Advanced Palladium-Catalyzed Synthesis of C-3 Benzyl Indolizine Compounds for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of C-3 Benzyl Indolizine Compounds for Commercial Scale-Up
The pharmaceutical industry continuously seeks efficient, scalable routes to nitrogen-containing heterocycles due to their prevalence in bioactive molecules. A significant breakthrough in this domain is detailed in patent CN110357879B, which discloses a novel preparation method for C-3 benzyl indolizine compounds. These scaffolds are critical structural motifs found in potent anticancer agents and phosphodiesterase inhibitors, demonstrating the high value of this chemical class in drug discovery. The disclosed methodology leverages a palladium-catalyzed annulation strategy that overcomes the limitations of traditional synthetic routes, offering a pathway that is both operationally simple and chemically robust. By utilizing readily available 2-alkylpyridines and propargyl carbonates, this process enables the rapid construction of complex indolizine cores with high atom economy. For R&D directors and procurement managers alike, understanding the nuances of this technology is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials for downstream API synthesis.
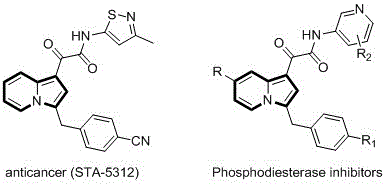
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of C-3 benzyl indolizine compounds has been fraught with significant challenges that hinder large-scale manufacturing and cost-effective production. Prior art methods, such as the reaction of 2-vinylpyridine with chlorocarbene under heating or irradiation conditions, suffer from poor applicability regarding substituent groups. These conventional routes often require harsh reaction conditions that can degrade sensitive functional groups, leading to complex impurity profiles that are difficult to separate. Furthermore, the yields associated with these older methodologies are frequently low, resulting in substantial material loss and increased waste generation. From a supply chain perspective, the reliance on unstable or hazardous reagents like chlorocarbene introduces safety risks and regulatory hurdles that complicate the logistics of commercial scale-up of complex pharmaceutical intermediates. The inability to easily diversify the substrate scope also limits the utility of these methods in medicinal chemistry campaigns where rapid analog synthesis is required.
The Novel Approach
In stark contrast, the method described in patent CN110357879B represents a paradigm shift towards greener and more efficient synthesis. This novel approach employs a transition metal-catalyzed cyclization between 2-alkylpyridines and propargyl carbonates, facilitated by a zero-valent palladium catalyst and specific phosphine ligands. The reaction proceeds smoothly in polar organic solvents such as dimethyl sulfoxide (DMSO) or N,N-dimethylformamide (DMF) at moderate temperatures ranging from 100°C to 140°C. This methodology not only tolerates a wide variety of functional groups—including cyano, ester, and phenyl substituents—but also delivers the target indolizine products in significantly improved yields. The operational simplicity is a key advantage; the reaction mixture can be directly filtered and purified via standard column chromatography, eliminating the need for complex workup procedures. This streamlined process directly addresses the need for cost reduction in pharmaceutical intermediate manufacturing by minimizing unit operations and maximizing throughput.
Mechanistic Insights into Palladium-Catalyzed Cyclization
The success of this synthetic route lies in the intricate interplay between the palladium catalyst and the phosphine ligand, which orchestrates the formation of the indolizine ring system through a well-defined catalytic cycle. The reaction initiates with the oxidative addition of the zero-valent palladium species into the propargyl carbonate, generating a reactive pi-allyl palladium intermediate. This electrophilic species is then intercepted by the nucleophilic 2-alkylpyridine substrate, likely through a C-H activation or nucleophilic attack mechanism depending on the specific electronic nature of the pyridine ring. The presence of bulky, electron-rich phosphine ligands, such as 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene (Xantphos derivatives), is crucial for stabilizing the active catalytic species and promoting the subsequent migratory insertion step. This step effectively constructs the new carbon-carbon bonds required to close the five-membered ring of the indolizine core. Finally, reductive elimination releases the desired C-3 benzyl indolizine product and regenerates the active palladium(0) catalyst, allowing the cycle to continue. Understanding this mechanism allows chemists to fine-tune reaction parameters for optimal performance.
