Advanced Palladium-Catalyzed Synthesis of Isochroman Amides for Commercial Scale-Up
Introduction to Next-Generation Amide Synthesis
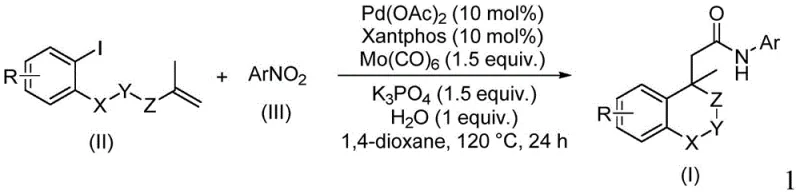
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds, particularly those containing amide linkages which are ubiquitous in bioactive molecules. Patent CN114539198A introduces a groundbreaking preparation method for amide compounds containing (iso)chroman structures, addressing critical bottlenecks in traditional synthetic routes. This innovation leverages a palladium-catalyzed cyclocarbopalladation and aminocarbonylation strategy, utilizing nitroarenes as a versatile nitrogen source and molybdenum carbonyl as a dual-purpose carbonyl source and reducing agent. For R&D directors and procurement specialists, this technology represents a significant leap forward in process efficiency, offering a pathway to high-purity intermediates with simplified operational protocols. The ability to tolerate a wide range of functional groups while maintaining high reaction efficiency makes this method particularly attractive for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of amide-containing heterocycles has relied heavily on the acylation of amines with carboxylic acids or their activated derivatives, a process that often requires harsh coupling reagents and generates substantial stoichiometric waste. Alternatively, transition metal-catalyzed carbonylation of halogenated aryl compounds typically necessitates the use of toxic, high-pressure carbon monoxide gas, posing severe safety risks and requiring specialized high-pressure reactor infrastructure that increases capital expenditure. Furthermore, conventional methods often struggle with substrate scope, particularly when dealing with sensitive functional groups that may degrade under acidic or basic activation conditions. These limitations result in higher production costs, extended lead times for high-purity intermediates, and complex purification workflows that hinder rapid process development.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes a reductive aminocarbonylation strategy that circumvents the need for gaseous CO and pre-functionalized amines. By employing nitroarenes as the nitrogen source, the process taps into a class of abundant, stable, and inexpensive raw materials that are widely available in the global chemical market. The use of molybdenum carbonyl serves as a safe, solid surrogate for carbon monoxide, releasing CO in situ under thermal conditions while simultaneously acting as a reducing agent to convert the nitro group into the requisite amine functionality. This tandem transformation allows for the direct construction of the amide bond and the heterocyclic ring in a single operational step, drastically simplifying the synthetic sequence and enhancing overall atom economy.
Mechanistic Insights into Pd-Catalyzed Cyclocarbopalladation
The core of this transformative chemistry lies in the intricate catalytic cycle driven by the palladium/Xantphos system. The reaction initiates with the oxidative addition of the iodoarene substrate to the active palladium(0) species, generating an aryl-palladium(II) intermediate. This is followed by a crucial intramolecular Heck-type cyclization, where the palladium center inserts across the pendant alkene moiety to form a sigma-alkyl-palladium species. This cyclization step is pivotal for constructing the rigid (iso)chroman core structure, establishing the stereochemical and structural integrity of the final product. The presence of the bulky Xantphos ligand is essential here, as it stabilizes the palladium center and facilitates the reductive elimination steps necessary for catalyst turnover.
Subsequent to cyclization, the sigma-alkyl-palladium intermediate undergoes carbon monoxide insertion, facilitated by the CO released from the thermal decomposition of molybdenum carbonyl. This generates an acyl-palladium species, which is then intercepted by the nitrogen nucleophile. Crucially, the nitroarene is reduced in situ, likely through interaction with the molybdenum species or the palladium hydride intermediates formed during the cycle, to generate the reactive amine species that attacks the acyl-palladium complex. This reductive aminocarbonylation sequence ensures that the amide bond is formed with high fidelity. The mechanism inherently suppresses side reactions such as homocoupling of the aryl halide or beta-hydride elimination, thereby ensuring a clean impurity profile that is critical for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize Isochroman Amides Efficiently
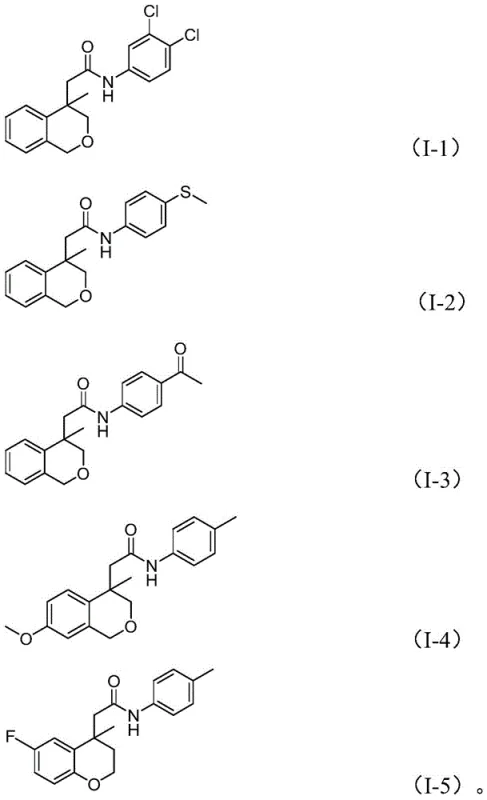
Executing this synthesis requires precise control over reaction parameters to maximize yield and purity. The protocol involves charging a sealed tube with the iodoarene substrate, the specific nitroarene coupling partner, palladium acetate catalyst, Xantphos ligand, molybdenum carbonyl, potassium phosphate base, and a stoichiometric amount of water. The reaction is conducted in 1,4-dioxane at an elevated temperature of 120°C for approximately 24 hours. This specific thermal profile ensures the complete decomposition of the carbonyl source and drives the equilibrium towards the desired amide product. Following the reaction period, the mixture is subjected to a straightforward workup involving filtration to remove metal residues and inorganic salts, followed by silica gel adsorption and column chromatography purification. Detailed standardized synthesis steps are provided in the guide below.
