Advanced Palladium-Catalyzed Synthesis of C-3 Benzyl Indolizine Compounds for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of C-3 Benzyl Indolizine Compounds for Commercial Scale-up
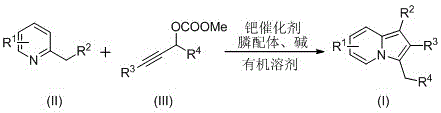
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN110357879B, published in April 2021, introduces a highly efficient preparation method for C-3 benzyl indolizine compounds, a structural motif found in potent antitumor agents and phosphodiesterase inhibitors. This technology represents a significant leap forward in synthetic methodology, moving away from harsh, low-yielding traditional processes toward a streamlined palladium-catalyzed cyclization. By utilizing readily available starting materials such as 2-alkylpyridines and propargyl carbonates, this invention addresses critical pain points regarding operational complexity and substrate scope. For R&D directors and procurement managers alike, understanding the nuances of this patented process is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity building blocks for next-generation drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of C-3 benzyl indolizine compounds has been fraught with significant technical challenges that hinder efficient commercial production. Prior art methods, such as the one-step reaction of 2-vinylpyridine with chlorocarbene under heating or illumination conditions, are severely limited by their narrow substrate scope and inconsistent outcomes. These conventional pathways often struggle with poor applicability when diverse substituent groups are introduced, leading to unpredictable reaction profiles and difficult purification processes. Furthermore, the reliance on reactive carbene intermediates can pose safety hazards and necessitates stringent control measures that increase operational costs. The low yields associated with these older methodologies result in substantial material waste, making them economically unviable for large-scale manufacturing where cost reduction in API manufacturing is a primary objective. Consequently, there has been a persistent demand for a more versatile and reliable synthetic strategy that can accommodate structural diversity without compromising efficiency.
The Novel Approach
The innovative method disclosed in CN110357879B overcomes these historical barriers through a sophisticated palladium-catalyzed annulation strategy. This novel approach involves the reaction of 2-alkylpyridines with propargyl carbonates in the presence of a metal palladium catalyst, a phosphine ligand, and a base within an organic solvent. The process operates at elevated temperatures ranging from 100°C to 140°C, ensuring complete conversion of starting materials into the desired C-3 benzyl indolizine framework. Unlike the restrictive nature of previous techniques, this method exhibits strong substrate designability, allowing chemists to tailor the R-groups on both the pyridine and alkyne components to access a wide library of analogues. The use of stable propargyl carbonates instead of hazardous carbenes significantly enhances process safety and ease of handling. Moreover, the post-treatment procedure is remarkably simple, involving basic filtration and column chromatography, which streamlines the workflow and reduces the time required to obtain high-purity products suitable for biological testing.
Mechanistic Insights into Palladium-Catalyzed Cyclization
The success of this transformation relies heavily on the precise interplay between the zero-valent palladium catalyst and the specialized phosphine ligands. The catalytic cycle likely initiates with the oxidative addition of the propargyl carbonate to the Pd(0) species, generating a reactive pi-allyl palladium intermediate. This electrophilic complex then undergoes nucleophilic attack by the nitrogen atom of the 2-alkylpyridine, forming a key C-N bond. Subsequent intramolecular cyclization and deprotonation steps, facilitated by the inorganic base such as potassium carbonate or cesium carbonate, lead to the formation of the indolizine ring system. The choice of ligand is critical; bulky, electron-rich bidentate phosphines like 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene (Xantphos derivatives) stabilize the active palladium species and promote the reductive elimination step that releases the final product. This mechanistic pathway ensures high turnover numbers and minimizes the formation of side products, which is crucial for maintaining the integrity of the impurity profile in pharmaceutical intermediates.
From an impurity control perspective, the specific stoichiometry employed in this patent plays a vital role in suppressing unwanted byproducts. The protocol recommends using an excess of 2-alkylpyridine relative to the propargyl carbonate, typically in a molar ratio of 2:1 to 4:1. This excess drives the equilibrium towards product formation and prevents the homocoupling of the alkyne substrate, a common side reaction in palladium chemistry. Additionally, the selection of polar aprotic solvents like dimethyl sulfoxide (DMSO) or N,N-dimethylformamide (DMF) not only ensures excellent solubility of all reaction components but also stabilizes the charged intermediates involved in the cyclization. By optimizing the catalyst loading to between 0.05 and 0.15 mmol per mmol of substrate, the process achieves a balance between reaction rate and cost efficiency, avoiding the accumulation of palladium residues that could complicate downstream purification. This level of mechanistic control is what distinguishes a lab-scale curiosity from a commercially viable manufacturing process.
How to Synthesize C-3 Benzyl Indolizine Efficiently
Implementing this synthesis requires careful attention to the specific reaction parameters outlined in the patent to ensure reproducibility and high yield. The process begins by combining the metal catalyst, phosphine ligand, 2-alkylpyridine, propargyl carbonate, and base in a suitable reactor equipped with heating and stirring capabilities. The mixture is then heated to the specified temperature range and maintained for a duration of 10 to 20 hours, depending on the specific substrates used. Monitoring the reaction progress is essential to determine the optimal endpoint, preventing over-reaction which could lead to decomposition. Once the reaction is deemed complete, the workup involves a straightforward filtration to remove insoluble salts, followed by adsorption onto silica gel and purification via column chromatography using ethyl acetate and petroleum ether mixtures. For detailed operational specifics and exact stoichiometric ratios tailored to your specific target molecule, please refer to the standardized synthesis steps provided below.
