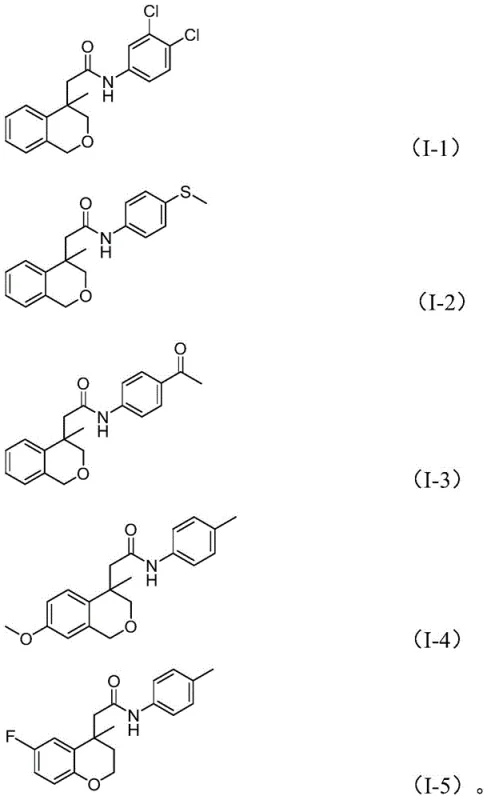
Advanced Palladium-Catalyzed Synthesis of Isochroman Amides for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently. Patent CN114539198A introduces a groundbreaking preparation method for amide compounds containing (iso)chroman structures, which are privileged motifs in numerous bioactive molecules and drug candidates. This technology leverages a sophisticated palladium-catalyzed cascade reaction that merges cyclocarbopalladation with aminocarbonylation in a single operational sequence. By utilizing nitroarenes as the nitrogen source and molybdenum hexacarbonyl as both the carbonyl source and reducing agent, the process circumvents many traditional synthetic bottlenecks. For R&D directors and process chemists, this represents a significant leap forward in atom economy and step reduction, offering a streamlined pathway to access high-value intermediates that were previously costly or difficult to manufacture on a large scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of amide-functionalized isochromans has relied on multi-step sequences involving the separate preparation of carboxylic acid derivatives and amines, followed by coupling reactions. These classical approaches often necessitate the use of hazardous coupling reagents, generate substantial stoichiometric waste, and require stringent moisture-free conditions. Furthermore, conventional carbonylation strategies typically depend on high-pressure carbon monoxide gas, which poses severe safety risks and requires specialized high-pressure autoclaves that are not universally available in standard pilot plants. The reliance on pre-functionalized amines also limits the scope, as many amines are unstable, expensive, or commercially unavailable, creating supply chain vulnerabilities and inflating the cost of goods for the final active pharmaceutical ingredient.
The Novel Approach
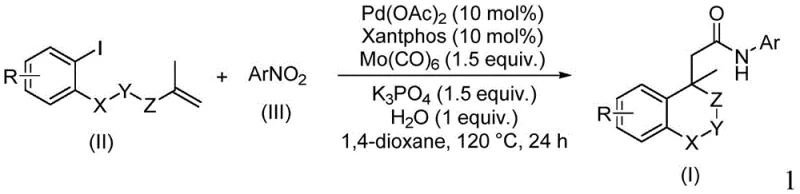
The methodology disclosed in the patent revolutionizes this landscape by employing a tandem catalytic cycle that constructs the isochroman ring and the amide bond simultaneously. Starting from readily available iodoarenes and nitroarenes, the reaction proceeds through an intramolecular Heck-type cyclization followed by CO insertion and in-situ reduction of the nitro group. This strategy not only simplifies the operational procedure to a simple mix-and-heat protocol but also dramatically expands the substrate scope. As illustrated in the specific examples below, the system tolerates a wide array of substituents, enabling the rapid generation of diverse chemical libraries for drug discovery without the need for complex protecting group manipulations.
Mechanistic Insights into Pd-Catalyzed Cyclocarbopalladation/Aminocarbonylation
The core of this transformation lies in the elegant interplay between the palladium catalyst and the molybdenum carbonyl complex. The reaction initiates with the oxidative addition of the aryl iodide to the Pd(0) species, generated in situ from palladium acetate and the Xantphos ligand. This is followed by a migratory insertion into the pendant alkene, forming a critical sigma-alkyl-palladium intermediate that establishes the isochroman core. Subsequently, carbon monoxide, released thermally from the molybdenum hexacarbonyl, inserts into the palladium-carbon bond to form an acyl-palladium species. Concurrently, the nitroarene undergoes reduction, likely facilitated by the molybdenum species and water, generating the reactive amine nucleophile in situ which then attacks the acyl-palladium complex to release the final amide product and regenerate the catalyst.