- Combine palladium acetate, Xantphos ligand, molybdenum carbonyl, potassium phosphate, water, iodoarene substrate, and nitroarene in 1,4-dioxane.
- Heat the reaction mixture to 120°C and maintain stirring for 24 hours to ensure complete conversion.
- Filter the reaction mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target amide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling advantages that directly address the pain points of modern supply chain management and cost engineering. By shifting away from hazardous gaseous reagents and expensive activated acid derivatives, the method significantly reduces the operational complexity and safety overhead associated with manufacturing. The reliance on commodity chemicals like nitroarenes and solid molybdenum carbonyl ensures a stable and resilient supply chain, mitigating the risks of raw material shortages that often plague specialty chemical production. Furthermore, the high functional group tolerance means that fewer protection and deprotection steps are required, leading to a shorter overall manufacturing timeline and reduced solvent consumption.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide infrastructure removes a major capital expenditure barrier, allowing production to occur in standard glass-lined or stainless steel reactors without specialized pressure ratings. Additionally, the use of nitroarenes as nitrogen sources is inherently more cost-effective than purchasing pre-functionalized anilines or amines, which often carry higher price tags due to additional synthetic steps in their own production. The simplified downstream processing, characterized by simple filtration and standard chromatography, further lowers labor and utility costs per kilogram of product, driving substantial cost savings in API manufacturing without compromising quality.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, including various substituted iodoarenes and nitroarenes, are widely produced commodities with robust global supply networks. This abundance ensures that procurement teams can secure raw materials with short lead times and minimal risk of disruption. The stability of the reagents also allows for long-term storage without significant degradation, enabling manufacturers to maintain strategic stockpiles. This reliability is crucial for maintaining continuous production schedules and meeting the rigorous delivery deadlines demanded by downstream pharmaceutical clients, effectively reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: The reaction conditions are mild enough to be scaled from gram-scale laboratory synthesis to multi-ton commercial production without encountering significant exothermic hazards or pressure spikes. The use of 1,4-dioxane, while requiring careful handling, is a well-understood solvent in industrial settings with established recovery and recycling protocols. Moreover, the atom-economic nature of the reaction, which incorporates most of the reactant atoms into the final product, results in lower E-factors and reduced waste generation. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, making the process easier to permit and operate in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and mechanistic understanding presented in the patent documentation, providing clarity for technical teams evaluating the feasibility of adoption. Understanding these nuances is essential for accurate process modeling and risk assessment.
Q: What is the primary advantage of using nitroarenes in this synthesis?
A: Nitroarenes serve as both the nitrogen source and the oxidant, eliminating the need for pre-functionalized amines and simplifying the supply chain for raw materials.
Q: How does molybdenum carbonyl function in this reaction system?
A: Molybdenum carbonyl acts as a solid carbon monoxide source and a reducing agent, avoiding the safety hazards associated with handling high-pressure CO gas cylinders.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of stable solid reagents and standard solvents like 1,4-dioxane makes the process highly scalable and compatible with existing industrial reactor infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isochroman Amides Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed technology for the production of next-generation pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including the safe management of molybdenum residues and the efficient recovery of precious palladium catalysts. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of isochroman amide meets the highest international standards for impurity profiles and assay potency.
We invite you to collaborate with us to leverage this innovative synthetic route for your specific drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this method can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data for similar structures and comprehensive route feasibility assessments. Let us help you accelerate your timeline to market with a reliable, cost-effective, and scalable supply solution.
