Revolutionizing Triazole Synthesis: Scalable Iodine-Catalyzed Process for High-Purity Pharmaceutical Intermediates
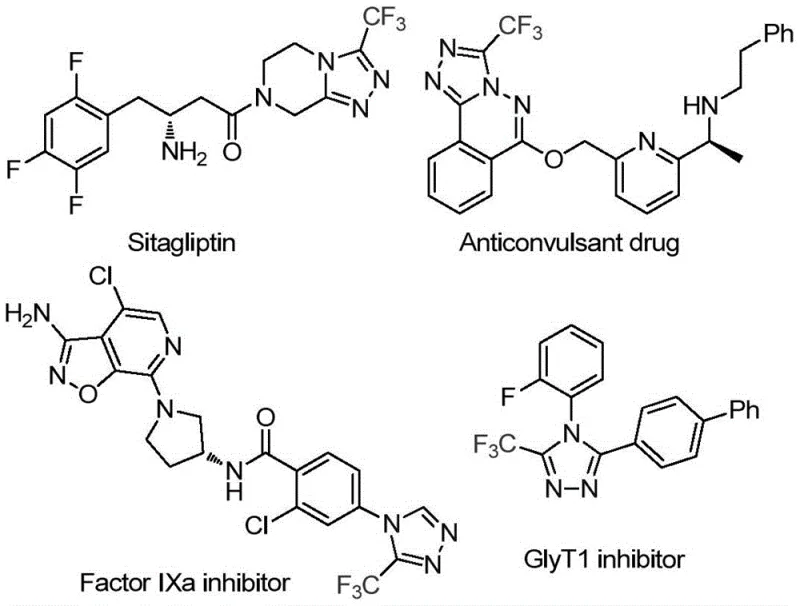
Patent CN114920707B introduces a groundbreaking synthetic methodology for producing 3-trifluoromethyl substituted 1,2,4-triazole compounds, which serve as critical building blocks in numerous pharmaceutical applications including factor IXa inhibitors and GlyT1 inhibitors. This innovative approach leverages dimethylformamide (DMF) not merely as a conventional solvent but as an integrated carbon source within the reaction matrix, enabling a streamlined process that operates effectively under ambient atmospheric conditions without requiring specialized anhydrous or anaerobic environments. The methodology represents a significant advancement over traditional triazole synthesis techniques by eliminating complex setup requirements while maintaining high functional group tolerance across diverse aromatic substrates. By utilizing molecular iodine as a catalyst at moderate temperatures of 120°C for 12 hours, the process achieves consistent conversion rates while accommodating various substituents on the aromatic ring including alkyl, alkoxy, halogen, and trifluoromethyl groups. This patent addresses longstanding industry challenges in producing fluorinated heterocyclic compounds by providing a practical solution that balances synthetic efficiency with operational simplicity, making it particularly valuable for pharmaceutical manufacturers seeking reliable access to high-purity triazole intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethyl-substituted 1,2,4-triazole compounds typically require stringent anhydrous and anaerobic conditions that necessitate specialized equipment and careful handling procedures, significantly increasing operational complexity and production costs. These conventional methods often employ transition metal catalysts that introduce additional purification challenges due to potential metal contamination in the final product, which is particularly problematic for pharmaceutical applications requiring ultra-high purity standards. The multi-step nature of existing synthetic routes frequently involves unstable intermediates that demand precise temperature control and immediate processing, creating bottlenecks in manufacturing scalability and supply chain reliability. Furthermore, the limited substrate scope of traditional methodologies restricts the structural diversity of accessible triazole derivatives, hindering medicinal chemistry optimization efforts for drug discovery programs. The requirement for expensive ligands and specialized reaction vessels further compounds the economic burden, making large-scale production economically challenging for many pharmaceutical intermediates manufacturers.