From an impurity control perspective, this mechanism offers distinct advantages. The use of a solid CO source like Mo(CO)6 ensures a slow, steady release of carbon monoxide, preventing the accumulation of high concentrations that could lead to side reactions such as homocoupling or over-carbonylation. Additionally, the in-situ generation of the amine from the nitro precursor avoids the handling of free amines which can sometimes lead to oligomerization or degradation issues. The presence of water in the system is crucial for the reduction step but is carefully balanced to prevent hydrolysis of the sensitive intermediates, ensuring a clean reaction profile that simplifies downstream purification and enhances the overall purity of the crude product.
How to Synthesize Isochroman Amides Efficiently
To implement this synthesis effectively, precise control over reaction parameters is essential to maximize yield and minimize byproduct formation. The process involves charging a sealed vessel with the iodoarene substrate, the nitroarene coupling partner, and the catalytic system comprising palladium acetate and Xantphos. Molybdenum hexacarbonyl is added as the carbonyl source along with potassium phosphate as the base and a stoichiometric amount of water. The mixture is suspended in 1,4-dioxane and heated to 120°C for approximately 24 hours. Detailed standardized synthesis steps for this protocol are provided in the guide below.
- Combine palladium acetate, Xantphos ligand, molybdenum hexacarbonyl, potassium phosphate, water, iodoarene substrate, and nitroarene in 1,4-dioxane solvent.
- Heat the reaction mixture to 120°C and maintain stirring for 24 hours to allow for cyclocarbopalladation and subsequent carbonyl insertion.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target isochroman amide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits regarding cost stability and operational safety. The shift away from gaseous carbon monoxide to a solid carbonyl source eliminates the need for expensive high-pressure infrastructure and reduces regulatory compliance burdens associated with toxic gas storage. Furthermore, the reliance on nitroarenes, which are commodity chemicals with stable pricing and abundant global supply, mitigates the risk of raw material shortages that often plague projects dependent on specialized, custom-synthesized amines. This robustness ensures a more predictable manufacturing timeline and protects against market volatility.
- Cost Reduction in Manufacturing: The elimination of external CO gas cylinders and high-pressure reactors significantly lowers capital expenditure and operational overhead. Additionally, the use of inexpensive nitroarenes instead of costly amines reduces the raw material bill of materials. The one-pot nature of the reaction minimizes solvent usage and labor hours associated with intermediate isolation, leading to substantial overall cost savings in the production of these complex intermediates.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials such as iodoarenes and nitroarenes, the supply chain becomes less fragile. These precursors are produced on a multi-ton scale globally, ensuring consistent availability. The simplified process flow also reduces the number of unit operations, decreasing the likelihood of batch failures and ensuring a steady, reliable output of high-quality intermediates for downstream API synthesis.
- Scalability and Environmental Compliance: The reaction conditions are mild enough to be scaled in standard glass-lined or stainless steel reactors without requiring exotic metallurgy for high-pressure containment. The atom-economic nature of the carbonylation reduces waste generation, aligning with green chemistry principles. This facilitates easier environmental permitting and waste disposal management, making the process highly attractive for large-scale commercial manufacturing under strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and mechanistic understanding presented in the patent documentation, providing clarity for technical teams evaluating this route for their specific projects.
Q: What is the advantage of using nitroarenes over amines in this synthesis?
A: Nitroarenes serve as stable, inexpensive, and readily available nitrogen sources. Unlike sensitive amines, they do not require strict anhydrous conditions for storage and handling, simplifying the supply chain and reducing raw material costs significantly.
Q: How does the process handle carbon monoxide safety concerns?
A: The process utilizes molybdenum hexacarbonyl (Mo(CO)6) as a solid carbonyl source. This eliminates the need for high-pressure CO gas cylinders and specialized gas handling infrastructure, drastically improving operational safety and facilitating easier scale-up in standard reactors.
Q: What is the functional group tolerance of this catalytic system?
A: The palladium/Xantphos system demonstrates excellent tolerance for various functional groups including halogens (F, Cl, Br), ethers, thioethers, and ketones. This allows for the direct synthesis of complex intermediates without extensive protecting group strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isochroman Amides Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in accelerating drug development timelines. Our team of expert process chemists has extensively evaluated the technology described in CN114539198A and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. Our state-of-the-art facilities are designed to handle complex catalytic reactions safely, and our rigorous QC labs enforce stringent purity specifications to guarantee the quality of every batch.
We invite you to collaborate with us to leverage this advanced chemistry for your next program. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce your overall cost of goods.
