Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Novel Molybdenum-Copper Catalysis
Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Novel Molybdenum-Copper Catalysis
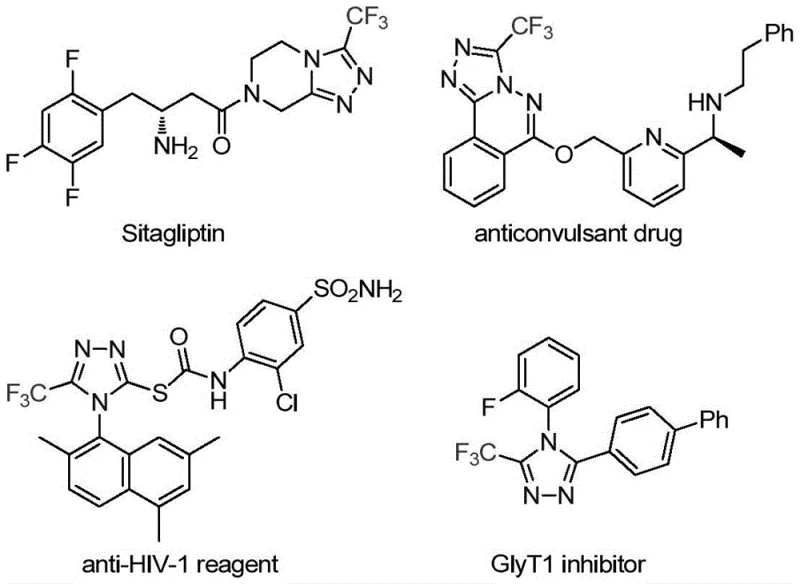
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine atoms which are pivotal for enhancing drug efficacy. A significant breakthrough in this domain is detailed in patent CN113307778A, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These structural motifs are not merely academic curiosities; they are foundational scaffolds found in numerous bioactive molecules, including prominent drugs like Sitagliptin and various anticonvulsants. The introduction of a trifluoromethyl group significantly modulates physicochemical properties such as lipophilicity, metabolic stability, and bioavailability, making these intermediates indispensable for modern medicinal chemistry. This report analyzes the technical merits of this novel synthetic route, evaluating its potential to serve as a reliable platform for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on methodologies that often suffer from significant operational drawbacks and safety concerns. Traditional literature reports predominantly describe the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These classical routes frequently necessitate harsh reaction conditions, involving strong acids or bases and elevated temperatures that can degrade sensitive functional groups. Furthermore, alternative multi-component reactions utilizing diazonium salts or trifluorodiazoethane introduce severe safety hazards due to the explosive nature of diazo compounds, posing unacceptable risks for large-scale manufacturing environments. Additionally, many existing protocols struggle with regioselectivity, often yielding mixtures of fully substituted triazoles rather than the specific 3,4-disubstituted isomers required for targeted drug design, thereby complicating downstream purification and reducing overall process efficiency.
The Novel Approach
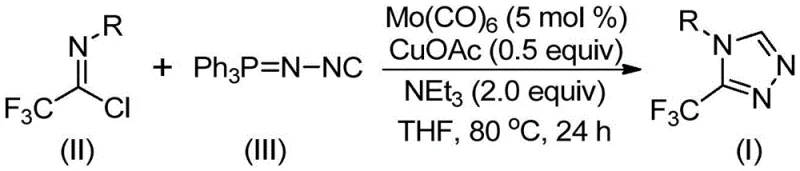
In stark contrast to these legacy methods, the technology disclosed in patent CN113307778A presents a paradigm shift by employing a molybdenum and copper co-catalyzed cycloaddition strategy. This innovative approach utilizes trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as the primary building blocks, reacting them under remarkably mild conditions ranging from 70 to 90°C. The use of molybdenum hexacarbonyl as a metal activator alongside cuprous acetate creates a unique catalytic environment that facilitates the formation of the five-membered triazole ring with high precision. This method eliminates the need for hazardous diazo reagents and avoids the extreme pH conditions associated with hydrazine cyclizations. Moreover, the reaction demonstrates exceptional substrate tolerance, allowing for the incorporation of diverse aryl and alkyl groups without compromising yield, thus offering a versatile and safer pathway for generating high-purity pharmaceutical intermediates suitable for rigorous regulatory standards.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The core of this synthetic advancement lies in the synergistic interaction between the molybdenum and copper catalysts, which orchestrates a sophisticated [3+2] cycloaddition mechanism. Initially, the molybdenum hexacarbonyl interacts with the functionalized isonitrile to form a reactive metal-isocyanide complex, effectively activating the carbon-nitrogen triple bond for nucleophilic attack. Subsequently, the cuprous acetate promotes the cycloaddition of this activated species to the trifluoroethylimidoyl chloride, leading to the formation of a transient five-membered ring intermediate. This step is critical as it establishes the triazole core while maintaining the integrity of the trifluoromethyl group. The final transformation involves the elimination of triphenylphosphine oxide, driven by trace water in the system, to yield the stable 3-trifluoromethyl-substituted 1,2,4-triazole product. This mechanistic pathway ensures high atom economy and minimizes the generation of toxic byproducts, aligning with green chemistry principles essential for sustainable manufacturing.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in ensuring product quality. By operating at moderate temperatures (70-90°C) rather than the high thermal stress required by traditional cyclizations, the formation of thermal degradation products and polymerization byproducts is significantly suppressed. The specific choice of tetrahydrofuran (THF) as the preferred solvent further enhances selectivity, as it effectively dissolves all reactants while stabilizing the catalytic species. The use of molecular sieves in the reaction mixture helps to manage moisture levels, preventing hydrolysis of the sensitive imidoyl chloride starting material while still allowing the necessary water-mediated elimination step to proceed at the end of the cycle. This precise control over the reaction environment results in a cleaner crude profile, reducing the burden on downstream purification processes such as column chromatography and facilitating the isolation of the target compound with high purity specifications.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, making it highly attractive for process chemistry teams aiming to transition from discovery to development scales. The procedure involves charging a reactor with the catalytic system comprising molybdenum hexacarbonyl and cuprous acetate, along with triethylamine as a base and molecular sieves to regulate water content. Once the catalyst mixture is prepared in the organic solvent, the trifluoroethylimidoyl chloride and the functionalized isonitrile are introduced, initiating the cycloaddition process. The reaction is allowed to proceed for a duration of 18 to 30 hours, ensuring complete conversion of the starting materials. Following the reaction period, the workup is straightforward, involving filtration to remove solid residues and standard silica gel chromatography to isolate the pure product. For a comprehensive breakdown of the specific stoichiometric ratios and temperature profiles optimized for different substrates, please refer to the standardized synthesis steps outlined below.