The Novel Approach
The patented methodology overcomes these limitations through an elegant iodine-catalyzed process that operates efficiently under ambient atmospheric conditions without requiring moisture-sensitive handling or inert gas environments. By utilizing DMF as both solvent and carbon source, the reaction eliminates the need for additional carbon-containing reagents while maintaining high functional group tolerance across a wide range of aromatic substrates including those with electron-donating and electron-withdrawing substituents. The process demonstrates remarkable simplicity with only three components required: trifluoroethyliminohydrazide, DMF, and molecular iodine at a stoichiometric ratio of 1:1.5. Operating at moderate temperatures of 120°C for 12 hours in standard glassware, this approach achieves consistent conversion rates while avoiding transition metal catalysts that could contaminate the final product. The broad substrate scope accommodates various substituents including methyl, methoxy, methylthio, fluorine, chlorine, and trifluoromethyl groups at different positions on the aromatic ring, enabling pharmaceutical companies to access diverse triazole derivatives for structure-activity relationship studies without modifying the core synthetic protocol.
Mechanistic Insights into Iodine-Catalyzed Triazole Formation
The reaction mechanism reveals two distinct pathways through which DMF contributes to triazole formation, demonstrating the versatility of this solvent as a dual-function reagent. When the formyl group serves as the carbon source, it undergoes condensation with trifluoroethyliminohydrazide to form a hydrazone intermediate, followed by intramolecular cyclization that eliminates dimethylamine to yield the target triazole product. Alternatively, when the N-methyl group acts as the carbon source, DMF first reacts with iodine to generate an amine salt that eliminates hydrogen iodide before undergoing nucleophilic addition with the hydrazide component. This pathway proceeds through elimination of N-methylformamide to form an azadiene intermediate, which then undergoes intramolecular nucleophilic addition and oxidative aromatization to produce the final triazole structure. Both mechanisms operate simultaneously under the reaction conditions, contributing to the overall efficiency of the process while maintaining high selectivity for the desired product formation.
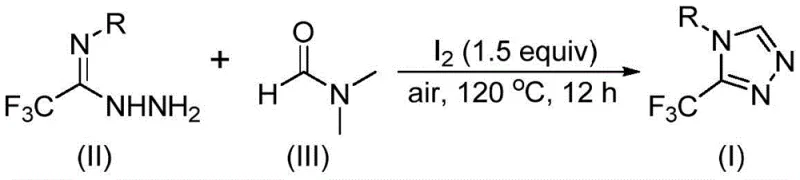
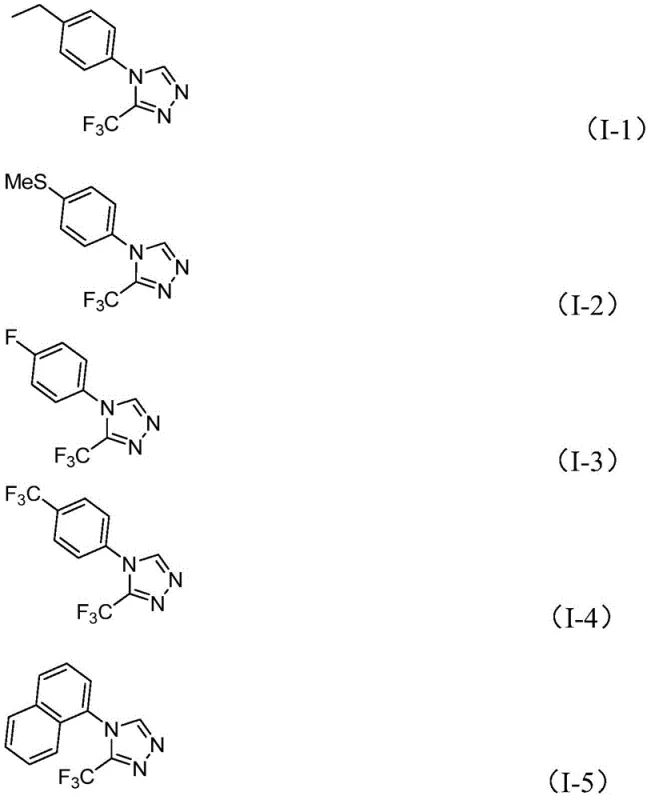
The dual reactivity pathways provide inherent robustness to the synthetic process by ensuring continuous product formation even if one pathway becomes less favorable due to specific substrate characteristics. This mechanistic flexibility explains the broad substrate scope observed in the patent examples, where various substituted aromatic amines successfully produce the corresponding triazole derivatives with consistent yields ranging from 42% to 61%. The absence of transition metals in this iodine-catalyzed system eliminates concerns about metal contamination in the final product, which is particularly advantageous for pharmaceutical applications requiring stringent purity specifications. Furthermore, the reaction's tolerance to atmospheric conditions stems from iodine's ability to mediate the transformation without being deactivated by oxygen or moisture, a significant improvement over conventional methods that require carefully controlled environments.
