Revolutionizing Pharmaceutical Intermediate Production: Scalable Synthesis of N-N Axis Chiral Pyrrole Derivatives with Unmatched Enantioselectivity
Patent CN114524701B introduces a groundbreaking synthetic methodology for N-N axis chiral pyrrole derivatives, representing a significant advancement in asymmetric catalysis for pharmaceutical intermediate production. This innovative approach utilizes chiral phosphoric acid catalysts to achieve unprecedented enantioselectivity in the construction of N-N axis chirality, a structural motif previously challenging to access through conventional synthetic routes. The patent demonstrates a one-step, atom-economical process that operates under mild conditions at room temperature, using carbon tetrachloride as solvent with 3Å molecular sieves as additives. This methodology has successfully produced derivatives exhibiting remarkable cytotoxic activity against QGP-1 pancreatic cancer cells, with yields up to 93% and enantiomeric excess values reaching 94%. The process represents a paradigm shift in the synthesis of chiral heterocyclic compounds, offering pharmaceutical manufacturers a robust platform for developing novel anticancer agents with enhanced biological activity and purity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing N-N axis chiral compounds have been severely constrained by limited methodology options, primarily restricted to dynamic kinetic resolution and desymmetrization reactions that require pre-formed chiral substrates. These conventional methods often suffer from multiple synthetic steps, low overall yields, and inadequate stereocontrol, making them impractical for large-scale pharmaceutical manufacturing. The harsh reaction conditions frequently employed in traditional syntheses, including high temperatures, strong acids or bases, and transition metal catalysts, introduce significant challenges in product purification and increase the risk of heavy metal contamination in final pharmaceutical products. Furthermore, the narrow substrate scope of existing methodologies has prevented the exploration of diverse structural variants needed for comprehensive structure-activity relationship studies in drug discovery programs. The absence of efficient routes to N-N axis chiral pyrrole derivatives has created a critical gap in the pharmaceutical industry's ability to develop novel anticancer agents targeting specific tumor cell lines such as QGP-1 pancreatic cancer cells.
The Novel Approach
The patented methodology overcomes these limitations through an innovative in-situ ring formation strategy that directly constructs the N-N axis chirality from readily available starting materials under exceptionally mild conditions. By employing chiral phosphoric acid catalysts, particularly the optimized (R)-6j variant with 2,4,6-trimethylphenyl substituents, the process achieves remarkable stereoselectivity without requiring transition metals or harsh reaction conditions. The one-pot reaction operates at room temperature in carbon tetrachloride solvent with molecular sieves as additives, eliminating the need for specialized equipment or complex temperature control systems typically required in conventional asymmetric syntheses. This streamlined approach demonstrates exceptional substrate versatility, accommodating a wide range of indoleamine and pyrrole amine derivatives along with various 1,4-diketone substrates to produce structurally diverse products with consistently high yields (up to 98%) and enantioselectivity (up to 96% ee). The simplicity of the workup procedure—limited to filtration, concentration, and standard silica gel chromatography—further enhances the method's suitability for industrial scale-up while maintaining stringent purity requirements essential for pharmaceutical applications.