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 70-90°C and stir for 18-30 hours, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling strategic advantages that extend beyond mere chemical elegance. The primary benefit lies in the accessibility and cost-effectiveness of the raw materials; both trifluoroethylimidoyl chloride and the functionalized isonitrile precursors are commercially available or can be synthesized from inexpensive commodity chemicals like aromatic amines and trifluoroacetic acid. This reliance on abundant feedstocks mitigates the risk of supply chain disruptions often associated with exotic or proprietary reagents. Furthermore, the elimination of hazardous diazo compounds removes the need for specialized explosion-proof infrastructure and complex safety protocols, thereby drastically simplifying facility requirements and reducing capital expenditure related to safety compliance. The robustness of the reaction also implies a more predictable manufacturing timeline, as the mild conditions reduce the likelihood of batch failures due to thermal runaways or side reactions.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the use of relatively inexpensive catalysts such as cuprous acetate and molybdenum hexacarbonyl, which are used in catalytic quantities rather than stoichiometric amounts. Unlike traditional methods that may require expensive ligands or precious metals like palladium, this system utilizes base metals that offer a favorable balance between activity and cost. The high reaction efficiency and conversion rates observed across a wide range of substrates mean that less raw material is wasted, directly improving the overall mass balance and reducing the cost of goods sold. Additionally, the simplified post-treatment process, which avoids complex extraction or neutralization steps typical of acidic or basic cyclizations, leads to substantial savings in labor and waste disposal costs.
- Enhanced Supply Chain Reliability: The versatility of this synthetic method allows for the production of a diverse library of triazole derivatives from a common set of starting materials, providing supply chain flexibility. Since the reaction tolerates various substituents including halogens, alkyl groups, and electron-withdrawing groups, manufacturers can produce multiple intermediates using the same equipment and general protocol. This standardization reduces the need for dedicated production lines for specific analogs, thereby increasing asset utilization and responsiveness to market demand. The ability to source starting materials from multiple global suppliers further de-risks the supply chain, ensuring continuity of supply even if one vendor faces production issues, which is critical for maintaining the uninterrupted manufacture of downstream active pharmaceutical ingredients.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this method has been explicitly validated for expansion to gram-level reactions with clear potential for kilogram and tonne-scale production. The use of THF, a common industrial solvent with well-established recovery and recycling protocols, facilitates easy integration into existing manufacturing infrastructure. From an environmental standpoint, the avoidance of toxic hydrazines and explosive diazo compounds significantly lowers the environmental footprint of the process. The generation of triphenylphosphine oxide as a byproduct is manageable and can often be recycled or disposed of safely, aligning with increasingly stringent global regulations on chemical waste. This compliance readiness ensures that the manufacturing process remains sustainable and viable in the long term without requiring costly retrofits for waste treatment.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of a new synthetic methodology is crucial for stakeholders involved in process development and quality assurance. The following questions address common inquiries regarding the operational parameters and scope of this Mo/Cu co-catalyzed triazole synthesis. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for evaluating the technology's fit for your specific project requirements. Whether you are concerned about substrate compatibility or reaction scalability, these insights aim to clarify the practical implications of adopting this advanced chemical route.
Q: What are the key advantages of this Mo/Cu co-catalyzed method over traditional triazole synthesis?
A: This method utilizes mild reaction conditions (70-90°C) compared to harsh thermal cyclizations, employs cheap and readily available starting materials like trifluoroethylimidoyl chloride, and achieves high reaction efficiency with broad substrate tolerance.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the patent explicitly states that the method can be expanded to gram-level reactions and provides a possibility for subsequent scale production due to its simple operation and high conversion rates in solvents like THF.
Q: What types of substituents are tolerated on the aryl ring of the product?
A: The method demonstrates wide functional group tolerance, successfully accommodating substituents such as methyl, methoxy, fluoro, chloro, nitro, and tert-butyl groups at ortho, meta, or para positions on the phenyl ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
The technological advancements described in patent CN113307778A represent a significant opportunity for optimizing the supply chain of critical heterocyclic intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage this novel Mo/Cu co-catalyzed methodology for the benefit of our global partners. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole meets the exacting standards required for pharmaceutical applications. Our commitment to quality and consistency makes us a trusted partner for companies seeking to secure a stable supply of high-value chemical building blocks.
We invite you to explore how this innovative synthesis route can enhance your project's economics and timeline. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to contact our technical procurement team to request specific COA data for our existing inventory or to discuss route feasibility assessments for custom derivatives. By collaborating with us, you gain access to cutting-edge synthetic technologies combined with the reliability of a seasoned CDMO partner, positioning your supply chain for success in a competitive market.