How to Synthesize Trifluoromethyl-Triazoles Efficiently
This patented methodology provides a robust framework for producing high-purity trifluoromethyl-substituted triazole intermediates through a streamlined synthetic route that eliminates traditional process complexities. The following standardized procedure details the precise implementation of this innovative approach for consistent manufacturing outcomes across diverse production scales.
- Combine trifluoroethyliminohydrazide and DMF in Schlenk tube with iodine catalyst under air atmosphere
- Heat reaction mixture to 120°C and maintain for 12 hours with continuous stirring
- Perform standard workup including filtration, silica gel mixing, and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic approach delivers substantial value to procurement and supply chain operations by addressing critical pain points associated with traditional triazole production methods. The elimination of specialized equipment requirements and complex environmental controls translates directly into reduced capital expenditure and operational complexity for manufacturing partners.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes expensive purification steps required to remove metal contaminants from pharmaceutical intermediates, resulting in significant cost savings throughout the production process. The use of commercially available DMF as both solvent and reactant streamlines material sourcing while reducing raw material costs compared to conventional methods requiring additional carbon sources. Simplified process requirements enable existing manufacturing facilities to adopt this technology without major capital investments in new equipment or specialized infrastructure.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials including molecular iodine and standard organic solvents ensures consistent supply chain performance without vulnerability to specialty chemical shortages. The ambient atmosphere operation eliminates dependency on specialized gas handling systems that can create production bottlenecks during supply disruptions. The broad substrate compatibility allows for flexible sourcing strategies where different aromatic amines can be used interchangeably based on market availability without requiring process revalidation.
- Scalability and Environmental Compliance: The straightforward reaction protocol with minimal byproducts facilitates seamless scale-up from laboratory to commercial production volumes while maintaining high purity standards required for pharmaceutical applications. The absence of hazardous metal catalysts simplifies waste stream management and reduces environmental compliance burdens associated with heavy metal disposal. Standard workup procedures using conventional purification techniques ensure consistent product quality across all production scales without requiring specialized equipment or expertise.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented synthetic methodology for pharmaceutical intermediate production.
Q: How does this method eliminate the need for anhydrous and anaerobic conditions?
A: The iodine-catalyzed reaction proceeds efficiently in air using DMF as both solvent and carbon source, eliminating the need for specialized inert atmosphere equipment and moisture-sensitive handling procedures that characterize conventional triazole synthesis methods.
Q: What is the significance of DMF serving dual roles as solvent and reactant?
A: DMF's dual functionality as both reaction medium and carbon source simplifies the process by reducing the number of required components while providing consistent reaction efficiency across diverse substrate variations without additional purification steps.
Q: How does this method support scalable production of pharmaceutical intermediates?
A: The straightforward reaction protocol with commercially available materials, ambient atmosphere operation, and simple workup procedures enables seamless scale-up from laboratory to commercial production volumes while maintaining high purity standards required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl-Triazole Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex pharmaceutical intermediates while maintaining stringent purity specifications through rigorous QC labs and advanced analytical capabilities. This patented methodology aligns perfectly with our commitment to delivering innovative solutions that balance synthetic efficiency with commercial viability for global pharmaceutical partners seeking reliable access to high-value fluorinated heterocyclic compounds.
Request a Customized Cost-Saving Analysis from our technical procurement team to understand how this innovative synthesis approach can optimize your specific supply chain requirements. We welcome inquiries for specific COA data and route feasibility assessments tailored to your pharmaceutical development needs.