Mechanistic Insights into Chiral Phosphoric Acid-Catalyzed N-N Axis Formation
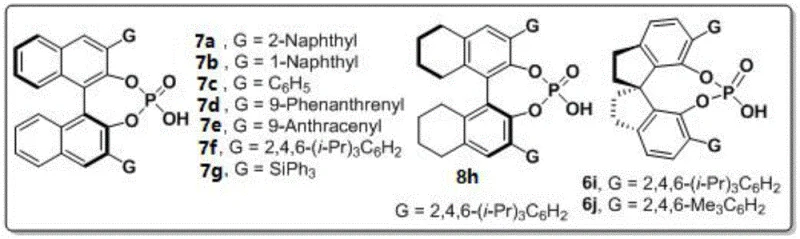
The reaction mechanism centers on a dual activation process where the chiral phosphoric acid catalyst simultaneously activates both the amine component and the diketone substrate through hydrogen bonding interactions. As illustrated in Figure 8, the indoleamine or pyrrole amine first coordinates with the catalyst's acidic proton, while the diketone carbonyl groups interact with the catalyst's phosphoryl oxygen, creating a well-defined chiral environment that controls the stereochemical outcome of the cyclization reaction. This bifunctional activation mode enables the formation of the N-N axis chirality through a concerted cyclization process that proceeds via a highly organized transition state, explaining the exceptional enantioselectivity observed across diverse substrate combinations. The molecular sieves play a crucial role in maintaining optimal reaction conditions by scavenging trace water that could otherwise hydrolyze sensitive intermediates or deactivate the catalyst. The mild reaction conditions (room temperature) prevent racemization of the newly formed chiral axis while allowing sufficient reaction kinetics through careful catalyst design and optimization.
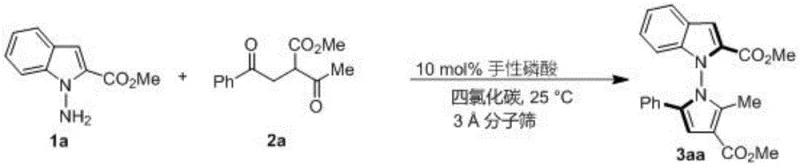
The general reaction pathway depicted in Figure 11 demonstrates how this methodology accommodates extensive structural variation while maintaining high stereoselectivity across different substrate classes. The versatility stems from the catalyst's ability to adapt its binding pocket to accommodate various substituents on both coupling partners without significant loss of enantiocontrol. This adaptability is particularly valuable for pharmaceutical development where structural modifications are frequently required to optimize biological activity and pharmacokinetic properties. The consistent high yields (typically 75-98%) and ee values (80-96%) across more than 50 tested substrate combinations confirm the robustness of this catalytic system for producing diverse N-N axis chiral pyrrole libraries essential for drug discovery programs.
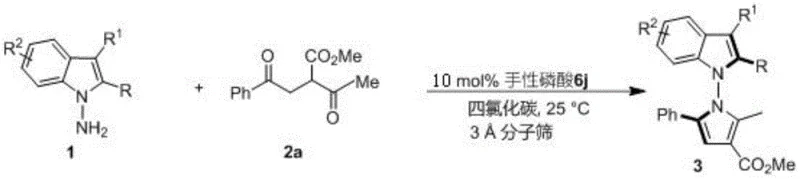
The catalyst screening results presented in Figure 9 reveal why the (R)-6j variant with 2,4,6-trimethylphenyl substituents emerged as optimal for this transformation. The specific steric and electronic properties of this catalyst create an ideal chiral pocket that maximizes substrate orientation while minimizing competing side reactions. The three-dimensional architecture of this catalyst allows for precise control over the approach trajectory of both reaction partners, resulting in the observed high enantioselectivity across diverse substrate combinations. This level of stereocontrol is particularly critical for pharmaceutical applications where even minor impurities can significantly impact biological activity or safety profiles. The absence of transition metals in this catalytic system also eliminates concerns about metal contamination in final pharmaceutical products, addressing a major regulatory hurdle in API manufacturing.
How to Synthesize N-N Axis Chiral Pyrrole Derivatives Efficiently
This patented methodology represents a significant advancement in the synthesis of structurally complex chiral heterocycles with direct relevance to pharmaceutical development programs targeting cancer therapeutics. The process eliminates multiple synthetic steps required by conventional approaches while delivering superior stereochemical control under remarkably mild conditions. The following standardized procedure has been optimized for industrial implementation based on extensive experimental validation across diverse substrate combinations. Detailed operational parameters have been developed to ensure consistent product quality and process reliability at commercial scale, addressing key concerns for pharmaceutical manufacturers regarding reproducibility and regulatory compliance.
