Advanced 5-Trifluoromethyl-1,2,4-Triazole Manufacturing: Scalable Solutions for Pharmaceutical Innovation
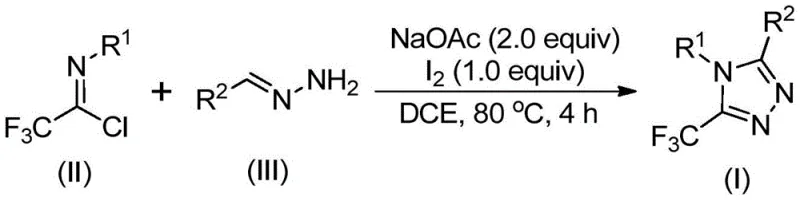
Patent CN110467579B introduces a groundbreaking synthetic methodology for producing 5-trifluoromethyl-substituted 1,2,4-triazole compounds, representing a significant advancement in heterocyclic chemistry with profound implications for pharmaceutical manufacturing. This novel approach eliminates the need for heavy metal catalysts while operating under standard atmospheric conditions, addressing critical limitations in conventional triazole synthesis that have constrained industrial adoption. The process leverages readily available starting materials including sodium acetate, trifluoroethyl imidoyl chloride (II), and hydrazone (III) in dichloroethane solvent at 80°C, demonstrating exceptional operational simplicity and robustness. By avoiding stringent anhydrous and anaerobic requirements typical of existing methods, this innovation substantially reduces manufacturing complexity while maintaining high product quality standards essential for pharmaceutical intermediates. The methodology's versatility in accommodating diverse substituents positions it as a strategic solution for producing complex triazole derivatives required in modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethylated triazoles typically require expensive and hazardous trifluoromethylation reagents or specialized transition metal catalysts that introduce significant purification challenges due to metal residue contamination. These methods often demand strictly controlled anhydrous and anaerobic conditions that substantially increase operational complexity and equipment requirements, making scale-up economically unviable for many manufacturers. The reliance on rare or toxic catalysts creates supply chain vulnerabilities while generating complex waste streams that require costly treatment processes to meet environmental regulations. Furthermore, conventional routes frequently exhibit narrow substrate scope with limited tolerance for functional groups, restricting their applicability in producing diverse triazole derivatives needed for pharmaceutical development. The multi-step nature of existing protocols also contributes to lower overall yields and higher production costs that undermine commercial viability for large-scale manufacturing operations.
The Novel Approach
The patented methodology overcomes these limitations through an elegant iodine-mediated cyclization process that operates under ambient conditions without requiring specialized equipment or hazardous reagents. By utilizing elemental iodine as a catalyst and sodium acetate as a base in dichloroethane solvent at moderate temperatures (80°C), the process achieves high efficiency while eliminating toxic metal residues that complicate traditional manufacturing routes. The reaction demonstrates remarkable functional group tolerance across diverse substrates (R1 = aryl; R2 = alkenyl/aryl/heteroaryl), enabling the production of various substituted triazole derivatives through simple precursor modifications. Crucially, the absence of anhydrous and anaerobic requirements significantly simplifies process engineering while reducing operational costs associated with specialized reactor systems and inert gas handling. This streamlined approach maintains excellent yield profiles across multiple substrate variations while providing a robust foundation for industrial scale-up without compromising product quality or purity standards required in pharmaceutical applications.
Mechanistic Insights into Iodine-Mediated Triazole Cyclization
The reaction mechanism proceeds through a well-defined sequence beginning with base-promoted intermolecular carbon-nitrogen bond formation between trifluoroethyl imidoyl chloride (II) and hydrazone (III), yielding a trifluoroacetamidine intermediate that subsequently undergoes isomerization. This key intermediate then experiences base-promoted oxidative iodination facilitated by elemental iodine to form an iodinated species that serves as the critical precursor for cyclization. The iodinated intermediate undergoes intramolecular electrophilic substitution followed by aromatization to form the final 5-trifluoromethyl-substituted 1,2,4-triazole product (I) with high regioselectivity. This mechanistic pathway avoids radical pathways that typically generate impurities in conventional syntheses while maintaining excellent control over the reaction trajectory through the carefully balanced molar ratios of sodium acetate (2.0 equiv) to iodine (1.0 equiv). The non-radical nature of this transformation significantly reduces side product formation compared to alternative methods that rely on unstable trifluoromethylation reagents or transition metal catalysts.
