Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are critical for enhancing metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in Chinese patent CN113307778A, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology addresses the growing demand for reliable pharmaceutical intermediate suppliers by offering a pathway that utilizes cheap and easily obtainable starting materials while maintaining high reaction efficiency. The core innovation lies in the co-catalytic system employing molybdenum hexacarbonyl and cuprous acetate, which facilitates the cycloaddition of functionalized isonitriles with trifluoroethylimidoyl chloride under remarkably mild conditions. This approach not only simplifies the synthetic route but also ensures that the resulting molecular frameworks are suitable for further elaboration into active pharmaceutical ingredients (APIs) and agrochemical agents.
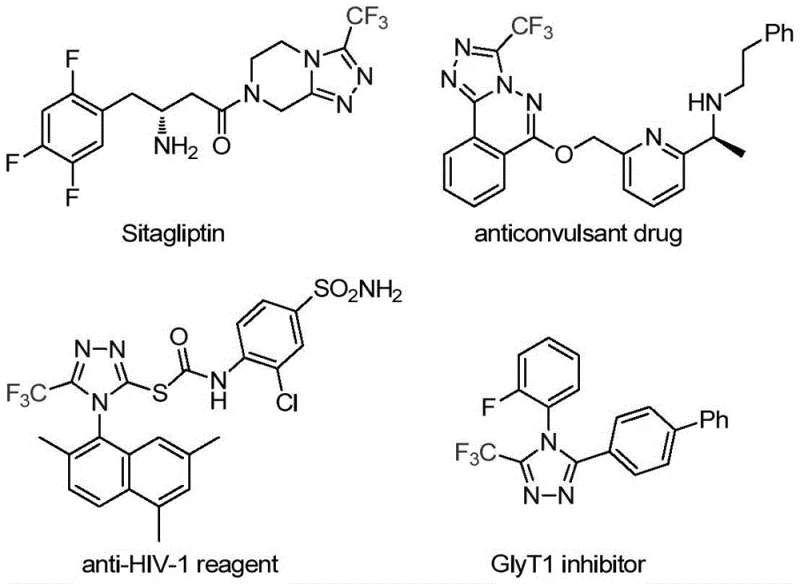
The strategic importance of the 1,2,4-triazole scaffold cannot be overstated, as evidenced by its presence in blockbuster drugs like Sitagliptin and various antifungal agents. The introduction of a trifluoromethyl group at the 3-position further amplifies the physicochemical properties of these molecules, making them indispensable building blocks in modern medicinal chemistry. By leveraging the methodology described in CN113307778A, manufacturers can access a versatile library of these valuable intermediates, thereby supporting the development of next-generation therapeutics with improved pharmacokinetic profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been plagued by significant operational challenges and safety concerns that hinder large-scale production. Traditional literature reports predominantly rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones, processes that often require harsh reaction conditions and generate substantial waste. Furthermore, alternative copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane pose severe safety risks due to the explosive nature of diazo compounds, making them unsuitable for cost reduction in API manufacturing environments. These legacy methods frequently suffer from limited substrate scope, poor atom economy, and the necessity for expensive or difficult-to-handle reagents, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
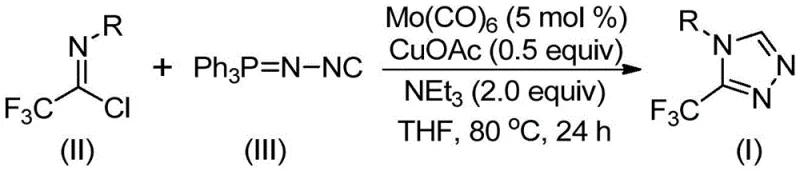
In stark contrast to these archaic techniques, the novel approach presented in the patent utilizes a transition metal co-catalyzed strategy that dramatically simplifies the synthetic landscape. By employing trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as the primary building blocks, the reaction proceeds through a streamlined [3+2] cycloaddition mechanism that avoids the use of hazardous diazo species. The reaction conditions are exceptionally mild, typically operating between 70~90℃, which significantly reduces energy consumption and thermal stress on sensitive functional groups. This method allows for the direct construction of the triazole ring with high regioselectivity, yielding 3,4-disubstituted products that are otherwise difficult to access. The operational simplicity, combined with the use of commercially available catalysts like Mo(CO)6 and CuOAc, positions this technology as a superior choice for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The success of this synthetic route is underpinned by a sophisticated dual-metal catalytic cycle that orchestrates the assembly of the triazole ring with precision. Mechanistically, the reaction initiates with the activation of the functionalized isonitrile by molybdenum hexacarbonyl, forming a reactive metal-isocyanide complex that primes the carbon center for nucleophilic attack. Simultaneously, the cuprous acetate promoter facilitates the coordination of the trifluoroethylimidoyl chloride, lowering the activation energy for the subsequent cycloaddition step. This synergistic interaction enables a smooth [3+2] cyclization to form a five-membered ring intermediate, which is a critical juncture in the formation of the heterocyclic core. The elegance of this mechanism lies in its ability to proceed without the need for external oxidants or extreme temperatures, thereby preserving the integrity of diverse functional groups attached to the aromatic rings.