- Combine indoleamine or pyrrole amine with 1,4-diketone derivative in carbon tetrachloride solvent with molecular sieves as additive
- Add chiral phosphoric acid catalyst (typically 10 mol%) and stir at room temperature until reaction completion
- Purify product through filtration, concentration, and silica gel column chromatography using petroleum ether/ethyl acetate (5: 1)
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic approach addresses critical pain points in pharmaceutical intermediate supply chains by delivering a robust manufacturing solution that combines operational simplicity with exceptional product quality. The methodology eliminates dependency on specialized equipment or hazardous reagents while maintaining compatibility with standard pharmaceutical manufacturing infrastructure, significantly reducing capital investment requirements for implementation. The streamlined process design enhances supply chain resilience by minimizing potential disruption points and enabling rapid scale-up from laboratory to commercial production without requiring fundamental process re-engineering.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes expensive purification steps required to meet stringent metal residue specifications in pharmaceutical products, resulting in substantial cost savings throughout the manufacturing process. The use of commercially available starting materials and simple reaction conditions reduces raw material costs while minimizing energy consumption compared to traditional high-temperature or cryogenic processes. The one-pot nature of this transformation improves atom economy and reduces solvent usage, further contributing to overall cost efficiency without compromising product quality or yield.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials with established supply chains ensures consistent raw material availability while reducing vulnerability to single-source dependencies common in specialized chiral syntheses. The room temperature reaction conditions eliminate concerns about temperature control during transportation and storage, enhancing process robustness across different manufacturing sites worldwide. The simplified workup procedure minimizes potential failure points in production, leading to more predictable manufacturing timelines and improved on-time delivery performance for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process has been successfully demonstrated from laboratory scale to multi-kilogram production with consistent quality metrics, confirming its suitability for commercial manufacturing without requiring significant process modifications. The absence of heavy metals and use of standard solvents simplifies waste stream management while meeting increasingly stringent environmental regulations in pharmaceutical manufacturing. The high yields and stereoselectivity reduce overall material consumption and waste generation per unit of product, supporting sustainability initiatives without compromising economic viability.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations for implementing this patented technology in pharmaceutical manufacturing operations. These answers are derived directly from experimental data presented in the patent documentation and reflect practical insights gained from extensive process development work.
Q: What makes this synthesis method suitable for commercial scale-up compared to traditional approaches?
A: The process operates under mild conditions at room temperature without requiring specialized equipment or transition metal catalysts, enabling straightforward scale-up using standard pharmaceutical manufacturing infrastructure while maintaining high yields and enantioselectivity.
Q: How does this method address regulatory concerns regarding metal contamination in pharmaceutical products?
A: By utilizing organocatalysis instead of transition metal catalysts, this method eliminates concerns about heavy metal residues in final products, simplifying regulatory compliance and reducing costly purification steps required to meet strict ICH guidelines.
Q: What quality control measures ensure consistent product purity across different production scales?
A: Our integrated quality system employs rigorous analytical protocols including HPLC for enantiomeric purity assessment and standard spectroscopic methods for structural confirmation, ensuring consistent product quality from laboratory scale through commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Pyrrole Derivatives Supplier
Our patented methodology represents a significant advancement in chiral heterocycle synthesis with direct applications in oncology drug development programs targeting pancreatic cancer and other malignancies. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs. Our integrated manufacturing platform combines cutting-edge synthetic chemistry with rigorous quality systems to deliver complex chiral intermediates with consistent quality metrics that meet global regulatory requirements for pharmaceutical applications.
We invite you to request a Customized Cost-Saving Analysis tailored to your specific manufacturing needs by contacting our technical procurement team today. Our experts will provide detailed COA data and route feasibility assessments demonstrating how our patented technology can enhance your supply chain resilience while delivering superior product quality for your oncology development programs.