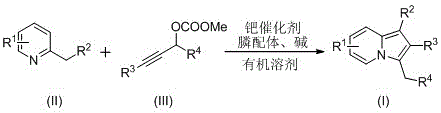
Controlling the impurity profile is paramount for any process intended for pharmaceutical applications, and this catalytic system offers inherent advantages in selectivity. The use of specific ligand-catalyst combinations ensures high regioselectivity, directing the cyclization exclusively to the C-3 position of the indolizine ring. This precision minimizes the formation of regioisomers that would otherwise require costly and yield-reducing separation steps. Furthermore, the mild basic conditions employed, typically using potassium carbonate or cesium carbonate, prevent the decomposition of base-sensitive functionalities that might be present on the substrate. The reaction conditions are optimized to balance conversion rates with product stability; extending the reaction time beyond 20 hours or exceeding 140°C can lead to increased costs and potential degradation, while insufficient time compromises completion. By adhering to the specified molar ratios—typically a slight excess of the 2-alkylpyridine relative to the propargyl carbonate—the process drives the equilibrium towards the product, ensuring high purity and reducing the burden on downstream purification teams.
How to Synthesize C-3 Benzyl Indolizine Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction monitoring to achieve the reported yields of up to 67%. The process begins by charging a reaction vessel with the metal palladium catalyst, the chosen phosphine ligand, the 2-alkylpyridine substrate, the propargyl carbonate, and a suitable inorganic base. These components are suspended or dissolved in an organic solvent, with DMSO and DMF proving to be the most effective media for solubilizing the reactants and promoting the catalytic cycle. The detailed standardized synthesis steps, including specific molar equivalents and purification protocols, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- Combine metal palladium catalyst, phosphine ligand, 2-alkylpyridine, propargyl carbonate, and base in an organic solvent such as DMSO or DMF.
- Heat the reaction mixture to a temperature range of 100-140°C and maintain stirring for 10 to 20 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography using ethyl acetate and petroleum ether to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible benefits that extend beyond mere chemical yield. The shift from hazardous, multi-step traditional syntheses to this direct, one-pot catalytic process fundamentally alters the cost structure and risk profile of producing these valuable intermediates. By simplifying the synthetic route, manufacturers can reduce the number of processing units required, lower energy consumption, and minimize the volume of solvent waste generated. These factors collectively contribute to a more sustainable and economically viable supply chain, ensuring that critical drug candidates can be advanced without being bottlenecked by material availability or prohibitive costs.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like chlorocarbene, replaced by stable and commercially available propargyl carbonates, leads to substantial raw material cost savings. Additionally, the high efficiency of the palladium catalyst, used in low loading amounts (0.05-0.15 mmol per mmol of substrate), reduces the financial burden associated with precious metal usage. The simplified post-processing workflow, which avoids complex extractions or recrystallizations in favor of direct filtration and chromatography, further lowers labor and utility costs. These cumulative efficiencies result in a significantly reduced cost of goods sold (COGS), allowing for more competitive pricing in the global market for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of this reaction conditions means that production is less susceptible to variations in raw material quality or minor fluctuations in process parameters. The use of common, off-the-shelf solvents and bases ensures that supply disruptions are unlikely, providing a stable foundation for long-term manufacturing contracts. Furthermore, the broad substrate scope demonstrated in the patent allows for the flexible production of various analogs without needing to revalidate entirely new processes. This flexibility is crucial for maintaining continuity of supply in the face of changing clinical demands, effectively reducing lead time for high-purity pharmaceutical intermediates needed for urgent drug development programs.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or tonne scale is straightforward due to the absence of extreme pressures or temperatures. The reaction operates safely within standard glass-lined or stainless steel reactors, facilitating easy technology transfer from R&D to commercial production. From an environmental standpoint, the improved atom economy and reduced waste generation align with modern green chemistry principles, helping companies meet increasingly stringent regulatory requirements. The ability to recycle solvents and the minimized use of toxic reagents further enhance the environmental profile of the manufacturing process, making it an attractive option for companies committed to sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is vital for assessing the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the optimal reaction conditions for this synthesis?
A: The patent specifies heating the reaction mixture to 100-140°C for 10-20 hours in polar solvents like DMSO or DMF, using a zero-valent palladium catalyst and a phosphine ligand.
Q: Which catalysts and ligands provide the best results?
A: Tris(dibenzylideneacetone)dipalladium or tetrakis(triphenylphosphine)palladium are preferred catalysts, paired with ligands such as 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene for optimal yield.
Q: Is this method scalable for industrial production?
A: Yes, the method utilizes inexpensive and readily available raw materials with simple post-processing steps like filtration and column chromatography, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C-3 Benzyl Indolizine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN110357879B for the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the bench to the plant. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of specific indolizine derivatives or large-scale supply of standard intermediates, our infrastructure is designed to support your timeline and quality requirements with unwavering reliability.
We invite you to engage with our technical sales team to discuss how this advanced palladium-catalyzed technology can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this superior manufacturing method. We encourage potential partners to contact our technical procurement team to obtain specific COA data and route feasibility assessments tailored to your unique molecular targets. Let us collaborate to accelerate your drug development journey with high-quality, cost-effective chemical solutions.
