the solid CO substitute (TFBen) thermally decomposes to release carbon monoxide in situ, which then inserts into the benzyl-palladium bond to form an acyl-palladium species. This acyl intermediate subsequently undergoes nucleophilic attack by the propargyl amine, triggering a cyclization event that forms the five-membered ring palladium intermediate. The process continues with a second carbon monoxide insertion, expanding the coordination sphere before final reductive elimination releases the desired 1,5-dihydro-2H-pyrrole-2-one product and regenerates the active palladium catalyst. This intricate dance of insertion and elimination ensures that the carbonyl groups are positioned precisely within the lactam ring, minimizing the formation of regioisomers.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-temperature thermal cyclizations. The well-defined organometallic pathway suppresses side reactions such as polymerization of the alkyne or homocoupling of the benzyl chloride, which are common pitfalls in less controlled environments. The use of triethylamine as a base further aids in scavenging the hydrochloric acid byproduct generated during the oxidative addition, preventing acid-catalyzed degradation of the sensitive lactam product. Consequently, the crude reaction mixture is remarkably clean, often requiring only simple filtration and silica gel treatment followed by column chromatography to achieve analytical purity. This high level of chemoselectivity is vital for R&D directors who must ensure that the impurity profile of the intermediate meets stringent regulatory standards for pharmaceutical applications, reducing the burden on downstream purification units.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise adherence to the stoichiometric ratios and thermal conditions outlined in the patent to maximize efficiency. The protocol dictates the use of 10 mol% palladium acetate and 20 mol% DPPP ligand relative to the limiting reagent, ensuring sufficient catalytic turnover without excessive metal loading that could complicate downstream metal removal. The reaction is typically conducted in acetonitrile (MeCN) at a concentration that allows for effective mixing, with the mixture heated to 110 °C for approximately 24 hours. Monitoring the reaction progress via TLC or HPLC is recommended to confirm complete consumption of the benzyl chloride starting material before proceeding to workup. The detailed standardized synthesis steps, including specific quenching procedures and purification parameters, are provided in the technical guide below to assist process chemists in replicating these high-yielding results.
- Charge a reaction vessel with palladium acetate catalyst, DPPP ligand, solid CO substitute (TFBen), triethylamine base, propargyl amine substrate, and benzyl chloride in acetonitrile solvent.
- Heat the reaction mixture to 110 °C and maintain stirring for 24 hours under inert atmosphere to facilitate the bis-carbonylation cyclization.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the high-purity 1,5-dihydro-2H-pyrrole-2-one product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology represents a strategic opportunity to de-risk the sourcing of critical heterocyclic building blocks. The shift away from hazardous gaseous reagents to stable solid surrogates drastically simplifies the logistics of raw material handling, removing the need for specialized gas cylinder storage and high-pressure reactor certification. This simplification directly translates to lower overhead costs and faster turnaround times for production batches, enhancing the overall agility of the manufacturing site. Furthermore, the reliance on commodity chemicals like benzyl chlorides and propargyl amines ensures that the supply chain is not vulnerable to the volatility often seen with exotic or highly specialized reagents. This stability allows for better long-term planning and inventory management, securing a continuous flow of materials essential for uninterrupted API production schedules.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of high-pressure infrastructure and the use of inexpensive, commercially available starting materials. By avoiding the capital intensity associated with CO gas handling, manufacturers can significantly reduce their fixed asset depreciation costs per kilogram of product. Additionally, the high reaction efficiency and yields (often exceeding 80%) minimize raw material waste, improving the overall mass balance and reducing the cost of goods sold (COGS). The simplified workup procedure, which avoids complex extractions or distillations, further lowers utility consumption and labor hours, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against varying electronic properties of the substrates ensures consistent output regardless of minor fluctuations in raw material quality. Since the starting materials are bulk chemicals produced by multiple global vendors, the risk of single-source supply disruption is mitigated. This diversification of the supply base provides procurement teams with greater negotiating power and flexibility. Moreover, the mild reaction conditions reduce the likelihood of equipment failure or unplanned downtime due to thermal runaway or pressure excursions, guaranteeing a more predictable delivery schedule to downstream customers.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage scale is straightforward due to the absence of gas-liquid mass transfer limitations that typically plague carbonylation reactions. The homogeneous nature of the reaction mixture allows for easy heat management in larger vessels, facilitating a smooth transition from pilot to commercial production. From an environmental standpoint, the use of a solid CO source minimizes the release of volatile organic compounds and toxic gases into the atmosphere, aligning with increasingly stringent global environmental regulations. The reduced waste generation and lower energy footprint make this method a sustainable choice for companies aiming to improve their green chemistry metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: Unlike traditional methods that require handling toxic, high-pressure carbon monoxide gas cylinders, this patented process utilizes a solid carbon monoxide substitute (TFBen). This eliminates the need for specialized high-pressure autoclaves and significantly reduces logistical hazards and regulatory burdens associated with gaseous CO transport and storage.
