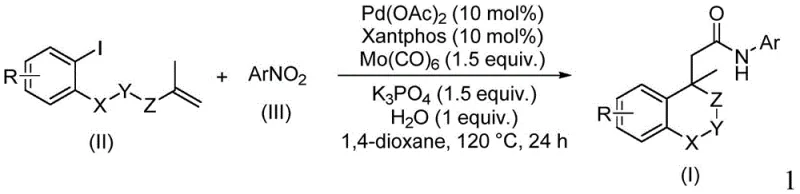
Advanced Palladium-Catalyzed Synthesis of Isochroman Amides for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly those containing amide linkages which are ubiquitous in bioactive molecules. A significant breakthrough in this domain is detailed in patent CN114539198A, which discloses a novel preparation method for amide compounds containing (iso)chroman structures. This technology represents a paradigm shift from traditional acylation methods, utilizing a sophisticated palladium-catalyzed cyclocarbopalladation and aminocarbonylation cascade. By leveraging nitroaromatic hydrocarbons as the nitrogen source and molybdenum carbonyl as both the carbonyl source and reducing agent, this method achieves high reaction efficiency with exceptional functional group tolerance. For R&D directors and procurement strategists, this innovation offers a compelling route to access high-value intermediates with reduced operational complexity and enhanced atom economy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of amide-functionalized heterocycles has relied heavily on the activation of carboxylic acids or their derivatives, such as acid chlorides or anhydrides, followed by nucleophilic attack by amines. While effective on a small scale, these conventional routes often suffer from significant drawbacks when applied to complex substrates like isochromans. The requirement for pre-functionalized starting materials increases the step count and overall cost, while the generation of stoichiometric waste during activation poses environmental challenges. Furthermore, traditional methods often struggle with chemoselectivity when multiple reactive sites are present on the molecule, leading to difficult purification processes and lower overall yields. The reliance on gaseous carbon monoxide for carbonylation reactions also introduces severe safety hazards and requires specialized high-pressure equipment, creating bottlenecks for supply chain continuity and commercial scalability.
The Novel Approach
The methodology described in the patent overcomes these historical barriers by integrating the ring-closing step and amide formation into a single catalytic cycle. Instead of using hazardous CO gas or pre-activated acids, the process employs solid molybdenum carbonyl [Mo(CO)₆], which safely releases CO in situ under thermal conditions. Crucially, the use of nitroarenes as nitrogen surrogates eliminates the need for sensitive amine starting materials, which can be prone to oxidation or difficult to store. This tandem reaction sequence, driven by a palladium catalyst and Xantphos ligand, constructs the isochroman core and installs the amide moiety simultaneously. This convergence not only drastically shortens the synthetic route but also improves the overall mass balance of the process, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pd-Catalyzed Cyclocarbopalladation and Aminocarbonylation
The core of this transformation lies in a meticulously orchestrated catalytic cycle initiated by the oxidative addition of the aryl iodide to the palladium(0) species generated in situ. Following this activation, an intramolecular Heck-type cyclization occurs, where the palladium inserts across the pendant alkene to form a critical sigma-alkyl-palladium intermediate. This step is pivotal for constructing the rigid isochroman skeleton with high stereochemical control. Subsequently, the insertion of carbon monoxide, derived from the decomposition of molybdenum carbonyl, generates an acyl-palladium complex. Unlike traditional carbonylations that require external nucleophiles, this system uniquely utilizes the nitroarene, which is concurrently reduced by the molybdenum species to an amine or nitroso intermediate. This reactive nitrogen species then attacks the acyl-palladium center, facilitating reductive elimination to release the final amide product and regenerate the active palladium catalyst.
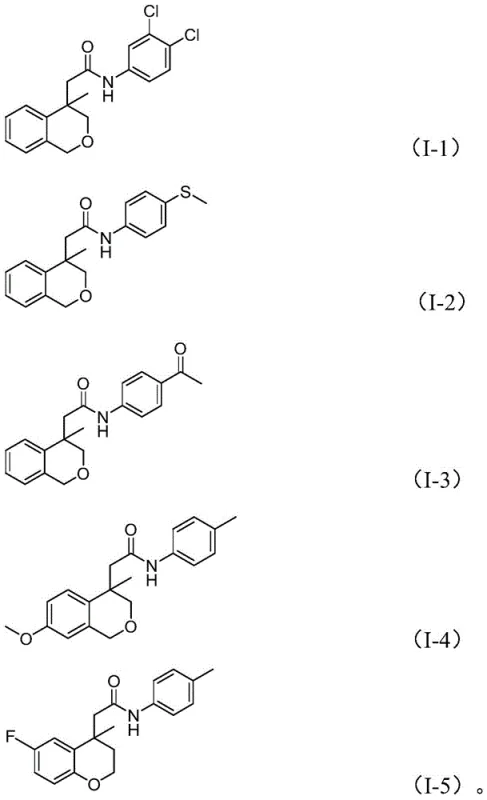
From an impurity control perspective, this mechanism offers distinct advantages over stepwise syntheses. Because the ring closure and amidation occur in a concerted manner within the coordination sphere of the metal, there is minimal opportunity for intermolecular side reactions or polymerization of the alkene moiety. The use of water as a crucial additive further assists in the reduction of the nitro group and stabilizes the catalytic cycle, ensuring that side products such as ureas or over-reduced amines are kept to a minimum. The broad substrate scope demonstrated in the patent, tolerating electron-withdrawing groups like halogens and trifluoromethyl groups as well as electron-donating methoxy groups, confirms the robustness of this catalytic system. This level of precision is essential for producing high-purity OLED material or pharmaceutical intermediates where trace impurities can compromise biological activity or material performance.
