Advanced Synthesis of N-N Axis Chiral Pyrrole Derivatives for Oncology Drug Development
Introduction to Novel N-N Axis Chiral Pyrrole Chemistry
The landscape of asymmetric synthesis is continually evolving, driven by the demand for complex chiral scaffolds in modern drug discovery. Patent CN114524701B introduces a groundbreaking methodology for the construction of N-N axis chiral pyrrole derivatives, a class of compounds that has historically been challenging to access with high stereochemical fidelity. This technology leverages a sophisticated organocatalytic system to achieve in-situ ring formation, bypassing the limitations of conventional resolution techniques. The resulting derivatives, characterized by their unique N-N axial chirality, have demonstrated remarkable biological potential, specifically exhibiting high sensitivity and strong cytotoxic activity against QGP-1 tumor cells. 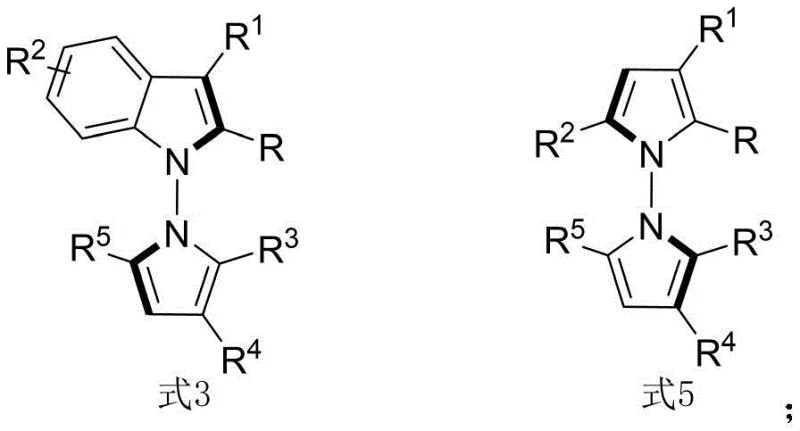 For pharmaceutical developers, this represents a significant opportunity to explore new chemical space for oncology therapeutics. The ability to synthesize these structures efficiently opens doors for reliable pharmaceutical intermediate supplier partnerships focused on cutting-edge anticancer research.
For pharmaceutical developers, this represents a significant opportunity to explore new chemical space for oncology therapeutics. The ability to synthesize these structures efficiently opens doors for reliable pharmaceutical intermediate supplier partnerships focused on cutting-edge anticancer research.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of axially chiral compounds has relied heavily on dynamic kinetic resolution (DKR) or desymmetrization strategies. These classical approaches often suffer from inherent theoretical yield limits, typically capping at 50% unless a dynamic equilibrium is perfectly managed, which is rarely the case in complex heterocyclic systems. Furthermore, existing methods for generating N-N axis chirality are extremely scarce in the literature, limiting the structural diversity available to medicinal chemists. The reliance on transition metal catalysts in some older protocols also introduces significant downstream processing challenges, such as the need for rigorous heavy metal removal to meet stringent regulatory standards for API manufacturing. These factors collectively result in higher production costs, longer lead times, and a restricted library of accessible analogs for biological screening.
The Novel Approach
The methodology disclosed in CN114524701B revolutionizes this field by employing a direct, organocatalytic cyclization strategy. Instead of resolving a racemic mixture, this process constructs the chiral axis de novo from achiral or pro-chiral precursors. By utilizing 1,4-diketone derivatives and indoleamine or pyrrole amine substrates, the reaction proceeds through a highly selective pathway mediated by a chiral phosphoric acid catalyst. 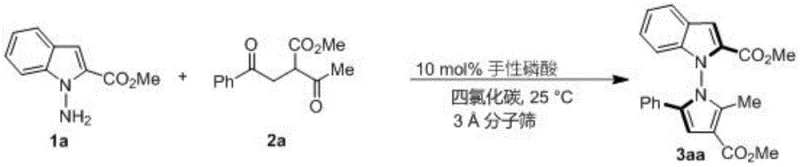 This approach not only achieves excellent yields (often exceeding 90%) but also delivers exceptional enantioselectivity, with ee values reaching up to 96%. The use of carbon tetrachloride as a solvent and 3A molecular sieves as additives ensures a controlled reaction environment that favors the desired stereochemical outcome. This shift from resolution to asymmetric synthesis fundamentally changes the economics of producing these high-value intermediates.
