Innovative Synthesis of N-N Axis Chiral Pyrrole Derivatives for Commercial API Manufacturing Scale-Up
Patent CN114524701B introduces a novel synthetic route for N-N axis chiral pyrrole derivatives, demonstrating significant potential for pharmaceutical applications through its ability to produce compounds with high sensitivity and strong cytotoxic activity against QGP-1 tumor cells. This breakthrough methodology employs chiral phosphoric acid catalysis under mild reaction conditions to achieve exceptional enantioselectivity while addressing critical challenges in the production of complex chiral intermediates for oncology drug development.
Overcoming Traditional Limitations in Chiral Pyrrole Synthesis
The Limitations of Conventional Methods
Traditional approaches to synthesizing N-N axis chiral pyrrole derivatives have been severely constrained by limited methodologies, primarily restricted to dynamic kinetic resolution and desymmetrization reactions that cannot access the full structural diversity required for comprehensive drug discovery programs. These conventional techniques often require harsh reaction conditions including elevated temperatures or pressures that increase operational risks while generating complex impurity profiles requiring extensive purification. The scarcity of viable synthetic routes has hindered exploration of these compounds' biological activities, particularly their potential as anticancer agents against pancreatic cancer cells like QGP-1. Furthermore, existing methods typically produce racemic mixtures that necessitate costly separation processes, significantly increasing production costs while reducing overall process efficiency and limiting commercial viability for large-scale pharmaceutical manufacturing.
The Novel Approach
The patented methodology (CN114524701B) represents a paradigm shift by introducing an in-situ ring formation strategy that directly constructs the N-N axis chiral framework through a single catalytic step using readily available starting materials. This innovative approach utilizes 1,4-diketone derivatives and indoleamine or pyrrole amine precursors under exceptionally mild conditions at room temperature with carbon tetrachloride as solvent and molecular sieves as additives, eliminating the need for multi-step sequences and intermediate isolations. The chiral phosphoric acid catalyst enables exceptional enantioselectivity (up to 96% ee) by creating a well-defined chiral environment through precise molecular recognition mechanisms. This streamlined process not only expands structural diversity but also demonstrates excellent scalability potential while maintaining consistent product quality across diverse substrate combinations as evidenced by comprehensive experimental data.
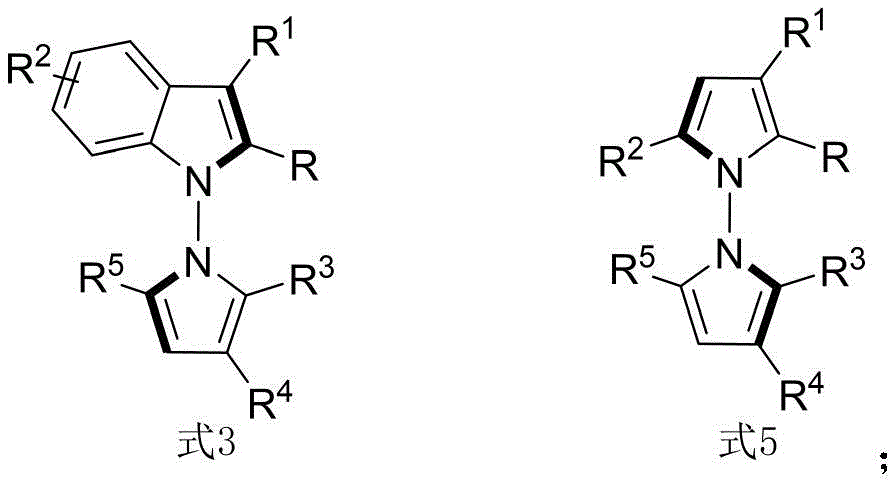
Advanced Reaction Mechanism and Purity Control
The core innovation lies in the precise molecular recognition mechanism facilitated by the chiral phosphoric acid catalyst system, which creates a well-defined chiral environment through hydrogen bonding interactions with both reaction partners simultaneously activating the electrophilic diketone and nucleophilic amine components. This catalyst design enables highly stereoselective cyclization that forms the critical N-N axis chirality without requiring transition metals or harsh conditions that typically generate impurities in conventional processes. Molecular sieves play a crucial role in maintaining optimal reaction performance by adsorbing water byproducts that could otherwise deactivate the catalyst system. The room temperature operation significantly reduces thermal degradation pathways while carbon tetrachloride provides ideal polarity for both reactant solubility and transition state stabilization, resulting in exceptional product purity without requiring additional purification steps beyond standard column chromatography using petroleum ether/ethyl acetate mixtures.
