Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Manufacturing
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust synthetic methodologies that can deliver complex heterocyclic scaffolds with high efficiency and purity. A significant breakthrough in this domain is detailed in patent CN113045503A, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These structures are pivotal in medicinal chemistry due to the enhanced metabolic stability and lipophilicity imparted by the trifluoromethyl group. The patent outlines a transition metal palladium-catalyzed carbonylation tandem reaction that utilizes readily available trifluoroethylimidoyl chloride and various amines as starting materials. This approach represents a paradigm shift from traditional cyclization methods, offering a pathway that is not only operationally simple but also highly compatible with diverse functional groups, thereby addressing critical needs for reliable pharmaceutical intermediate suppliers seeking to optimize their synthetic routes for active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl substituted quinazolinone compounds has been fraught with significant technical and economic challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Conventional literature reports typically rely on the cyclization of anthranilamide with ethyl trifluoroacetate, trifluoroacetic anhydride, or trifluoroacetic acid, often necessitating harsh reaction conditions that can degrade sensitive functional groups. Alternative strategies involving the cyclization of anthranilic acid esters with unstable trifluoroacetamides or the use of isatoic anhydride frequently suffer from low yields and narrow substrate scopes. Furthermore, methods promoted by coupling reagents like T3P, while effective in some contexts, introduce substantial cost burdens due to the expense of the reagents and the complexity of the resulting waste streams. These limitations collectively restrict the ability of procurement teams to secure cost-effective supplies of high-purity intermediates, creating bottlenecks in the development of new drug candidates.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN113045503A introduces a streamlined catalytic cycle that dramatically simplifies the construction of the quinazolinone core. By employing cheap and easily obtained trifluoroethylimidoyl chloride and amines as the foundational building blocks, this novel approach eliminates the need for pre-activation steps and expensive stoichiometric coupling agents. The reaction proceeds via a palladium-catalyzed carbonylation tandem sequence in an organic solvent such as dioxane at a moderate temperature of 110°C. This methodology not only achieves high reaction efficiency with yields often exceeding 90% for various substrates but also demonstrates exceptional substrate designability. The ability to synthesize different groups of substituted trifluoromethyl quinazolinone compounds through simple substrate modification provides medicinal chemists with a powerful tool for structure-activity relationship (SAR) studies, ensuring a reliable supply of diverse chemical entities for drug discovery programs.
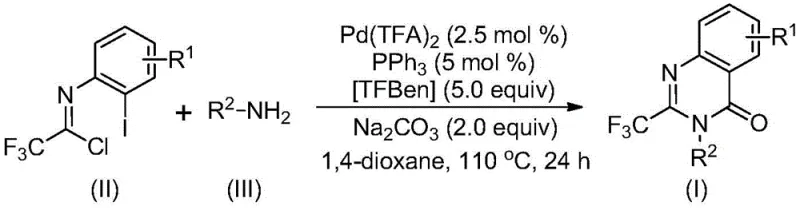
Mechanistic Insights into Palladium-Catalyzed Carbonylation Tandem Reaction
The mechanistic elegance of this transformation lies in its multi-step tandem nature, which constructs the heterocyclic ring and installs the carbonyl functionality in a single operational sequence. The reaction likely initiates with an alkali-promoted intermolecular carbon-nitrogen bond coupling between the amine and the imidoyl chloride to generate a trifluoroacetamidine derivative in situ. Subsequently, the palladium catalyst, specifically palladium trifluoroacetate coordinated with triphenylphosphine, undergoes oxidative addition into the carbon-iodine bond of the aromatic ring. A crucial step involves the release of carbon monoxide from the additive TFBen (1,3,5-tricarboxylic acid phenol ester) under heating, which then inserts into the carbon-palladium bond to form an acyl palladium intermediate. This acyl species then undergoes intramolecular cyclization promoted by the base to form a seven-membered ring palladium intermediate, followed by reductive elimination to release the final 2-trifluoromethyl-substituted quinazolinone product and regenerate the active catalyst.
From a quality control perspective, the choice of catalyst and ligand system is critical for minimizing impurity profiles. The use of palladium trifluoroacetate in conjunction with triphenylphosphine ensures high conversion rates while suppressing side reactions such as homocoupling or incomplete cyclization. The patent data indicates that this catalytic system tolerates a wide range of substituents, including halogens (F, Cl, Br), alkyl groups (methyl, tert-butyl), and electron-withdrawing groups (trifluoromethyl), without significant degradation in yield. For R&D directors focused on purity, the post-treatment procedure is equally advantageous; the reaction mixture can be simply filtered and purified via standard column chromatography. This simplicity in workup reduces the risk of introducing extraneous contaminants and facilitates the isolation of high-purity intermediates required for subsequent biological testing or further synthetic elaboration.