This approach not only achieves excellent yields (often exceeding 90%) but also delivers exceptional enantioselectivity, with ee values reaching up to 96%. The use of carbon tetrachloride as a solvent and 3A molecular sieves as additives ensures a controlled reaction environment that favors the desired stereochemical outcome. This shift from resolution to asymmetric synthesis fundamentally changes the economics of producing these high-value intermediates.
Mechanistic Insights into Chiral Phosphoric Acid Catalysis
The success of this transformation hinges on the precise design of the chiral phosphoric acid catalyst. The patent details an extensive screening of various catalyst backbones, including binaphthyl, octahydrobinaphthyl, and spiro-type derivatives. The data unequivocally points to the superiority of the spiro-type chiral phosphoric acid, specifically compound 6j bearing 2,4,6-trimethylphenyl groups. 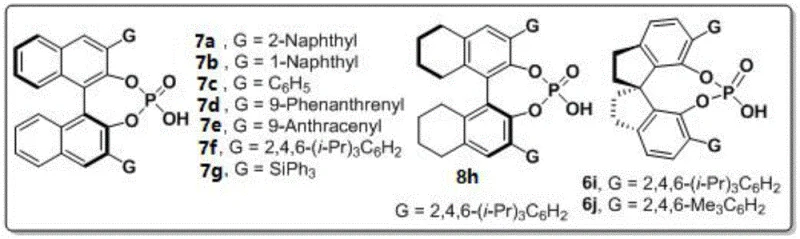 Mechanistically, the catalyst likely operates through a dual hydrogen-bonding activation mode. The acidic proton of the phosphoric acid activates the carbonyl group of the 1,4-diketone, while the phosphoryl oxygen may coordinate with the nucleophilic amine species. The bulky 3,3'-substituents on the spiro backbone create a confined chiral pocket that sterically directs the approach of the reactants, thereby enforcing the formation of one specific atropisomer over the other. This level of stereocontrol is critical for ensuring the biological consistency of the final drug substance.
Mechanistically, the catalyst likely operates through a dual hydrogen-bonding activation mode. The acidic proton of the phosphoric acid activates the carbonyl group of the 1,4-diketone, while the phosphoryl oxygen may coordinate with the nucleophilic amine species. The bulky 3,3'-substituents on the spiro backbone create a confined chiral pocket that sterically directs the approach of the reactants, thereby enforcing the formation of one specific atropisomer over the other. This level of stereocontrol is critical for ensuring the biological consistency of the final drug substance.
Furthermore, the robustness of this catalytic system allows for a wide tolerance of functional groups on both the amine and diketone components. Substituents such as halogens, alkoxy groups, and various ester functionalities are well-tolerated without compromising the enantiomeric excess. This substrate flexibility is a key advantage for process chemists aiming to generate structure-activity relationship (SAR) libraries rapidly. The mechanism avoids the formation of stable metal-complex intermediates, which simplifies the reaction profile and reduces the risk of side reactions associated with metal coordination. Consequently, the impurity profile of the crude reaction mixture is cleaner, facilitating easier purification and higher overall process efficiency.
How to Synthesize N-N Axis Chiral Pyrrole Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and stereoselectivity. The process is designed to be operationally simple, avoiding the need for cryogenic temperatures or inert atmosphere gloveboxes, which significantly lowers the barrier to entry for adoption. The standard protocol involves mixing the amine substrate and the 1,4-diketone derivative in carbon tetrachloride, followed by the addition of the catalyst and molecular sieves. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety compliance during scale-up operations.
