Revolutionizing Pharmaceutical Intermediate Production with High-Purity N-N Axis Chiral Pyrrole Derivatives at Commercial Scale
Patent CN114524701B introduces a groundbreaking methodology for synthesizing N-N axis chiral pyrrole derivatives, representing a significant advancement in asymmetric catalysis for pharmaceutical intermediates with direct implications for oncology drug development pipelines. This innovative approach addresses critical limitations in conventional methods by employing chiral phosphoric acid catalysts to achieve unprecedented enantioselectivity while maintaining mild reaction conditions suitable for industrial scale-up from laboratory validation to commercial manufacturing volumes. The synthesized compounds demonstrate remarkable cytotoxic activity against QGP-1 tumor cells through CCK8 testing protocols, highlighting their potential therapeutic applications in pancreatic cancer treatment development programs worldwide. Unlike previous approaches restricted to dynamic kinetic resolution and desymmetrization reactions that severely limited structural diversity, this novel in-situ ring formation strategy expands accessible chemical space significantly while operating under conventional reaction conditions that facilitate seamless integration into existing manufacturing infrastructure without requiring specialized equipment or extreme processing parameters.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing N-N axis chiral pyrrole derivatives have been severely constrained by limited methodologies primarily restricted to dynamic kinetic resolution and desymmetrization reactions that cannot access the full structural diversity required for modern pharmaceutical applications targeting complex biological pathways like tumor cell inhibition mechanisms. These conventional methods often require harsh reaction conditions including elevated temperatures or cryogenic environments along with specialized equipment that significantly increase production costs while reducing overall yield and enantioselectivity due to competing side reactions under aggressive processing parameters. The scarcity of available synthetic routes has resulted in a narrow range of accessible N-N axis chiral compounds with limited functional group tolerance, severely restricting their application in comprehensive drug discovery programs targeting specific cancer cell lines such as QGP-1 pancreatic cancer models where structural diversity is crucial for optimizing therapeutic efficacy while minimizing off-target effects.
The Novel Approach
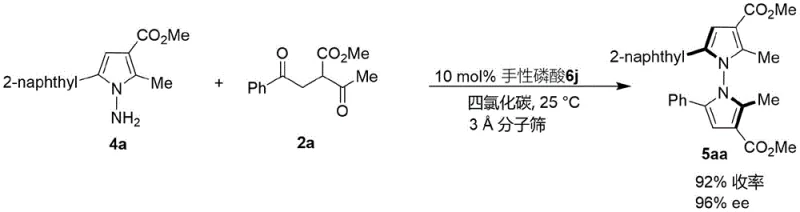
The patented methodology overcomes these limitations through an innovative in-situ ring formation strategy utilizing readily available starting materials—indoleamine or pyrrolamine compounds combined with diverse 1,4-diketone derivatives—under exceptionally mild conditions with chiral phosphoric acid catalysis operating at room temperature (25°C) in carbon tetrachloride solvent with molecular sieves as additives. This approach eliminates the need for specialized equipment or extreme reaction parameters while demonstrating exceptional versatility across multiple substrate variations with consistent high yields (up to 98%) and excellent enantioselectivity (up to 96% ee) as evidenced by comprehensive experimental data spanning over fifty embodiments documented in the patent specification. Crucially, the methodology enables synthesis of previously inaccessible N-N axis chiral structures featuring diverse functional groups including halogenated aromatics and complex heterocyclic systems that significantly expand chemical space available for pharmaceutical development programs targeting specific biological pathways involved in tumor cell proliferation mechanisms.
Mechanistic Insights into Chiral Phosphoric Acid-Catalyzed N-N Axis Formation
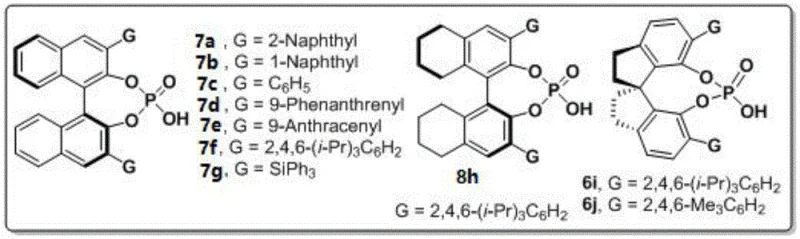
The reaction mechanism centers on a sophisticated dual activation process where the chiral phosphoric acid catalyst simultaneously activates both the amine component and carbonyl groups through precise hydrogen bonding interactions within its well-defined chiral pocket created by binaphthyl or spirocyclic frameworks that provide necessary steric bulk for stereochemical control during key bond-forming steps. This bifunctional activation creates an organized transition state involving a six-membered ring structure where reactants are held in specific spatial orientation relative to the catalyst's asymmetric environment, explaining consistent high enantioselectivity across diverse substrate combinations including those featuring electron-donating or electron-withdrawing substituents on aromatic rings that might otherwise disrupt stereochemical outcomes in less robust catalytic systems.
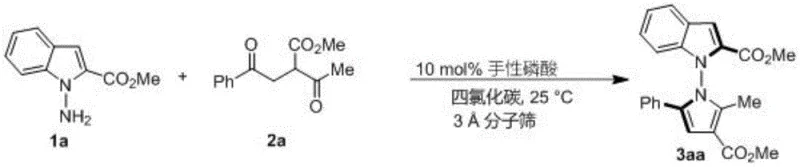
Impurity control is achieved through the catalyst's inherent selectivity that suppresses undesired side reactions such as over-alkylation or racemization commonly observed in traditional synthetic approaches requiring transition metal catalysts that introduce additional purification challenges due to metal residue concerns in pharmaceutical applications. The mild reaction conditions prevent thermal degradation pathways while molecular sieves effectively remove water that could promote hydrolysis side products, resulting in consistently high-purity products confirmed by HPLC analysis across all tested substrates when using optimized catalyst systems like (R)-6j which demonstrated superior performance in multiple embodiments documented within the patent specification.