Following the ring closure, the system undergoes a spontaneous elimination of triphenylphosphine oxide, driven by the thermodynamic stability of the final aromatic triazole product. This step is crucial for impurity control, as the phosphine oxide byproduct is easily removed during the standard workup procedures, ensuring a clean crude profile. The tolerance of the catalytic system towards various electronic environments on the aryl ring—ranging from electron-donating methyl and methoxy groups to electron-withdrawing fluoro and nitro substituents—demonstrates the robustness of the mechanism. Such mechanistic resilience is vital for R&D directors who require consistent purity and reproducible yields across different batches, as it minimizes the formation of side products that could complicate downstream purification.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings, requiring standard glassware and common organic solvents. The protocol involves charging a reaction vessel with the requisite molar ratios of trifluoroethylimidoyl chloride, functionalized isonitrile, molybdenum hexacarbonyl, cuprous acetate, and triethylamine in tetrahydrofuran (THF). The mixture is then heated to the specified temperature range and stirred until conversion is complete, typically monitored by TLC or HPLC. Detailed standardized synthesis steps see the guide below.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature range of 70~90℃ and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix the crude sample with silica gel, and perform column chromatography purification to isolate the final 3-trifluoromethyl-substituted 1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Mo/Cu co-catalyzed method translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the raw material portfolio; since both the trifluoroethylimidoyl chloride and the functionalized isonitrile are derived from readily available commodity chemicals, the risk of supply disruption is significantly minimized. This accessibility ensures a stable flow of materials, which is critical for maintaining continuous production schedules in a volatile global market. Furthermore, the elimination of hazardous diazo reagents removes the need for specialized explosion-proof infrastructure, thereby reducing capital expenditure and insurance costs associated with manufacturing these high-value intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the use of relatively inexpensive catalysts such as cuprous acetate and molybdenum hexacarbonyl, which are used in low catalytic loadings. By avoiding the use of precious metals like palladium or rhodium, the overall cost of goods sold (COGS) is substantially lowered, allowing for more competitive pricing in the marketplace. Additionally, the mild reaction conditions reduce energy overheads, and the straightforward workup procedure minimizes solvent usage and waste disposal fees, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or reagent quality, leading to higher batch-to-batch consistency. This reliability is paramount for supply chain planners who must guarantee delivery timelines to downstream API manufacturers. The ability to source starting materials from multiple vendors without compromising reaction performance further de-risks the supply chain, ensuring that production targets can be met even if one supplier faces logistical challenges.
- Scalability and Environmental Compliance: From an environmental perspective, the method aligns well with green chemistry principles by generating fewer hazardous byproducts and utilizing safer solvents like THF. The scalability of the reaction has been demonstrated up to the gram level in the patent examples, and the absence of exothermic hazards suggests a smooth path to kilogram and ton-scale production. This scalability, combined with reduced waste generation, facilitates easier regulatory compliance and supports the sustainability goals of modern chemical enterprises.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the reaction parameters and scope. These insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy and relevance for process development discussions. Understanding these nuances is essential for optimizing the synthesis for specific target molecules and integrating the process into existing manufacturing workflows.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies reacting at 70~90℃ for 18~30 hours in THF solvent, utilizing Mo(CO)6 and CuOAc as the catalytic system with triethylamine as the base.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates wide substrate tolerance, successfully synthesizing derivatives with substituents such as methyl, methoxy, fluoro, chloro, and nitro groups on the aromatic ring.
Q: Is the purification process complex for these intermediates?
A: The post-processing is straightforward, involving simple filtration followed by standard column chromatography purification, avoiding complex extraction or distillation steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug discovery and development pipeline. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. By partnering with us, you gain access to a supply chain that is not only reliable but also deeply knowledgeable about the intricacies of fluorine chemistry and heterocyclic synthesis.
We invite you to engage with our technical procurement team to discuss how this advanced Mo/Cu catalyzed technology can be tailored to your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new targets, we are here to provide the support you need. Contact us today to secure a sustainable and cost-effective supply of these vital pharmaceutical building blocks.