Impurity control is achieved through the precise stoichiometric balance between sodium acetate and iodine (2.0–3.0:1 molar ratio), which optimizes the oxidative iodination step while minimizing over-reaction or decomposition pathways. The moderate reaction temperature (80°C) prevents thermal degradation of sensitive intermediates while ensuring sufficient reaction kinetics for complete conversion within the specified timeframe. The use of dichloroethane as the preferred solvent provides optimal solubility for all reactants while maintaining inertness toward the iodine catalyst system. Post-reaction purification through standard column chromatography effectively removes any residual starting materials or minor byproducts without requiring specialized techniques to eliminate metal contaminants that plague traditional catalytic methods. This combination of controlled reaction parameters and straightforward purification delivers consistent high-purity products meeting stringent pharmaceutical quality requirements across diverse substrate variations.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
This innovative synthesis protocol represents a significant advancement in triazole chemistry by providing a practical route to valuable pharmaceutical intermediates without requiring specialized equipment or hazardous reagents. The process demonstrates exceptional operational simplicity through its ambient condition requirements and use of commercially available starting materials that eliminate supply chain constraints associated with rare catalysts or sensitive reagents. By following the standardized procedure outlined in the patent documentation, manufacturers can achieve consistent high-quality results across multiple production scales while maintaining excellent process control throughout the reaction sequence. Detailed standardized synthesis steps are provided below to ensure reliable implementation of this methodology in industrial settings.
- Combine sodium acetate (2.0 equiv), trifluoroethyl imidoyl chloride (II), and hydrazone (III) in dichloroethane solvent under ambient conditions without requiring anhydrous or anaerobic environments.
- Heat the reaction mixture to 80°C for 4 hours followed by addition of elemental iodine (1.0 equiv) and continue reaction for an additional hour to complete the cyclization process.
- Perform post-processing through filtration, silica gel mixing, and column chromatography purification to obtain high-purity 5-trifluoromethyl-substituted 1,2,4-triazole compounds (I).
Commercial Advantages for Procurement and Supply Chain Teams
This novel manufacturing approach delivers substantial value across procurement and supply chain functions by addressing critical pain points inherent in traditional triazole synthesis methods. The elimination of heavy metal catalysts removes significant cost drivers associated with catalyst procurement, specialized handling procedures, and extensive post-reaction purification steps required to eliminate toxic residues from final products. By operating under standard atmospheric conditions without requiring anhydrous or anaerobic environments, the process reduces capital expenditure on specialized reactor systems while lowering operational costs associated with inert gas consumption and moisture control systems. The use of readily available starting materials from established chemical suppliers enhances supply chain resilience while minimizing raw material cost volatility that often impacts pharmaceutical manufacturing economics.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts eliminates both the direct material costs and the substantial downstream processing expenses required to remove metal contaminants from final products. This streamlined approach reduces overall production complexity by avoiding multi-step purification protocols typically needed to achieve pharmaceutical-grade purity standards when using metal-catalyzed reactions.
- Enhanced Supply Chain Reliability: Utilizing commercially available reagents from multiple global suppliers creates robust sourcing options that mitigate single-source dependency risks common in specialized chemical manufacturing. The process's tolerance for standard industrial equipment enables rapid technology transfer between manufacturing sites without requiring significant capital investments in new infrastructure.
- Scalability and Environmental Compliance: The ambient condition operation and straightforward workup procedure facilitate seamless scale-up from laboratory to commercial production volumes while generating simpler waste streams that require less complex treatment compared to processes involving toxic metals or hazardous reagents. This environmental advantage aligns with growing regulatory pressures for sustainable manufacturing practices in the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on the patented methodology's specific advantages and implementation requirements. These insights are derived directly from the patent documentation and experimental validation data provided in the original filing.
Q: Why is this metal-free synthesis advantageous for pharmaceutical manufacturing?
A: The elimination of heavy metal catalysts removes critical purification steps required to eliminate toxic metal residues, significantly reducing both production costs and regulatory compliance risks while maintaining high product purity essential for pharmaceutical applications.
Q: How does the process ensure high purity without heavy metal catalysts?
A: The iodine-mediated cyclization mechanism provides selective transformation with minimal byproduct formation, while the straightforward post-processing protocol effectively removes impurities without requiring specialized metal-scavenging procedures that complicate traditional manufacturing routes.
Q: What scalability has been demonstrated for commercial production?
A: The process operates under standard atmospheric conditions using readily available reagents and solvents, with documented gram-scale feasibility indicating strong potential for seamless scale-up to industrial production volumes without specialized equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic compounds while maintaining stringent purity specifications through rigorous QC labs and advanced analytical capabilities. As a specialized CDMO partner with deep expertise in triazole chemistry, we offer tailored manufacturing solutions that leverage this patented methodology to deliver high-quality intermediates meeting exacting pharmaceutical standards. Our integrated approach combines cutting-edge process chemistry with robust quality systems to ensure consistent product performance across all production scales.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis can optimize your specific manufacturing requirements. Contact us today to obtain detailed COA data and comprehensive route feasibility assessments for your target compounds.