- Prepare the reaction mixture by combining indoleamine or pyrrole amine substrates with 1,4-diketone derivatives in carbon tetrachloride solvent.
- Add 3A molecular sieves as an additive and introduce 10 mol% of the optimized chiral phosphoric acid catalyst (specifically spiro-type catalyst 6j).
- Stir the reaction at 25°C until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography to isolate the high-purity chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers compelling advantages that directly address the pain points of procurement managers and supply chain directors. The shift to an organocatalytic process eliminates the dependency on expensive and supply-constrained transition metals like palladium or rhodium. This substitution not only reduces the raw material cost but also simplifies the supply chain by removing the need for specialized metal scavengers and the associated validation testing for residual metals. The mild reaction conditions, operating effectively at 25°C, translate to significant energy savings compared to processes requiring heating or deep cooling, contributing to a lower carbon footprint and reduced utility costs.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes a major cost driver associated with both the catalyst itself and the downstream purification required to meet regulatory limits. Additionally, the high yields and enantioselectivity reported in the patent minimize the loss of valuable chiral material, effectively increasing the throughput of the manufacturing line without additional capital expenditure. The use of readily available starting materials further stabilizes the cost base, protecting against volatility in the pricing of exotic reagents.
- Enhanced Supply Chain Reliability: By utilizing a robust organocatalytic system, the manufacturing process becomes less susceptible to disruptions in the supply of specialized metal catalysts. The reagents involved, such as indoleamines and 1,4-diketones, are commodity chemicals with established global supply chains, ensuring consistent availability. The simplicity of the work-up procedure, involving basic filtration and concentration, reduces the turnaround time between batches, allowing for more responsive production scheduling to meet fluctuating market demands.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential due to its mild thermal profile and lack of hazardous metal waste streams. The absence of heavy metals simplifies waste treatment protocols, aligning with increasingly stringent environmental regulations regarding industrial effluent. The high atom economy of the cyclization reaction ensures that a maximum proportion of the input mass is converted into the desired product, reducing the volume of chemical waste generated per kilogram of API intermediate produced.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into your existing development pipeline.
Q: What is the primary advantage of this synthesis method over traditional dynamic kinetic resolution?
A: Unlike traditional methods limited to dynamic kinetic resolution or desymmetrization, this patent utilizes an in-situ ring formation strategy via organocatalysis. This approach significantly broadens the substrate scope, allowing for the creation of diverse N-N axis chiral structures that were previously inaccessible, while achieving superior enantioselectivity (up to 96% ee) under mild room temperature conditions.
Q: Why is the spiro-type chiral phosphoric acid catalyst (6j) preferred for this reaction?
A: Screening data within the patent indicates that the spiro-type chiral phosphoric acid catalyst 6j provides the highest stereocontrol compared to binaphthyl or octahydrobinaphthyl derivatives. Its unique rigid three-dimensional structure creates a more defined chiral environment, effectively differentiating the transition states during the cyclization process, which results in consistently higher enantiomeric excess values across various substrate combinations.
Q: What are the biological implications of these new N-N axis chiral pyrrole derivatives?
A: Biological activity assays described in the patent demonstrate that these synthesized derivatives exhibit potent cytotoxic activity against QGP-1 pancreatic tumor cells. The N-N axial chirality appears to play a critical role in enhancing sensitivity to these cancer cells, suggesting that these compounds serve as valuable leads for developing next-generation anticancer agents with improved therapeutic indices.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Pyrrole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in CN114524701B for the development of next-generation oncology therapeutics. As a leading CDMO partner, we possess the technical expertise to adapt and optimize this organocatalytic route for your specific project needs. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-N axis chiral pyrrole derivatives meets the highest quality standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us today to request specific COA data for our reference standards and comprehensive route feasibility assessments. Let us help you accelerate your path to market with reliable, high-quality chiral intermediates.
