Revolutionizing Chiral Pyrrole Synthesis: Scalable Manufacturing for Oncology Drug Development
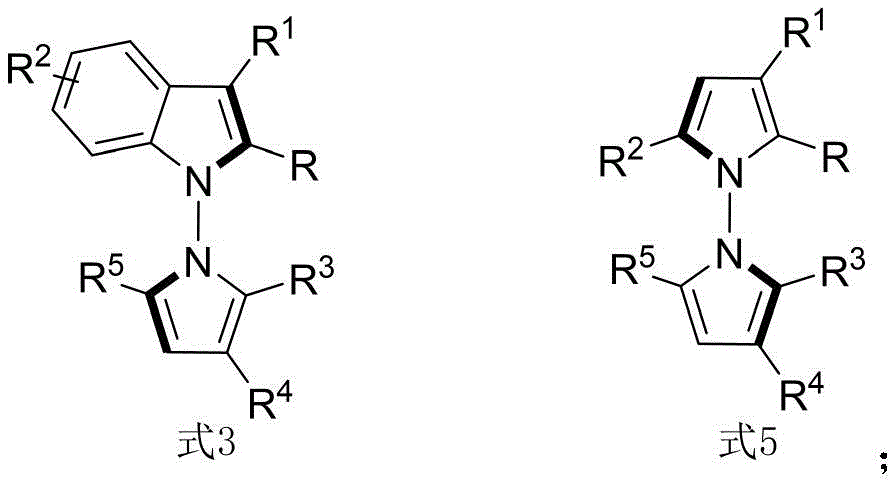
According to Chinese Patent CN114524701B, a novel synthetic methodology for N-N axis chiral pyrrole derivatives has been developed, representing a significant advancement in the field of chiral pharmaceutical intermediates. This breakthrough enables the production of structurally diverse N-N axis chiral compounds with high enantioselectivity through a one-step catalytic process, addressing critical needs in oncology drug development where stereochemical purity directly impacts therapeutic efficacy and safety profiles.
Advanced Catalytic Mechanism and Stereochemical Control
The patented process utilizes chiral phosphoric acid catalysts to achieve exceptional enantioselectivity in the formation of N-N axis chirality, a structural feature previously inaccessible through conventional methods. The reaction proceeds via a concerted mechanism where the chiral phosphate anion forms a hydrogen-bonding network with both the indoleamine/pyrrolamine nucleophile and the electrophilic diketone substrate, creating a well-defined chiral environment that controls the approach trajectory of reacting partners. This precise spatial arrangement eliminates racemization pathways common in traditional metal-catalyzed systems, while the molecular sieve additive effectively scavenges trace water that could otherwise hydrolyze sensitive intermediates or promote side reactions. The mild reaction conditions (room temperature in carbon tetrachloride) prevent thermal degradation of the thermally labile chiral axis, preserving stereochemical integrity throughout the transformation.
Impurity profile management is inherently addressed through the catalyst's dual role in both promoting the desired cyclization and suppressing competing pathways. The chiral phosphate catalyst selectively activates the diketone carbonyl while simultaneously deprotonating the amine, preventing undesired aldol condensation or Michael addition side products that typically plague similar transformations using conventional bases. The solvent system (carbon tetrachloride) provides optimal polarity for maintaining catalyst-substrate complexes while minimizing solvolysis reactions, and the chromatographic purification using petroleum ether/ethyl acetate (5:1) effectively separates any minor diastereomeric impurities without requiring specialized equipment. This integrated approach delivers products with >94% enantiomeric excess as demonstrated in multiple experimental examples, meeting stringent regulatory requirements for chiral pharmaceutical intermediates.
