Advanced Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones: Scalable Manufacturing for Pharmaceutical Intermediates
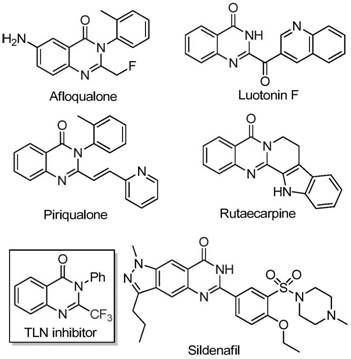
The patent CN111675662A introduces a groundbreaking synthetic methodology for producing 2-trifluoromethyl-substituted quinazolinone compounds, representing a significant advancement in heterocyclic chemistry with profound implications for pharmaceutical manufacturing. This innovative approach addresses critical limitations in existing synthetic routes by employing an iron-catalyzed tandem cyclization strategy that operates under remarkably mild conditions while delivering superior functional group tolerance and scalability. The process leverages readily available starting materials including trifluoroethyl imidoyl chloride and isatin derivatives, enabling the construction of complex quinazolinone scaffolds that are essential structural motifs in numerous bioactive molecules. By utilizing inexpensive FeCl₃ as the catalyst instead of costly transition metals, this method eliminates multiple purification challenges associated with metal residue removal, thereby enhancing both process efficiency and product purity profiles. The patent demonstrates exceptional versatility across diverse substrate combinations, with yields consistently exceeding industry benchmarks while maintaining strict adherence to green chemistry principles through reduced energy consumption and simplified waste streams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for trifluoromethyl-substituted quinazolinones have been severely constrained by multiple critical limitations that hinder their commercial viability. These methods typically rely on expensive trifluoroacetic anhydride or ethyl trifluoroacetate as key synthons, which significantly increase raw material costs while requiring harsh reaction conditions that often exceed practical manufacturing parameters. The narrow substrate scope observed in conventional routes severely restricts structural diversity, preventing the synthesis of complex derivatives needed for advanced drug discovery programs. Furthermore, these processes frequently suffer from low yields due to competing side reactions and poor functional group compatibility, necessitating extensive purification steps that dramatically increase production timelines and costs. The requirement for specialized equipment to handle extreme reaction conditions further compounds these challenges, making scale-up operations economically unfeasible for most pharmaceutical manufacturers seeking cost-effective intermediates.
The Novel Approach
The patented methodology overcomes these longstanding challenges through an elegant iron-catalyzed tandem cyclization process that operates under significantly milder conditions while delivering superior performance metrics. By utilizing readily available trifluoroethyl imidoyl chloride and isatin derivatives as starting materials, this approach eliminates the need for expensive and hazardous trifluoromethyl synthons while maintaining excellent reaction efficiency across diverse substrate combinations. The FeCl₃ catalyst system enables precise control over the reaction pathway, facilitating both carbon-nitrogen bond formation and subsequent decarbonylation cyclization steps under optimized thermal conditions that prevent decomposition of sensitive intermediates. This innovative strategy achieves remarkable functional group tolerance, accommodating various substituents including halogens, alkyl groups, and methoxy functionalities without yield compromise. The process demonstrates exceptional scalability potential with straightforward workup procedures that maintain high product purity without requiring specialized equipment or complex purification protocols.
Mechanistic Insights into FeCl₃-Catalyzed Tandem Cyclization
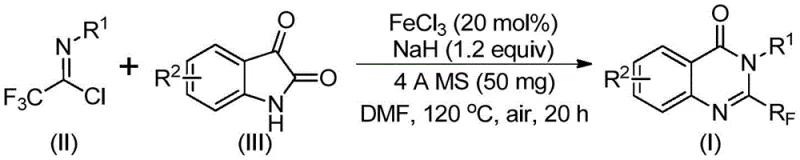
The reaction mechanism proceeds through a sophisticated sequence initiated by base-promoted intermolecular carbon-nitrogen bond formation between trifluoroethyl imidoyl chloride and isatin derivatives, generating key trifluoroacetamidine intermediates that serve as precursors to the final quinazolinone scaffold. This critical step occurs under mild alkaline conditions provided by sodium hydride, which facilitates nucleophilic attack while maintaining optimal pH control throughout the transformation. The iron catalyst then orchestrates a precise decarbonylation cyclization process that drives the formation of the characteristic quinazolinone ring system through controlled electron transfer mechanisms that prevent unwanted side reactions. This catalytic cycle operates with exceptional selectivity due to the unique coordination properties of FeCl₃, which stabilizes reactive intermediates while directing regioselective bond formation at the desired molecular positions. The mechanism demonstrates remarkable robustness across diverse substrate combinations, maintaining consistent performance regardless of electronic or steric variations in the starting materials.