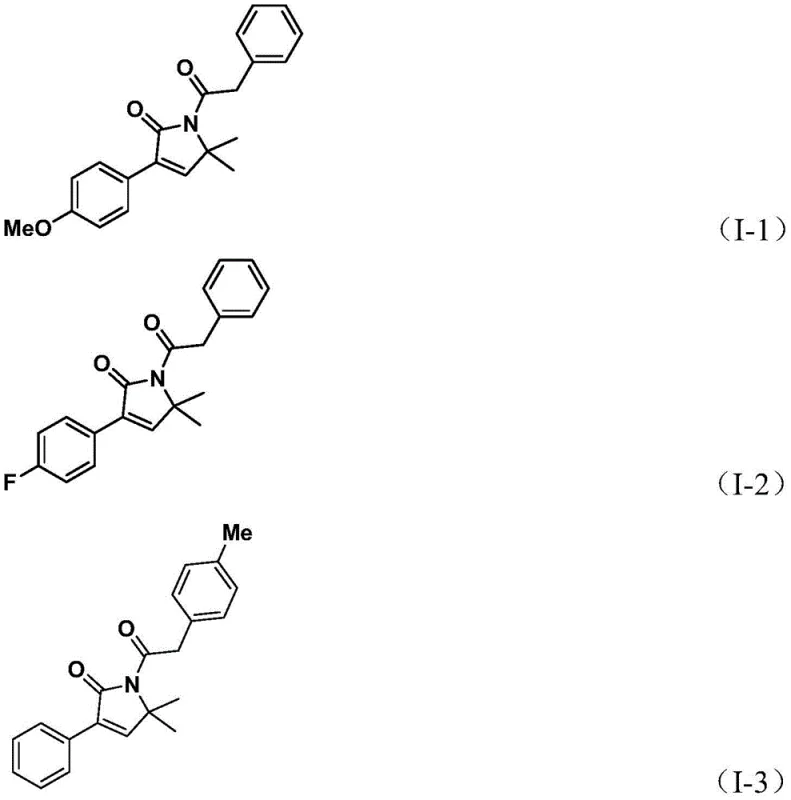
Q: What represents the maximum yield achieved for the 1,5-dihydro-2H-pyrrole-2-one scaffold in the provided examples?
A: In the optimized examples provided within the patent data, specifically Example 1 utilizing a 4-methoxy substituted benzyl chloride, the reaction achieved an isolated yield of 92%. This demonstrates high efficiency even with electron-donating groups on the aromatic ring.
Q: Is this process suitable for substrates containing sensitive functional groups like halogens or trifluoromethyl groups?
A: Yes, the method exhibits excellent substrate compatibility. The patent data confirms successful synthesis with substrates containing fluoro, chloro, bromo, and trifluoromethyl groups (Examples 2, 4, 5), yielding products in the range of 75% to 82%, indicating robust tolerance for diverse electronic environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality intermediates play in the development of life-saving medications. Our technical team has thoroughly analyzed the potential of the palladium-catalyzed bis-carbonylation route described in CN112694430B and is fully prepared to leverage this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,5-dihydro-2H-pyrrole-2-one delivered meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to explore how this advanced synthesis method can optimize your specific project requirements. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to this safer, more efficient route can impact your bottom line. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss custom route feasibility assessments for your proprietary targets. Let us be your partner in driving innovation and efficiency in your supply chain.