- Charge a reaction vessel with metal palladium catalyst, phosphine ligand, 2-alkylpyridine, propargyl carbonate, and base in an organic solvent such as DMSO or DMF.
- Heat the reaction mixture to a temperature between 100°C and 140°C and maintain stirring for 10 to 20 hours until the reaction is complete.
- Perform post-treatment by filtering the mixture, mixing with silica gel, and purifying via column chromatography to isolate the target C-3 benzyl indolizine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers distinct strategic advantages over legacy synthesis routes. The primary benefit lies in the accessibility and cost-effectiveness of the raw materials; 2-alkylpyridines are commodity chemicals available in bulk quantities, and propargyl carbonates are easily synthesized or sourced from established suppliers. This abundance of starting materials mitigates the risk of supply chain disruptions that often plague projects relying on exotic or custom-synthesized reagents. Furthermore, the simplified post-processing workflow eliminates the need for complex extraction sequences or specialized equipment, directly translating to reduced labor costs and faster batch turnover times. The ability to use common polar solvents also simplifies solvent recovery and recycling protocols, aligning with modern environmental compliance standards and reducing waste disposal expenses. These factors collectively contribute to a more resilient and cost-efficient supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive, commercially available starting materials and the elimination of hazardous reagents like chlorocarbene. By avoiding the need for specialized containment systems required for toxic gases, capital expenditure on facility upgrades is significantly minimized. Additionally, the high conversion rates achieved through optimized catalyst loading mean that less raw material is wasted, improving the overall atom economy of the synthesis. The straightforward purification via column chromatography, while standard in R&D, can be adapted to crystallization or distillation on scale, further driving down the cost of goods sold. This efficiency allows for competitive pricing strategies when sourcing high-purity indolizine derivatives for drug development pipelines.
- Enhanced Supply Chain Reliability: Supply continuity is paramount in the pharmaceutical sector, and this method enhances reliability by utilizing robust chemistry that is less sensitive to minor fluctuations in reaction conditions. The broad substrate scope means that if a specific substituted pyridine becomes unavailable, alternative analogues can often be synthesized using the same platform technology without re-optimizing the entire process. The use of stable solid bases like potassium carbonate or cesium carbonate ensures that the reaction is not dependent on sensitive liquid reagents that may degrade over time. This stability allows for longer shelf-life of pre-mixed reagent kits and simplifies inventory management. Consequently, manufacturers can maintain consistent production schedules and meet tight delivery deadlines for clinical trial materials.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram quantities is facilitated by the use of high-boiling polar solvents which maintain homogeneity even at high concentrations. The reaction temperature range of 100°C to 140°C is well within the operating limits of standard glass-lined or stainless steel reactors, removing the need for cryogenic cooling or high-pressure equipment. From an environmental standpoint, the absence of halogenated solvents in the reaction medium and the potential for solvent recycling aligns with green chemistry principles. The simplified workup reduces the volume of aqueous waste generated, lowering the burden on wastewater treatment facilities. These attributes make the process highly attractive for contract development and manufacturing organizations (CDMOs) looking to expand their portfolio of sustainable heterocyclic synthesis capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These answers are derived directly from the experimental data and beneficial effects described in patent CN110357879B, providing clarity on the practical application of this technology. Understanding these details helps stakeholders assess the feasibility of integrating this route into their existing manufacturing workflows. Whether you are concerned about catalyst removal, substrate compatibility, or regulatory compliance, the insights below offer a foundational understanding of the process capabilities.
Q: What are the key advantages of this Pd-catalyzed method over traditional synthesis?
A: Unlike traditional methods involving chlorocarbene which suffer from poor substituent applicability and low yields, this novel approach utilizes readily available 2-alkylpyridines and propargyl carbonates under mild conditions, resulting in significantly improved operational simplicity and substrate design flexibility.
Q: Which catalysts and ligands are optimal for this transformation?
A: The patent specifies zero-valent palladium sources such as tris(dibenzylideneacetone)dipalladium or tetrakis(triphenylphosphine)palladium, paired with bidentate phosphine ligands like 4,6-bis(diphenylphosphino)dibenzofuran or 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene to ensure high conversion rates.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses common polar solvents like DMSO and DMF which effectively dissolve raw materials, and the reaction temperatures of 100-140°C are easily manageable in standard industrial reactors, facilitating straightforward commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C-3 Benzyl Indolizine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We have rigorously evaluated the methodology described in CN110357879B and confirmed its potential for generating high-purity indolizine derivatives with stringent purity specifications. Our state-of-the-art rigorous QC labs are equipped to analyze complex impurity profiles, guaranteeing that every batch meets the exacting standards required by global regulatory bodies. By leveraging our expertise in palladium catalysis and heterocyclic chemistry, we can deliver customized solutions that optimize both yield and cost for your specific application.
We invite you to collaborate with us to explore the full potential of this advanced synthetic route for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can reduce your overall expenditure. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us be your partner in transforming innovative chemistry into commercial reality, ensuring a seamless supply of critical building blocks for your pharmaceutical pipeline.