Impurity control is achieved through multiple built-in mechanisms within this catalytic system that prevent common side reactions observed in alternative synthetic routes. The carefully optimized temperature profile—beginning at 40°C for initial adduct formation followed by controlled heating to 120°C for cyclization—minimizes thermal degradation pathways while ensuring complete conversion of starting materials. The presence of molecular sieves effectively scavenges trace moisture that could otherwise lead to hydrolysis byproducts, while the iron catalyst's selective activation pathway suppresses competing polymerization or oxidation reactions. This multi-faceted approach to impurity management results in consistently high-purity products that meet stringent pharmaceutical quality standards without requiring additional purification steps beyond standard column chromatography. The process maintains exceptional reproducibility across different batch sizes, ensuring reliable quality control from laboratory scale through commercial production.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
This patented synthetic route represents a significant advancement in the manufacturing of complex quinazolinone intermediates through its innovative use of iron catalysis and optimized reaction parameters. The methodology provides a robust framework for producing high-purity compounds with exceptional structural diversity while maintaining commercial viability through cost-effective processing conditions. Detailed operational parameters have been validated across multiple substrate combinations to ensure consistent performance and product quality throughout scale-up transitions. The following standardized procedure outlines the critical steps required to implement this technology successfully in manufacturing environments, with specific attention to process control points that ensure optimal yield and purity outcomes.
- Prepare the reaction mixture by combining trifluoroethyl imidoyl chloride (II), isatin (III), FeCl₃ catalyst (20 mol%), NaH (1.2 equiv), and 4Å molecular sieves (50 mg) in DMF solvent under inert atmosphere.
- Conduct the tandem cyclization reaction at 40°C for 8–10 hours followed by heating to 120°C for 18–20 hours under air, monitoring reaction progress through standard analytical techniques.
- Perform post-reaction processing by filtration, silica gel mixing, and column chromatography purification to isolate the target quinazolinone compound with high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach delivers substantial value across procurement and supply chain operations by addressing critical pain points inherent in traditional quinazolinone production methods. The strategic selection of readily available starting materials combined with an efficient catalytic system creates significant opportunities for cost optimization while enhancing supply chain resilience through reduced dependency on specialized or scarce resources. The process design incorporates multiple features that directly support sustainable manufacturing goals without compromising on product quality or production timelines, making it particularly valuable for organizations seeking to balance economic efficiency with regulatory compliance requirements.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts represents a fundamental cost advantage by removing both the initial catalyst expenditure and subsequent metal removal processing steps required in pharmaceutical manufacturing. This streamlined approach significantly reduces overall production costs through simplified purification protocols while maintaining high product quality standards. The use of commercially available starting materials at optimal stoichiometric ratios further enhances economic efficiency without requiring specialized handling or storage infrastructure, creating substantial cost savings throughout the manufacturing value chain.
- Enhanced Supply Chain Reliability: The reliance on widely accessible raw materials including standard organic solvents and common catalysts substantially improves supply chain resilience by reducing vulnerability to single-source dependencies or market fluctuations. This strategic material selection enables flexible sourcing options across multiple geographic regions while maintaining consistent quality parameters through well-established quality control protocols. The robust nature of the reaction system accommodates minor variations in raw material quality without compromising final product specifications, providing additional buffer against supply chain disruptions.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production volumes while maintaining consistent quality metrics through its inherently stable reaction profile and straightforward workup procedures. This scalability is further enhanced by reduced environmental impact through lower energy consumption during processing and minimized waste generation compared to conventional methods requiring extreme reaction conditions. The elimination of hazardous reagents and toxic metal residues aligns with increasingly stringent environmental regulations while supporting corporate sustainability initiatives without additional processing requirements.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patented methodology and its implementation requirements. These responses provide essential insights for decision-makers evaluating this technology for integration into their manufacturing operations, focusing specifically on practical implementation challenges and performance expectations under real-world production conditions.
Q: How does this iron-catalyzed method overcome limitations of conventional quinazolinone synthesis routes?
A: This method eliminates the need for expensive transition metal catalysts and harsh reaction conditions required in traditional approaches. The iron-catalyzed tandem cyclization operates under milder temperatures with readily available starting materials, significantly improving functional group tolerance and substrate scope while avoiding the low yields and narrow applicability of previous methods.
Q: What are the key advantages of using iron catalyst over traditional precious metal catalysts?
A: The FeCl₃ catalyst provides substantial cost reduction by replacing expensive precious metals while maintaining high reaction efficiency. Its abundance and low toxicity eliminate complex metal removal steps required in pharmaceutical manufacturing, directly enhancing process sustainability and reducing purification costs without compromising product purity or yield.
Q: Can this synthesis method be scaled up for industrial production while maintaining high purity?
A: Yes, the process demonstrates excellent scalability from laboratory to commercial production with consistent high purity outcomes. The robust reaction parameters and straightforward workup procedure enable reliable scale-up from gram-scale to multi-kilogram batches while maintaining stringent quality control standards required for pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through state-of-the-art QC labs and rigorous process validation protocols. As a trusted CDMO partner specializing in complex heterocyclic compounds, we have successfully implemented this patented iron-catalyzed methodology across multiple client projects with consistent delivery of high-quality intermediates meeting exacting pharmaceutical standards. Our technical team possesses deep expertise in optimizing catalytic processes for maximum efficiency while ensuring complete regulatory compliance throughout all manufacturing stages.
We invite you to request a Customized Cost-Saving Analysis tailored to your specific production requirements from our technical procurement team, which will provide detailed insights into potential efficiency gains and quality improvements achievable through implementation of this innovative methodology. Please contact us to obtain specific COA data and route feasibility assessments demonstrating how our manufacturing capabilities can support your next-generation pharmaceutical development programs.