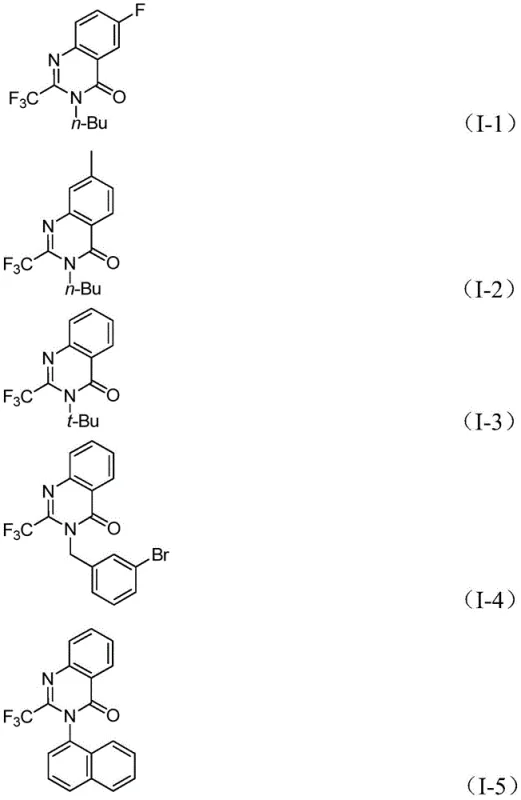
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves charging a reaction vessel with the specific molar ratios of palladium trifluoroacetate, triphenylphosphine, sodium carbonate, TFBen, trifluoroethylimidoyl chloride, and the desired amine in an aprotic solvent like 1,4-dioxane. The mixture is then heated to 110°C and stirred for a period ranging from 16 to 30 hours, depending on the specific electronic nature of the substrates. Upon completion, the reaction is cooled, filtered to remove inorganic salts and palladium residues, and the filtrate is concentrated. The crude product is then subjected to silica gel column chromatography to afford the pure target compound. For a detailed breakdown of the specific reagent quantities and step-by-step operational parameters, please refer to the standardized guide below.
- Combine palladium trifluoroacetate, triphenylphosphine, sodium carbonate, TFBen, trifluoroethylimidoyl chloride, and amine in an organic solvent like dioxane.
- Heat the reaction mixture to 110°C and stir for 16 to 30 hours to facilitate the carbonylation tandem reaction and cyclization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final 2-trifluoromethyl-substituted quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the significant cost reduction in pharmaceutical intermediate manufacturing driven by the use of commodity chemicals. Trifluoroethylimidoyl chloride and various amines are widely available in the global chemical market at competitive prices, unlike specialized activated esters or anhydrides used in older methods. Furthermore, the elimination of expensive coupling reagents like T3P drastically lowers the raw material cost per kilogram of product. The reaction conditions are mild enough to be performed in standard glass-lined reactors without requiring cryogenic cooling or extreme pressure, which simplifies equipment requirements and reduces energy consumption. These factors combine to create a highly economical process that enhances the overall margin profile for downstream API production.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the substitution of high-cost reagents with inexpensive, bulk-available starting materials. By utilizing a catalytic amount of palladium rather than stoichiometric amounts of activating agents, the method minimizes the generation of heavy metal waste and reduces the cost associated with waste disposal and catalyst recovery. Additionally, the high atom economy of the carbonylation step ensures that a greater proportion of the input mass is converted into the desired product, thereby reducing the effective cost of goods sold (COGS). This efficiency allows manufacturers to offer more competitive pricing for complex heterocyclic intermediates without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on chemically stable and commercially ubiquitous raw materials. Since trifluoroethylimidoyl chloride and the requisite amines are produced by multiple suppliers globally, the risk of supply disruption due to single-source dependency is mitigated. The robustness of the reaction conditions also means that the process is less susceptible to variations in utility availability, such as fluctuations in cooling water temperature or power supply, ensuring consistent batch-to-batch production. This reliability is crucial for maintaining continuous manufacturing schedules and meeting the stringent delivery timelines demanded by multinational pharmaceutical clients.
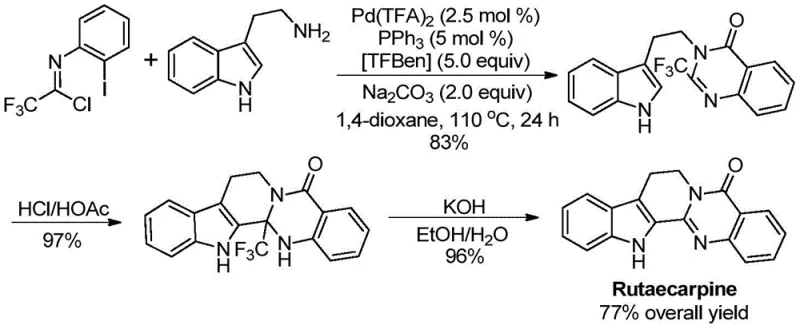
- Scalability and Environmental Compliance: The scalability of this synthesis is evidenced by its successful application in the gram-level preparation of diverse derivatives and the total synthesis of Rutaecarpine. The straightforward workup procedure, involving simple filtration and chromatography, translates well to larger scales where complex extraction protocols can become logistical nightmares. Moreover, the use of dioxane as a solvent, while requiring careful handling, is a well-established industrial solvent with established recovery and recycling protocols. The reduction in hazardous waste generation compared to traditional methods aligns with increasingly stringent environmental regulations, facilitating smoother regulatory approvals and enhancing the sustainability profile of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in patent CN113045503A, providing clarity on the method's versatility and practical application in industrial settings. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this Pd-catalyzed method over traditional quinazolinone synthesis?
A: Unlike conventional methods requiring harsh conditions or expensive activating agents like T3P, this patent utilizes cheap trifluoroethylimidoyl chloride and amines under mild conditions (110°C), achieving significantly higher yields and broader substrate compatibility.
Q: Can this synthesis route be applied to complex drug molecules like Rutaecarpine?
A: Yes, the patent explicitly demonstrates the successful application of this methodology in the high-yield total synthesis of Rutaecarpine, achieving an overall yield of 77% through a streamlined three-step process involving cyclization and hydrolysis.
Q: Is the catalyst system suitable for large-scale industrial production?
A: The method employs commercially available palladium trifluoroacetate and triphenylphosphine with simple post-treatment procedures like filtration and chromatography, indicating strong potential for scalability and cost-effective manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN113045503A for accelerating drug development timelines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest industry standards. We are committed to leveraging our technical expertise to support your R&D initiatives and commercial manufacturing needs with unmatched precision and reliability.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to this efficient catalytic process can impact your bottom line. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us be your partner in turning complex chemical challenges into commercial successes.