How to Synthesize Isochroman Amides Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the dual-function reagents and the thermal profile of the reaction. The process is designed to be operationally simple, utilizing standard laboratory glassware or stainless steel reactors capable of maintaining temperatures up to 120°C. The detailed protocol involves mixing the iodoarene substrate with the nitroarene coupling partner in 1,4-dioxane, followed by the addition of the palladium catalyst system and molybdenum carbonyl. The reaction proceeds under atmospheric pressure, removing the need for autoclaves typically associated with carbonylation chemistry. For a comprehensive understanding of the specific molar ratios, temperature gradients, and workup procedures required to achieve the reported yields of up to 96%, please refer to the standardized synthesis guide below.
- Combine palladium acetate, Xantphos ligand, molybdenum carbonyl, potassium phosphate, water, iodoarene compound, and nitroarene in 1,4-dioxane.
- Heat the reaction mixture to 120°C and maintain stirring for 24 hours to ensure complete conversion via cyclocarbopalladation and aminocarbonylation.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity isochroman amide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio. By replacing expensive, moisture-sensitive amines and hazardous acid chlorides with stable, commodity-grade nitroarenes and iodoarenes, companies can significantly mitigate supply risk. These starting materials are widely available from multiple global vendors, ensuring supply continuity even during market fluctuations. Furthermore, the elimination of high-pressure carbon monoxide gas removes a major safety liability and regulatory hurdle, allowing for production in a broader range of facilities without specialized gas handling infrastructure. This flexibility translates directly into reduced capital expenditure and lower insurance costs for manufacturing sites.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of molybdenum carbonyl as a dual-purpose reagent, which effectively combines the cost of the carbonyl source and the reducing agent into a single line item. This consolidation reduces the total number of reagents required per batch, lowering both material costs and waste disposal fees. Additionally, the high yields reported across various substrates mean that less raw material is wasted on failed batches or extensive purification, leading to a more favorable cost of goods sold (COGS). The simplicity of the post-treatment process, which involves basic filtration and chromatography, further reduces labor hours and solvent consumption compared to multi-step traditional syntheses.
- Enhanced Supply Chain Reliability: Reliance on nitroaromatic compounds as nitrogen sources provides a buffer against supply chain disruptions common with specialized amine building blocks. Nitroarenes are chemically stable and have long shelf lives, allowing manufacturers to maintain strategic stockpiles without degradation concerns. This stability ensures that production schedules can be maintained consistently, reducing lead time for high-purity pharmaceutical intermediates. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, providing a more forgiving and reliable manufacturing window that supports just-in-time delivery models.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the absence of gaseous reagents and the use of standard heating protocols. The reaction operates at moderate temperatures (120°C) which are easily achievable in standard jacketed reactors, avoiding the energy intensity of cryogenic or ultra-high-temperature processes. From an environmental standpoint, the atom economy is superior to classical methods, generating less saline waste and avoiding the use of chlorinating agents. This aligns with modern green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations, thereby avoiding potential fines and enhancing corporate sustainability profiles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this route for their specific product pipelines.
Q: What are the key advantages of using nitroarenes over traditional amines in this synthesis?
A: Nitroarenes serve as stable, inexpensive, and abundant nitrogen sources that eliminate the need for pre-synthesized amines, thereby reducing raw material costs and simplifying the supply chain for large-scale manufacturing.
Q: How does the molybdenum carbonyl function in this catalytic system?
A: Molybdenum carbonyl acts as a dual-function reagent, serving simultaneously as the carbonyl source for amide bond formation and as the reducing agent required to convert the nitro group into the reactive amine species in situ.
Q: Is this process scalable for industrial production of API intermediates?
A: Yes, the process utilizes commercially available reagents and standard heating conditions (120°C) without requiring high-pressure carbon monoxide gas, making it highly suitable for safe and efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isochroman Amides Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN114539198A for accelerating drug discovery and process development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle complex organometallic chemistry safely, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for clinical and commercial applications. We are committed to delivering high-purity isochroman amides that empower your research and production goals.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and reduce your overall manufacturing costs.