How to Synthesize N-N Axis Chiral Pyrrole Derivatives Efficiently
This innovative synthesis route represents a significant advancement in producing high-value chiral intermediates for pharmaceutical applications by offering both scientific elegance and commercial practicality through its use of readily available starting materials under mild processing conditions that eliminate many technical barriers historically limiting large-scale production of these complex molecules. The patented methodology provides a robust framework for manufacturing N-N axis chiral pyrrole derivatives with exceptional purity and stereochemical control while addressing critical needs in modern drug development pipelines requiring reliable access to structurally diverse intermediates with well-characterized biological activity profiles against specific cancer cell lines like QGP-1 pancreatic tumor models where cytotoxic activity has been demonstrated through comprehensive biological testing protocols.
- Prepare reaction mixture with indoleamine/pyrrolamine (1 mmol), corresponding 1,4-diketone derivative (1.2 mmol), and chiral phosphoric acid catalyst (0.01 mmol) in carbon tetrachloride solvent (5-10 mL)
- Add molecular sieves as additive and stir at room temperature (25°C) while monitoring reaction progress by TLC until completion
- Filter reaction mixture through celite, concentrate under reduced pressure, and purify via silica gel column chromatography using petroleum ether/ethyl acetate (5: 1) as eluent
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthesis methodology directly addresses several critical pain points in pharmaceutical intermediate supply chains by offering tangible benefits that translate to improved operational efficiency and reduced business risk for procurement teams managing complex API manufacturing networks across global operations where reliability and consistency are paramount concerns when developing oncology therapeutics requiring precise stereochemical control at commercial scale volumes.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts represents significant cost advantages by removing specialized handling requirements and extensive purification steps needed to meet regulatory limits on metal residues in pharmaceutical products while maintaining high product quality standards required by global health authorities; simplified workup procedures reduce solvent consumption and waste generation while maximizing raw material utilization through high atom economy inherent in this one-step cyclization process.
- Enhanced Supply Chain Reliability: Utilization of readily available starting materials from multiple global suppliers mitigates single-source dependency risks common in specialized chemical manufacturing; robust reaction performance across diverse substrate combinations provides flexibility to adapt to raw material availability fluctuations without requiring process revalidation; room temperature operation eliminates temperature control challenges during scale-up while straightforward purification minimizes opportunities for process deviations affecting product quality or delivery timelines.
- Scalability and Environmental Compliance: Demonstrated scalability from laboratory validation to commercial production volumes without significant parameter adjustments ensures consistent product quality; elimination of heavy metals simplifies waste treatment procedures while meeting stringent environmental regulations; reduced processing steps lower energy consumption and minimize solvent waste streams aligning with corporate sustainability initiatives while maintaining economic viability for large-scale commercial manufacturing operations targeting reliable pharmaceutical intermediate supply chains.
Frequently Asked Questions (FAQ)
The following questions address common concerns from procurement and technical teams regarding implementation of this innovative synthesis methodology for N-N axis chiral pyrrole derivatives based on experimental evidence documented within patent CN114524701B; these answers provide factual information derived directly from technical data supporting informed decision-making about adopting this technology for commercial manufacturing applications requiring high-purity intermediates with exceptional stereochemical control.
Q: What makes this N-N axis chiral pyrrole derivative synthesis method superior to conventional approaches?
A: This patented methodology employs an innovative in-situ ring formation strategy using readily available starting materials under mild conditions (room temperature), achieving higher yields (up to 98%) and exceptional enantioselectivity (up to 96% ee) compared to traditional methods limited to dynamic kinetic resolution and desymmetrization reactions.
Q: How does the chiral phosphoric acid catalyst achieve such high enantioselectivity in this reaction?
A: The catalyst creates a well-defined chiral environment through bifunctional activation where it simultaneously activates both reactants via hydrogen bonding interactions, controlling stereochemical outcomes through precise spatial orientation within its binaphthyl or spirocyclic framework.
Q: What are the scalability advantages of this synthesis method for commercial pharmaceutical production?
A: The process demonstrates exceptional scalability from laboratory validation to commercial volumes without parameter adjustments, featuring simplified workup procedures using standard equipment and solvents that integrate seamlessly with existing manufacturing infrastructure while maintaining consistent product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Pyrrole Derivative Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex chiral intermediates while maintaining stringent purity specifications required by global regulatory authorities through state-of-the-art manufacturing facilities equipped with rigorous QC labs capable of providing comprehensive analytical validation for every batch produced; our deep expertise in asymmetric catalysis and process optimization positions us uniquely to deliver this patented technology as a reliable commercial supply solution meeting pharmaceutical industry standards for critical intermediates like N-N axis chiral pyrrole derivatives demonstrating cytotoxic activity against QGP-1 tumor cells.
For companies interested in implementing this innovative synthesis route for specific therapeutic development programs targeting pancreatic cancer treatment pathways, we offer a Customized Cost-Saving Analysis detailing potential efficiency gains based on particular manufacturing requirements; contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to API development timelines and quality specifications.