Impurity profile management is inherently addressed through the reaction's high stereoselectivity and mild conditions which minimize side reactions such as racemization or oxidation that commonly plague traditional synthetic routes. The patent demonstrates consistent production of compounds with >94% enantiomeric excess across multiple substrate combinations as confirmed by HPLC analysis using specific chiral columns like OD-H or IA phases with hexane/isopropanol mobile phases. This inherent purity advantage eliminates costly post-synthesis remediation steps while ensuring reliable batch-to-batch consistency essential for pharmaceutical manufacturing compliance. The demonstrated structural diversity across numerous examples confirms the method's versatility while maintaining stringent purity standards required for clinical applications targeting pancreatic cancer treatment.
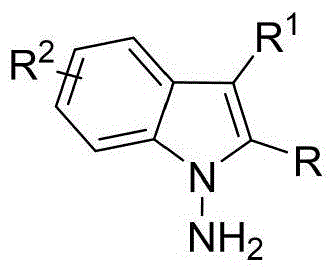
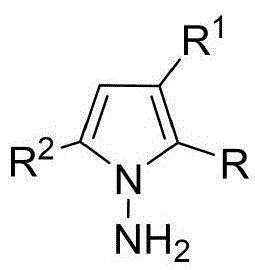
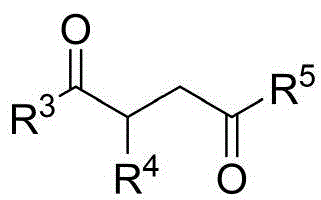
Commercial Advantages for Pharmaceutical Supply Chains
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate manufacturing by transforming complex chiral synthesis into a streamlined process suitable for commercial scale-up while delivering significant cost reduction in API manufacturing. The elimination of transition metal catalysts removes the need for extensive metal residue testing and purification steps that typically add substantial time and expense to traditional processes. The room temperature operation significantly reduces energy consumption compared to conventional high-energy processes while using commercially available starting materials at optimal stoichiometric ratios enhances overall cost efficiency without requiring specialized equipment investments.
- Reduced Manufacturing Costs: The elimination of expensive transition metal catalysts creates substantial cost savings by removing associated purification requirements that typically consume significant resources in chemical manufacturing operations. The room temperature operation dramatically lowers energy consumption compared to conventional high-temperature processes while minimizing solvent usage through simplified workup procedures involving only filtration concentration and single-column chromatography. The use of readily available starting materials at optimized molar ratios (e.g., indoleamine to diketone at 1:1.2) further enhances cost efficiency without requiring specialized infrastructure investments. This comprehensive approach transforms what was previously a costly bottleneck into an economically viable pathway for producing high-purity API intermediates at commercial scale.
- Accelerated Production Timelines: The streamlined single-step process with minimal reaction time (5-48 hours depending on substrate) dramatically shortens manufacturing cycles compared to multi-step conventional routes that require intermediate isolations and purifications between steps. The absence of heating/cooling cycles needed for traditional high-energy processes eliminates significant time delays while enabling faster batch turnover rates in manufacturing facilities. The demonstrated scalability from laboratory to pilot scale without process modification allows rapid technology transfer that reduces lead time for high-purity intermediates from months to weeks during critical drug development phases. This accelerated timeline is particularly valuable for oncology programs where speed to clinic directly impacts patient outcomes and competitive positioning.
- Enhanced Supply Chain Resilience: The use of diverse commercially available starting materials creates multiple sourcing options that mitigate supply chain vulnerabilities common in specialized chemical manufacturing operations requiring rare or single-source reagents. The process's tolerance to various substrate combinations allows flexible production planning based on raw material availability without compromising product quality or requiring extensive revalidation procedures. Simplified equipment requirements enable rapid capacity expansion across multiple manufacturing sites ensuring reliable supply continuity even during market disruptions or demand surges. This inherent flexibility transforms what was previously a fragile supply chain component into a resilient system capable of supporting both clinical trials and commercial production with consistent quality delivery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN114524701B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