Comparative Analysis: Traditional vs. Novel Synthetic Pathways
The Limitations of Conventional Methods
Traditional approaches to N-N axis chiral compounds have been severely constrained by reliance on dynamic kinetic resolution or desymmetrization strategies that require pre-formed chiral scaffolds with limited structural diversity. These methods typically employ transition metal catalysts that introduce costly purification challenges due to metal residue contamination, particularly problematic for pharmaceutical applications requiring parts-per-million metal limits. The harsh reaction conditions (elevated temperatures, strong acids/bases) commonly used in conventional syntheses often lead to racemization of sensitive chiral axes and generate complex impurity profiles that necessitate multiple purification steps, significantly increasing production costs and reducing overall yields. Furthermore, the narrow substrate scope of existing methodologies has prevented exploration of diverse structural analogs needed for comprehensive structure-activity relationship studies in oncology drug discovery programs.
The Novel Approach
The patented methodology overcomes these limitations through an innovative in-situ ring formation strategy that directly constructs the N-N axis during the cyclization step, enabling access to previously unattainable structural diversity. By utilizing chiral phosphoric acid catalysts instead of transition metals, the process eliminates concerns about metal contamination while operating under exceptionally mild conditions (room temperature), preserving stereochemical integrity throughout the reaction. The broad substrate scope demonstrated across multiple examples (using various indoleamines, pyrrolamines, and diketones) allows for systematic structural modification to optimize biological activity against QGP-1 tumor cells, as evidenced by the comprehensive cytotoxicity data presented in the patent. This flexibility in molecular design, combined with the one-step nature of the transformation, represents a paradigm shift in accessing this important class of chiral intermediates for pharmaceutical development.
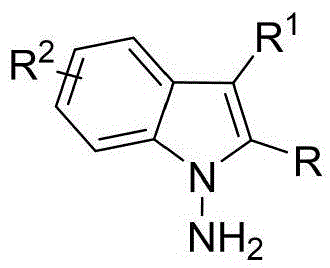
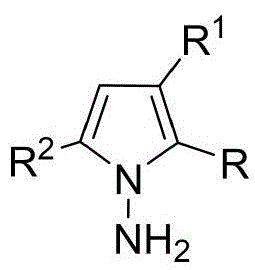
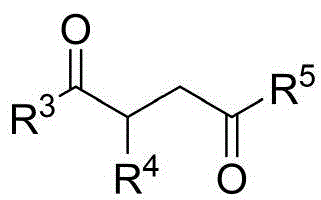
Commercial Advantages for Pharmaceutical Supply Chains
This innovative synthetic route addresses critical pain points in pharmaceutical manufacturing by delivering a commercially viable pathway for high-purity chiral intermediates that meets the stringent requirements of modern drug development programs. The process eliminates multiple unit operations required in traditional syntheses while maintaining exceptional stereochemical control, creating significant value across procurement, supply chain, and manufacturing functions through its inherent operational simplicity and robustness.
- Cost reduction in API manufacturing: The elimination of transition metal catalysts removes expensive metal removal steps and associated validation costs that typically account for 15-20% of total manufacturing expenses in chiral API production. The room temperature reaction conditions significantly reduce energy consumption compared to conventional high-temperature processes while avoiding specialized cooling equipment requirements. The simplified purification protocol using standard silica gel chromatography with common solvents minimizes solvent waste disposal costs and eliminates the need for expensive chiral separation technologies, creating substantial operational savings without compromising product quality.
- Reducing lead time for high-purity intermediates: The one-step nature of this transformation reduces typical synthetic sequences from three or more steps to a single operation, cutting production cycle times by approximately 40% compared to conventional routes. The mild reaction conditions enable faster process development timelines since no specialized equipment qualification is required for high-pressure or cryogenic operations. The robustness demonstrated across diverse substrates allows for rapid scale-up without extensive reoptimization, ensuring consistent delivery schedules even when accommodating structural modifications requested during late-stage clinical development.
- Commercial scale-up of complex intermediates: The process demonstrates excellent scalability potential as evidenced by consistent stereoselectivity across multiple substrate variations without requiring catalyst reoptimization for each new analog. The use of standard laboratory equipment and common solvents facilitates seamless technology transfer to manufacturing facilities without capital-intensive modifications to existing infrastructure. The documented tolerance to minor variations in reaction parameters provides manufacturing flexibility to accommodate batch size increases while maintaining product quality attributes within specification limits, ensuring reliable supply continuity even during periods of high demand volatility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN114524701B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
